By David Bautz, PhD
NASDAQ:VKTX
NASDAQ:LGND
On December 7, 2015, Viking Therapeutics, Inc. (NASDAQ:VKTX) announced new preclinical data on the effect of VK5211 on weight gain in cynomolgus monkeys after 13 weeks of treatment. The results showed progressive weight gain throughout the course of treatment, with a substantial portion of the weight gain maintained after treatment was stopped. The results were presented at the 8th International Conference on Cachexia, Sarcopenia, and Muscle Wasting in Paris, France.
VK5211
Viking’s lead drug candidate is VK5211 is a third-generation non-steroidal Selective Androgen Receptor Modulator (SARM) that is being developed for maintenance or improvement of lean body mass (LBM), bone mineral density (BMD), and function in patients recovering from non-elective hip fracture surgery. Hip fracture is associated with a number of morbidities, the majority of which are the result of deleterious changes in body composition following the injury. In the first year after a hip fracture, fat mass increases by up to 7% (Karlsson et al., 1996) while lean mass decreases by up to 11% (Fox et al., 2000). This is in comparison to healthy older females who lose approximately 1% of lean mass per year and gain approximately 1.7% in fat mass (Karlsson et al., 2000).
SARMs are a group of compounds designed to act as androgen receptor (AR) agonists in muscle and bone while being partial agonists in other areas of the body (e.g., prostate). The most prominent androgen, testosterone, stimulates the growth of muscle and bone (anabolic effects) as well as the prostate and sebaceous glands (androgenic effects), and is considered a non-tissue-selective androgen.
While androgens inhibit fat accumulation and increase skeletal muscle growth, two properties that make them ideal therapeutic candidates for restoring or preserving body composition following hip fracture, the use of testosterone therapy has a number of side effects including prostate growth (Meikle et al., 1997) and polycythemia (Snyder et al., 2000) in men and acne, alopecia, and hirsutism in women (Phillips et al., 1997) that precludes its use in a large number of patients. Thus, what would be most beneficial would be a product that provided the anabolic effects of androgen therapy with limited androgenic effects.
VK5211 (then known as LGD-4033) was originally developed by Ligand Pharmaceuticals, Inc. (NASDAQ:LGND) and was previously tested in preclinical models and early stage clinical trials. Two Phase 1 clinical trials showed the drug to be safe and well-tolerated at all doses following daily oral administration for up to 21 days. The drug selectively activates AR in muscle and bone, stimulating muscle and bone growth, while avoiding undesirable side effects, such as unwanted hair growth, acne, or prostate growth.
Preclinical Primate Data
To examine the safety and physiological changes that occur after 13 weeks of VK5211 dosing, cynomolgus monkeys were orally administered VK5211 once daily at 0, 0.6, 3, 15, or 75 mg/kg. The following figure shows a significant increase in body weight in both male and female monkeys during the 13 weeks of dosing. The fact that the results were seen in females is important, as the majority of hip fractures occur in females.
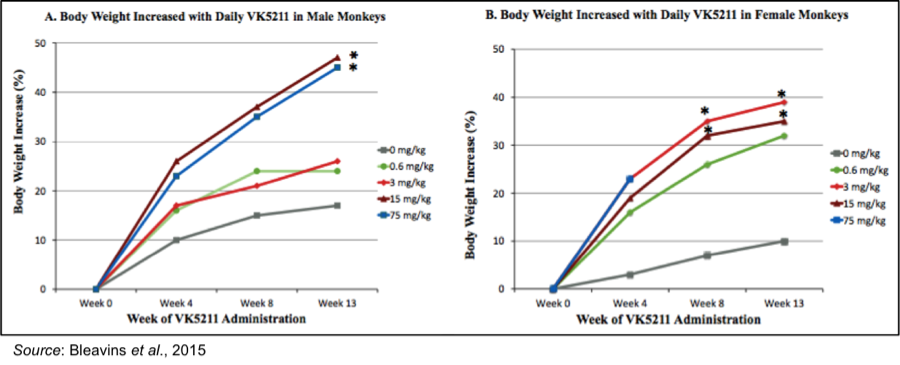
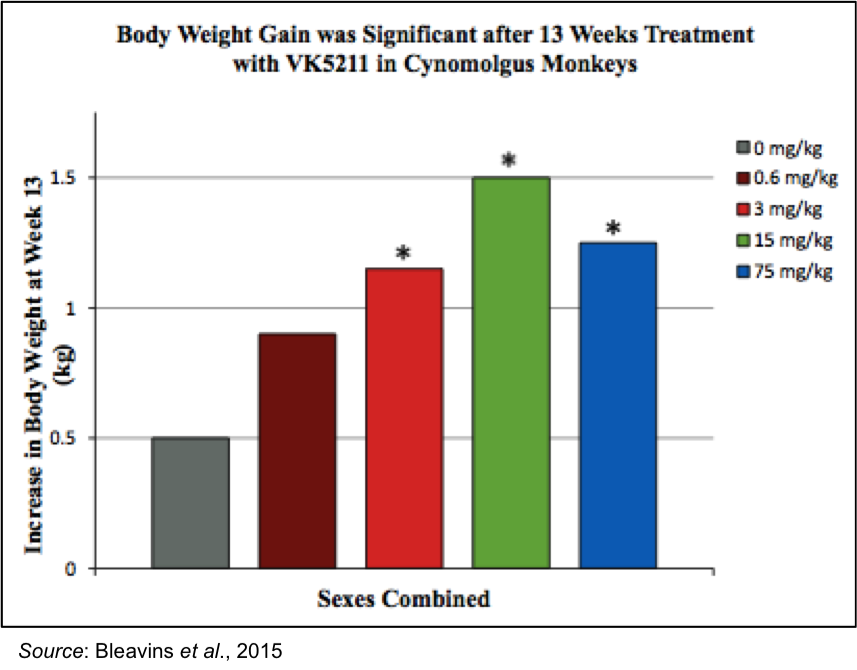
At the end of 13 weeks of dosing, there was a significant increase in body weight for the monkeys dosed with 3 and 15 mg/kg. The 75 mg/kg dosing was halted after 48 days due to signs of toxicity, however this is not a concern for the ongoing Phase 2 clinical trial as that dose is well above the 0.5, 1.0, and 2.0 mg doses being administered. In addition, 70% of the increased weight was retained after a four-week recovery period.
