By David Bautz, PhD
OBB:PIRS
Financial Update
On November 11, 2015, Pieris Pharmaceuticals, Inc. (OBB:PIRS) announced financial results for the third quarter of 2015 and provided a business update. The company reported revenues of $0.4 million, which was mainly comprised of a milestone payment received during the quarter. Net loss for the quarter was $3.9 million, or $0.10 per share, and was comprised of $2.1 million of R&D and $2.2 million of G&A expenses. Total cash burn for the quarter was $3.7 million.
The company exited the third quarter of 2015 with $32.3 million in cash and cash equivalents. In July 2015, the company raised gross proceeds of $28.3 million through a public offering of 9,090,909 shares of common stock at a price of $2.75 per share and an additional 1,211,827 shares pursuant to the over-allotment option exercised by the underwriters. Oppenheimer & Co. and JMP Securities acted as joint book-running managers for the offering. Roth Capital Partners and Trout Capital acted as co-managers for the offering. We anticipate the current cash position will be enough to fund operations for at least the next 12 months.
Business Update
Pieris is developing the Anticalin® class of biotherapeutic compounds targeting a wide range of medical conditions. Anticalins are a new class of novel, engineered proteins that are based on lipocalins, which are naturally-occurring proteins with variable loop regions supported by a rigid framework that bind a wide array of compounds (just like antibodies). Pieris scientists have developed a means of modifying the lipocalins to confer unique binding specificities against virtually any biological target, and dubbed the new compounds ‘Anticalins®’ (antibody + lipocalin). The company is currently developing PRS-080, which targets hepcidin for the treatment of functional iron deficiency in patients with chronic kidney disease; PRS-060, which targets the interleukin 4 receptor (IL4Ra) for the treatment of asthma; and PRS-343, an immuno-oncology product that works by “stepping on the gas” to ramp up an immune response in the tumor microenvironment.
PRS-080 Update
On November 5, 2015, Pieris Pharmaceuticals, Inc. (PIRS) announced the presentation of Phase 1 clinical data for PRS-080 at the 2015 American Society of Hematology (ASH) annual meeting. The abstract, entitled “A Phase I Study Investigating Safety, Tolerability, Pharmacokinetics and Pharmacodynamic Activity of the Hepcidin Antagonist PRS-080#022. Results from a Randomized, Placebo Controlled, Double-Blind Study Following Single Administration to Healthy Subjects” will be presented on Monday, December 7, 2015.
PRS-080 is an Anticalin® that targets hepcidin, a 25 amino acid peptide produced in the liver and excreted into the bloodstream where it binds to ferroportin, an iron transporter found on duodenal enterocytes, reticuloendothelial macrophages, and hepatocytes (Donovan et al., 2006). Binding of hepcidin to ferroportin induces its internalization and degradation, resulting in iron accumulation in enterocytes, macrophages, and the liver. Hepcidin expression is induced by inflammatory cytokines, which is hypothesized to result in the sequestration of iron from invading pathogens (Babitt et al., 2010). This increased hepcidin expression results in many of the hallmarks of chronic diseases, including iron sequestration, hypoferremia, and anemia.
Pieris is developing PRS-080 as a treatment for functional iron deficiency (FID) in anemic patients with chronic kidney disease (CKD), which is a progressive loss of kidney function over a period of months or years. FID in CKD patients is characterized by low circulating iron levels that limits erythropoiesis even when these patients have sufficient iron stores in their body. These patients have low serum transferrin (a measure of circulating iron) and normal or high serum ferritin (a marker of body iron stores). FID is characteristic of a chronic inflammatory state, which in CKD patients may be due to an increased incidence of infections and/or the induction of inflammatory cytokines by the hemodialysis procedure.
The data to be presented at ASH 2015 is from a placebo controlled, double blind, Phase 1 study with ascending doses of PRS-080 in six cohorts of healthy volunteers of 0.08, 0.4, 1.2, 4.0, 8.0, and 16.0 mg/kg (NCT02340572). Six patients in each cohort received drug while two patients received placebo through a two hour intravenous infusion. A total of 39 adverse events (AEs) were reported during or after treatment in 22 subjects. All AEs were mild or moderate and there were no serious AEs reported. Importantly, there were no infusion reactions or hypersensitivities noted during the study. In addition, no anti-drug antibodies were detected from participants treated with PRS-080.
