By David Bautz, PhD
NASDAQ:BCLI
On July 18, 2016, BrainStorm Cell Therapeutics (NASDAQ:BCLI) announced results from the company’s Phase 2 clinical trial of NurOwn™ in patients with amyotrophic lateral sclerosis (ALS). The data show that patients treated with NurOwn™ were more likely to see a decrease in the rate of decline as measured by the Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS-R) compared to those treated with placebo. Perhaps most importantly, the greatest benefit was seen in a subset of patients with faster-progressing disease (i.e., those whose health was deteriorating the most). Below is a brief background on ALS, NurOwn™, the Phase 2 clinical trial, and a discussion of the results.
Background on ALS
ALS is a rapidly progressing neurodegenerative disease whereby the nerve cells in the brain and spinal cord that control muscle movement degenerate. As the disease progresses, all patients will experience increased difficulty swallowing and speaking, with most patients not able to use their arms or legs. Eventually, patients in later stages of the disease may become completely paralyzed, which includes losing the ability to control their breathing. Patients typically do not live more than three to five years after being diagnosed with the disease, although there is considerable variability in disease progression from one patient to another. In the U.S., approximately 30,000 people are currently living with ALS.
The rate at which disease progression occurs is measured utilizing a scoring system called the “ALS Functional Rating Scale Revised” (ALSFRS-R; Cedarbaum et al., 1999). The ALSFRS-R measures gross motor tasks, fine motor tasks, bulbar functions, and respiratory functions through a scoring system consisting of a series of 12 questions on basic tasks (speech, salivation, swallowing, handwriting, cutting food, dressing and hygiene, turning in bed, walking, climbing stairs, dyspnea, orthopnea, and respiratory insufficiency) that are rated on a five-point scale where 0 = can’t do and 4 = normal ability. The individual items are summed to produce a score of between 0 = worst and 48 = best.
There are only a limited number of treatment options available for ALS patients, which are mostly designed to relieve symptoms and improve quality of life. Sanofi’s Riluzole® (rilutek) is the only treatment shown to improve survival, but only for two to three months, and it does not reverse nerve damage that has already occurred. Sales of Riluzole® peaked at around $50 million per year. It is now available as a generic.
NurOwn™
BrainStorm is developing adult stem cells therapies for the treatment of a range of neurodegenerative diseases. The company has a proprietary process called NurOwn™ that harvests and propagates autologous Mesenchymal Stem Cells (MSC) and then induces their differentiation into neurotrophic factor (NTF) secreting cells, called MSC-NTF. The cells are then returned to the patient at or near the target area for treatment. Because these cells are autologous, there is virtually no risk of rejection or tumor formation. Below is a graph showing the dramatic increase in various neurotrophic factors secreted by NurOwn™ cells (red) compared to normal mesenchymal stem cells (blue).
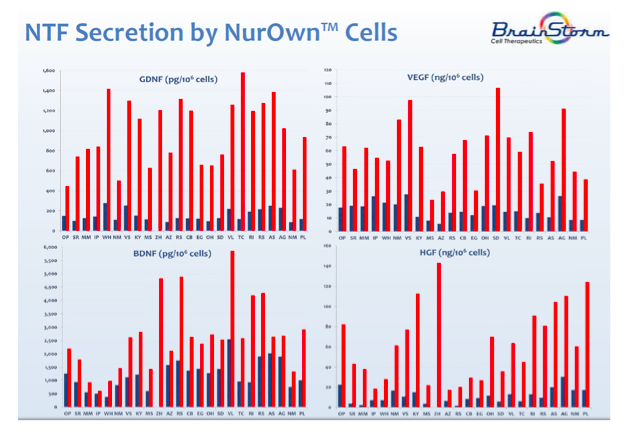
BrainStorm previously conducted a Phase 1/2 and a Phase 2a clinical trial of NurOwn™, both of which took place at Hadassah Medical Center between June 2011 and October 2014. All patients in the trials had a three-month run-in period, were treated with NurOwn™, and then evaluated during a six-month follow-up time. The Phase 1/2 trial consisted of 12 patients (six patients received NurOwn™ intramuscularly (IM) while six patients received NurOwn™ intrathecally (IT)) and the study met its primary endpoints of safety and tolerability, with no treatment-related adverse events reported. The Phase 2a study consisted of 14 patients that received both IM and IT injections of NurOwn™. The results from these studies were published in January 2016 in the journal JAMA Neurology and discussed in a previous Seeking Alpha article.