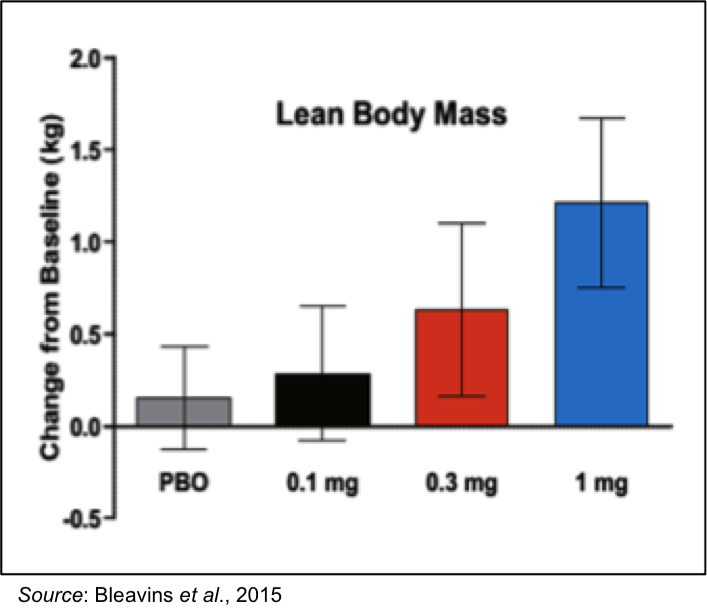
The primate data compares quite well with previously disclosed data from a Phase 1 multiple ascending dose clinical trial in 76 healthy men that received daily doses of placebo, 0.1 mg, 0.3 mg, or 1 mg of VK5211 for 21 consecutive days (Basaria et al., 2013). Those results showed a dose dependent increase in LBM and no serious adverse events.
VK5211 Clinical Trial Underway
Viking recently initiated a Phase 2 study in patients ≥ 65 years of age who have suffered a hip fracture within the past three to seven weeks. This is a multicenter, randomized, parallel group, double blind, placebo controlled trial, where patients will be administered placebo or 0.5 mg, 1.0 mg, or 2.0 mg of VK5211 once-daily for 12 weeks (NCT02578095). A total of 120 patients are expected to enroll in the trial evenly split between the four treatment groups. The primary outcome of the trial is the change in LBM after 12 weeks of treatment. Secondary and exploratory endpoints include assessments of functional performance, quality-of-life, and activities of daily living. We anticipate topline results being available in the second half of 2016.
Conclusion
The primate data for VK5211 is quite encouraging, particularly the magnitude of the effect, the durability of response, and the lack of any serious adverse events. The company now has a considerable amount of data to support the development of VK5211 in maintaining body mass following hip fracture, and we are looking forward to analyzing the results from the ongoing Phase 2 clinical trial when they are reported, which should occur in the second half of 2016.
In addition to VK5211, Viking is also developing VK2809 for the treatment of hypercholesterolemia and NASH, and VK0214 for the treatment of adrenoleukodystrophy (ALD). The company recently announced the filing of an Investigational New Drug (IND) application with the FDA such that a Phase 2 clinical trial of VK2809 can be performed in patients suffering from hypercholesterolemia and fatty liver disease. Results from that study should be available at the end of 2016 or first part of 2017. For ALD, the company recently announced a research collaboration with the Kennedy Krieger Institute to evaluate VK0214 in in vivo models of ALD, which will support the initiation of IND-enabling studies, with the goal of getting VK0214 into clinical trials in 2017.
We initiated coverage of Viking a few weeks ago, and we invite investors to look over the full report, which can be found here. We believe that Viking’s products represent potential best-in-class treatments due to their unique mechanisms of action and strong safety profile in early clinical testing. With a market cap of less than $30 million, we believe the market does not fully appreciate the multiple billion-dollar opportunities Viking is pursuing, thus representing a compelling investment opportunity. We have assigned a ‘Buy’ rating to the shares with a price target of $15.
READ THE LATEST FULL RESEARCH REPORT HERE
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR and to view our disclaimer.