A decrease in free hepcidin was noted beginning one hour after administration of PRS-080 along with a transient increase in serum iron concentration and transferrin saturation. Serum iron concentrations reached approximately 50 μM in individual subjects but did not increase further with increasing dose. The normal range for serum iron concentration is approximately 11-30 μM. We believe this is a very important data point to come out of this trial, because if PRS-080 is able to increase normal iron levels to above normal, it stands to reason that it should be able to increase anemic patients iron levels to within the normal range.
The company is planning to move quickly into a Phase 1b clinical trial of PRS-080 in anemic patients with chronic kidney disease, which should commence before the end of 2015. Data from that study should be available in the second half of 2016.
PRS-060 Update
PRS-060 is an Anticalin® that has very high affinity for IL4Ra (Kd ~ 20 pM) and does not bind to the related cytokine receptors IL6R, IL18R, and IL23R. By targeting IL4Ra, PRS-060 inhibits binding of both IL-4 and IL-13, as depicted in the following graphic.
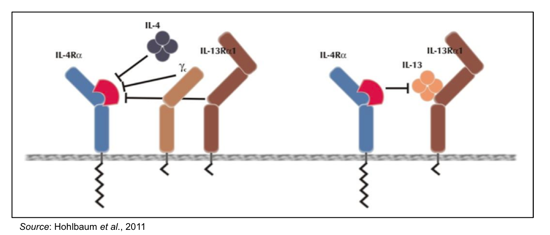
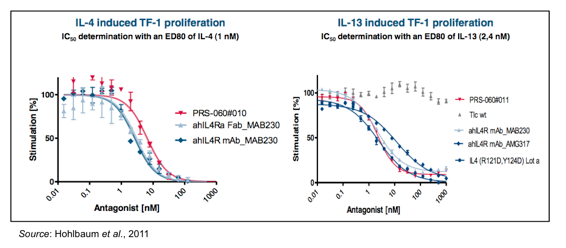
The inhibition of IL-4 and IL-13 binding by PRS-060 was shown in a series of preclinical experiments involving erythroleukemic (TF-1) cells that proliferate upon stimulation with a wide range of cytokines. The cells were preincubated for 30 min with PRS-060 or other IL4Ra antagonists prior to stimulation with IL-4 (left) or IL-13 (right). Cell proliferation was measured five days after treatment and graphed as a function of increasing amounts of IL4Ra antagonist. The results show that PRS-060 is able to inhibit both IL-4 and IL-13 dependent proliferation.
One of the advantages that Anticalins® have is their mode of delivery can be modified depending upon the therapeutic target. In the case of PRS-060, which is targeting IL4Ra expression in the lung, inhalation delivery would be optimal as it is likely to decrease the systemic side effects that could arise from a chronic, systemic Th2-targeting medication. Pieris has conducted inhalation studies with PRS-060 in mice and determined that systemic concentration of PRS-060 is minimal when dosed by inhalation. Since most other asthma medications are dosed by inhalation, the ability to dose PRS-060 by inhalation represents a more convenient dosing regimen than monoclonal antibody treatments, which must be administered by injection at a doctor’s office or other medical clinic.
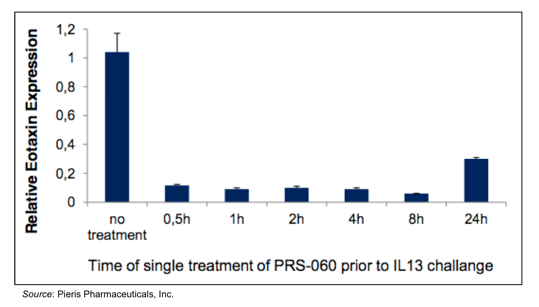
Efficacy of inhaled PRS-060 is shown in the following graphic, in which eotaxin expression (a marker of airway inflammation) was induced by administration of IL-13 to the lungs of mice after pre-treatment with inhaled PRS-060. The data shows that eotaxin expression is inhibited for up to 24 hours following a single inhaled dose of PRS-060.
Currently, PRS-060 is still in preclinical development with the final IND enabling studies being conducted in 2015 in preparation for an IND filing in 2016. Earlier in 2015, Pieris announced the receipt of a research grant in collaboration with the University of Melbourne to advance PRS-060 through preclinical studies for inhaled delivery to treat asthmatics. The lead investigator on that grant, Professor Gary Anderson, PhD, recently presented preclinical data on PRS-060 that included an overview of pharmacokinetic analysis and data demonstrating concentration-dependent antagonism of IL4Ra-mediated signaling and very low in silico predicted immunogenicity.