Phase 2 Study of NurOwn™
BrainStorm conducted a randomized, double blind, placebo controlled Phase 2 clinical trial to evaluate the safety and efficacy of a single dose of NurOwn™ in early-stage ALS patients (NCT02017912). The trial took place at three centers in the U.S. (Massachusetts General Hospital (MGH), University of Massachusetts Memorial Hospital, and the Mayo Clinic) and enrolled 48 patients randomized 3:1 to receive either NurOwn™ cells (n=36) or placebo (n=12).
Just as with the company’s Phase 2a trial, there was a three-month run-in (to determine the rate of decline in ALSFRS-R prior to treatment), followed by treatment and a six-month follow up. Patients were evaluated at two, four, eight, twelve, sixteen, and twenty-four weeks after treatment. The primary endpoint of the study was safety based on the number of patients with adverse events, with secondary efficacy endpoints including the change in ALSFRS-R.
Along with showing NurOwn™ was safe and well tolerated, the study also achieved a number of secondary efficacy endpoints. The results presented by BrainStorm are all in the context of a 2010 survey of ALS clinicians and researchers that showed all participants felt a 25% change in decline of the ALSFRS-R score would be at least somewhat clinically meaningful, and 93% of participants felt a 50% change in the decline of the ALSFRS-R score would be very clinically meaningful (Castrillo-Viguera et al., 2010).
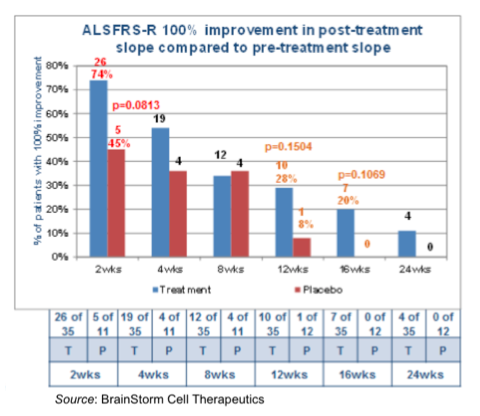
The following chart shows the number of patients that achieved 100% improvement in the post-treatment decline in ALSFRS-R compared to before treatment. A 100% improvement means that a patient had to have either stable disease or an improvement in ALSFRS-R score to count as a responder. All timepoints except for 8 weeks showed a greater percentage of NurOwn™ treated patients as responders compared to placebo. Of particular interest is that at 16 weeks post-treatment, 20% of those receiving NurOwn™ had stable disease (or improvement) compared to 0% of those receiving placebo.
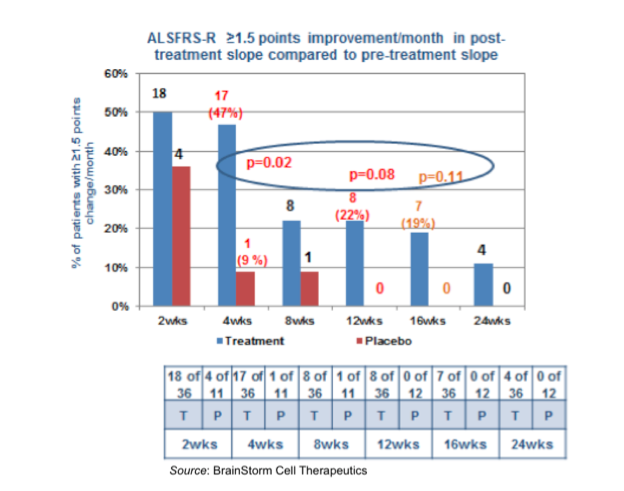
When examining the data based on an absolute point improvement in ALSFRS-R slope, again patients treated with NurOwn™ appeared to fare better than those treated with placebo. At four weeks post treatment there was a statistically significant difference in responders between NurOwn™ and placebo treated patients (p=0.03). This effect was also pronounced at 12 weeks post-treatment, where approximately 22% of those treated with NurOwn™ had a ≥1.5 point improvement/month in ALSFRS-R slope compared to 0% of those treated with placebo.
It is well known that there is a high degree of variability in the progression of ALS, which is exemplified in the following graph showing the decrease in ALSFRS for 30 randomly selected ALS patients from a clinic in Belgium (Swinnen et al., 2014).