The current clinical plan calls for Pieris to test PRS-060 in healthy volunteers and then in patients with severe asthma. The goal of these pilot studies is two-fold: 1) the company could establish proof-of-concept for inhaled Anticalins®, which would provide evidence for an additional mode of administration for the company’s products outside of subcutaneous and intravenous injections, the way that almost all biological therapies are administered now, and 2) establish that dosing of inhaled Anticalins® is practical and that the compound has the requisite pharmacokinetic/pharmacodynamic profile to be utilized as a therapeutic capable of positively impacting patients symptoms. We estimate that Phase 1 data for PRS-060 could be available in 2017.
PRS-343 Update
PRS-343 is a bispecific compound that contains a CD137-specific Anticalin® genetically linked to a HER2-specific monoclonal antibody, and is the lead compound from the company’s immuno-oncology program. CD137 (4-1BB, TNFSFR9) is a tumor necrosis factor receptor (TNFR) family member with costimulatory function that regulates T-cell proliferation and survival and is expressed on activated CD8+ and CD4+ T-cells (Watts, 2005). Since CD8+ T-cells play a pivotal role in tumor immunity, enhancing CD137 costimulation could promote the generation of protective antitumor immune responses. This idea has been validated through enhanced immunogenicity in tumor cells engineered to express CD137 ligand (Mogi et al., 2000) or anti-CD137 antibodies (Ye et al., 2002). Unfortunately, the use of systemic anti-CD137 antibodies can result in severe side effects such as liver toxicity (Grade 4 hepatitis), which resulted in the cancellation of a Phase 1 clinical trial (Yao et al., 2013). Thus, the ability to stimulate CD137 in the tumor microenvironment could mitigate these systemic toxic effects.
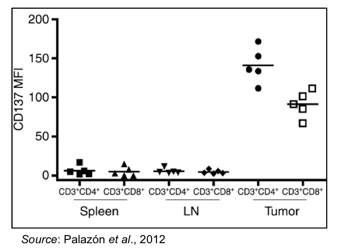
Another important aspect of CD137 is that it is preferentially expressed in high levels on intratumoral T-cells (Palazón et al., 2012). This is shown in the following graph, where CD4+ and CD8+ T-cells were extracted from the spleen, lymph nodes, and tumor of mice bearing CT26 tumors. Only T-cells purified from tumors showed expression of CD137, whereas T-cells from other locations from the same mice did not show increased expression of CD137.
These data were further corroborated in tissue samples from patients with ovarian cancer (Ye et al., 2014). Results from that study showed that tumor-infiltrating lymphocytes (TILs; found in the tumor microenvironment) and tumor-associated lymphocytes (TALs; found surrounding the tumor microenvironment) naturally expressed higher levels of CD137 than circulating T cells. In addition, CD137-expressing TILs, but not PD-1 positive or PD-1 negative CD137-negative cells, possessed autologous tumor reactivity both in vitro and in vivo. All told, there is significant data to support the use of CD137-targeting therapeutics as anti-cancer agents.
Pieris has done a number of studies on different CD137-specific Anticalin’s® to identify the lead molecule to advance into clinical testing. The company revealed that this lead compound has a very high affinity for CD137 (~2 nM), activates T-cells in an ex vivo model, and binds to CD137 in a “non-competitive fashion”. What this means is that binding of the Anticalin® to CD137 does not interfere with the binding of CD137 and its natural ligand. The flexibility of the Anticalin® platform is exemplified in the following graphic, which shows that no matter how the Anticalin® is attached to the monoclonal antibody, both entities retain the ability to bind to their respective ligands with essentially the same affinity.
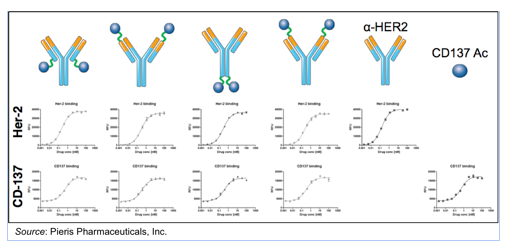
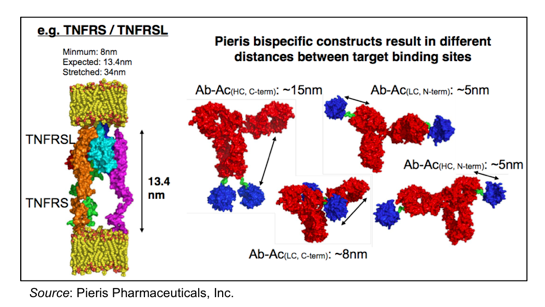
Another advantage of the flexibility of the Anticalin® platform is that the bispecific geometry of the molecule can be manipulated to create different pharmacodynamic effects. This is shown in the following graphic, where multiple distances between HER2 and CD137 binding can be tested to determine the optimal target synapse. Preclinical work such as this is ongoing, with selection of the drug candidate to advance into clinical testing most likely being completed by the end of 2015.
The company has made presentations on PRS-343 at multiple scientific conferences this fall including at CRI-CIMT-EATI-AACR Inaugural International Cancer Immunotherapy Conference, the Protein Engineering Summit Europe (PEGS), the Society for Immunotherapy of Cancer (SITC), and the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics. An overview of the data presented at CRI-CIMT-EATI-AACR can be found here. The following are key takeaways from the data presented thus far for PRS-343:
1) The anti-CD137 Anticalin® created by Pieris is a very stable protein that binds with high affinity to CD137 in a non-competitive fashion, which allows the interaction between CD137 and its natural ligand to still take place.
2) PRS-343 is a very stable molecule where both components retain their native binding affinities no matter how the Anticalin® is attached to the antibody.
3) The engineered IgG4 backbone of the monoclonal antibody component of PRS-343 does not bind to either FcγRI or FcγRIII, and retains the ability to bind to FcRN.
4) How the anti-CD137 Anticalin® is attached to the anti-HER2 monoclonal antibody influences the ex vivo activity of PRS-343, as some of the configurations result in T-cell activation (as measured by release of INF-γ and IL-2) and some do not.
Conclusion and Recommendation
We initiated coverage of Pieris in April 2015, and just since that time the company has achieved a number of important milestones, including 1) uplisting to the Nasdaq; 2) improving the balance sheet; and 3) announcing positive results from the Phase 1 study of PRS-080. However, the stock price has not appreciably increased since that time. We believe this will change as investors come to realize the potential of the Anticalin® platform, particularly with the positive data attained in the Phase 1 study of PRS-080.
The wide range of diseases being targeted by Pieris is indicative of the flexibility and adaptability of the Anticalin® platform, and could make Pieris highly sought after by larger biopharmaceutical companies over the next few years, both as a partner or an acquisition candidate. A number of companies with novel protein-binding platforms (most of which have been various monoclonal antibody technologies) have either been acquired or signed lucrative partnership deals over the past few years, as shown in the following table.
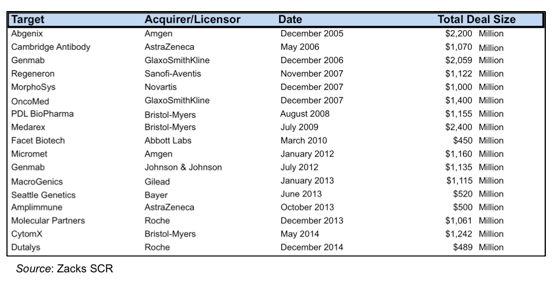
One example from the chart above that really stands out is that of Micromet, which Amgen acquired in January 2012 for $1.1 billion. Micromet was a German company that reverse merged with CancerVax Corporation in 2006. The company developed a novel antibody technology known as Bispecific T-cell engagers (BiTE) that work by binding both a tumor associated antigen and a T-cell receptor. The FDA approved Blinatumomab (BLINCYTO®), a BiTE compound that treats acute lymphoblastic leukemia, in December 2014.
In the first half of 2008, Micromet had a market cap of approximately $80 million. Approximately 4 years later, it was acquired for $1.1 billion, a greater than 13-fold return for investors who got in early. Pieris is a German company that went through a reverse merger and has a novel protein-binding technology that includes a bispecific compound that targets a tumor associated antigen and a T-cell receptor. Pieirs’ current market cap is approximately $80 million. Whether the Pieris story will have the same ending as Micromet’s remains to be seen, but we believe that the similarities are too great to not consider.
We are very optimistic about the future of Pieris and believe that the Anticalin® platform is a transformative technology that could lead to a number of novel treatments in a variety of diseases. We recently increased our price target based upon the positive data released in the abstract for the Phase 1 clinical trial of PRS-080 that will be presented in December. We believe the shares are worth $8 and we continue to believe that investors with a longer-term horizon will be handsomely rewarded by an investment in Pieris.
READ THE LATEST FULL RESEARCH REPORT HERE
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR and to view our disclaimer.