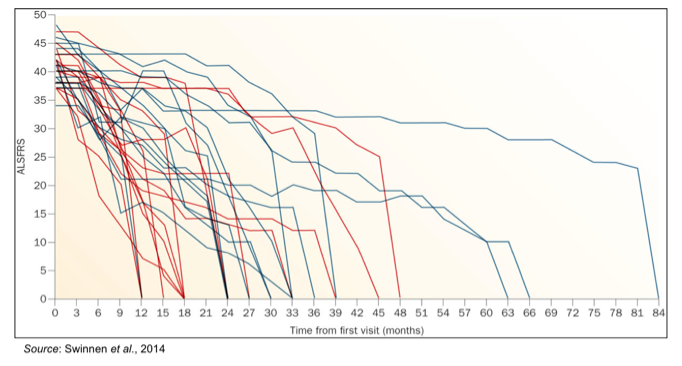
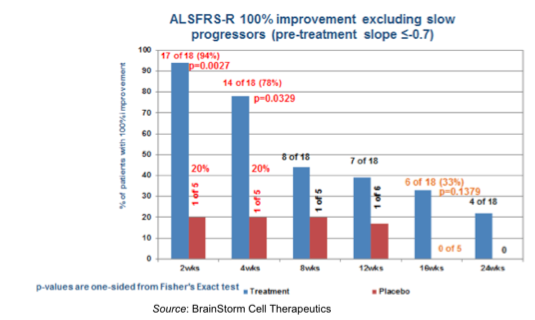
What is apparent from the above graph is that the progression of the disease is not constant, as most patients appear to progress slowly for some time before disease progression begins to increase. Thus, patients could be considered as “slow progressors” or “fast progressors” based upon the monthly change in ALSFRS-R. BrainStorm performed a subset analysis in patients with faster progressing disease (defined as an ALSFRS-R slope of >-0.7 points/month during the pre-treatment phase), as -0.7 point/month was the average rate of decline for patients in the study during the pre-treatment period.
The differences between NurOwn™ and placebo-treated patients were most stark when examining those patients who had 100% improvement in ALSFRS-R slope change following treatment. One month post treatment, 78% (14/18) of NurOwn™ treated patients (who had previously had steadily progressing disease with a decrease in ALSFRS-R slope of >-0.7 points/month) saw their progression stabilize or improve compared to only 20% (1/5) of placebo treated patients. Even out to 16 weeks, 33% (6/18) of NurOwn™ treated patients continued to be stable, compared to 0% of the placebo treated patients. While the numbers are small, these results are still very encouraging, and point to the necessity for a multi-dose trial to determine if additional doses of NurOwn™ could extend the amount of time that these patients see disease stabilization.
In order to better understand the biological effect of NurOwn™ cells, 35 cerebrospinal fluid (CSF) samples were collected both pre- and post-treatment with NurOwn™ to examine the levels of neurotrophic and inflammatory factors. The company reported that there was a statistically significant increase in levels of vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF) following treatment with NurOwn™ cells in the patients that responded to treatment. There was also a statistically significant decrease in inflammatory markers in patients treated with NurOwn™ that was not seen in those treated with placebo.
Conclusion
In addition to showing that NurOwn™ treatment was safe and well tolerated, the results from the Phase 2 clinical trial were described by one investigator as showing “clinically meaningful beneficial response” and another was “very encouraged and excited by the trial results” having conducted clinical studies in ALS for 30 years. Thus, it appears as though clinicians who work with ALS patients are excited about the data.
In our opinion, the most encouraging data may have to do with the cohort of patients whose disease was progressing the fastest in the pre-treatment period (a decrease in ALSFRS-R slope of >-0.7 points/month), as these are the patients most in need of an effective treatment and who appeared to respond best to NurOwn™ treatment. The fact that BrainStorm has identified this cohort as being most responsive to treatment with NurOwn™ will aid in designing future clinical trials, as the company can specifically target this group of patients, with one possibility being that those early in the disease would not receive treatment with NurOwn™ until their disease begins to progress more rapidly.
These results are also highly supportive of the notion that multiple doses of NurOwn™ will be necessary to see an extended clinical benefit. The company is conducting a multi-dose clinical trial of NurOwn™ in Israel, in which 24 patients will be treated with three consecutive doses of the drug in order to explore the safety and efficacy of a multi-dose treatment. The company is also planning to meet with the FDA in order to plan a placebo controlled, randomized, double blind, multi-dose clinical trial in the U.S. and Israel.
From a valuation standpoint, we continue to believe BrainStorm’s shares remain highly attractive. Before becoming generic, Riluzole® cost $50,000 per year and was shown to only extend survival by two to three months. The results shown above indicate that NurOwn™ may be able to slow down the progression of ALS, and for some patients it may even lead to disease stabilization. Multiple doses of NurOwn™ could increase these effects. As such, we currently model for NurOwn™ to cost $100,000 per year. We have also increased the probability of success to 40% based upon the results of the Phase 2 trial, and continue to believe that upon approval NurOwn™ could generate peak revenues of $1.6 billion. Our current valuation for BrainStorm is $12 per share.
READ THE FULL RESEARCH REPORT HERE
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR and to view our disclaimer.