By John Vandermosten, CFA
OTC:BIOAF
READ THE FULL BIOAF RESEARCH REPORT
Initiating Coverage
We are initiating coverage of Bioasis Technologies Inc. (OTC: BIOAF) with a current valuation of $0.601per share. This present value is based on our estimates for development of the xB3 platform in multiple indications. Bioasis is advancing several candidates in its pipeline, including one in the IND-enabling phase that we expect to enter the clinic by the end of next year. The company has developed an amino acid sequence derived from a human protein called p97 or melanotransferrin that is able to deliver biologics and oligonucleotides across the blood brain barrier (BBB) without impacting the efficacy of the payload.
Key reasons to own Bioasis shares:
‣ Best in class xB3 blood brain barrier penetrating technology
◦ Non-transferrin based transcytotic pathway
‣ Addresses a major hurdle to CNS therapy
‣ High selectivity for BBB and CNS parenchyma
‣ Preserves payload function and pharmacodynamics
‣ Can deliver antibodies, enzymes, siRNA and small molecules
‣ Lead indications in HER2+ metastatic breast cancer and Gaucher Disease
‣ Broad licensing and funding opportunities with partners
◦ Chiesi Group
◦ Prothena Corporation
◦ Leading pharma company
‣ Diversified preclinical portfolio able to address multiple CNS disorders
‣ 120+ patents and pending applications across 10+ patent families
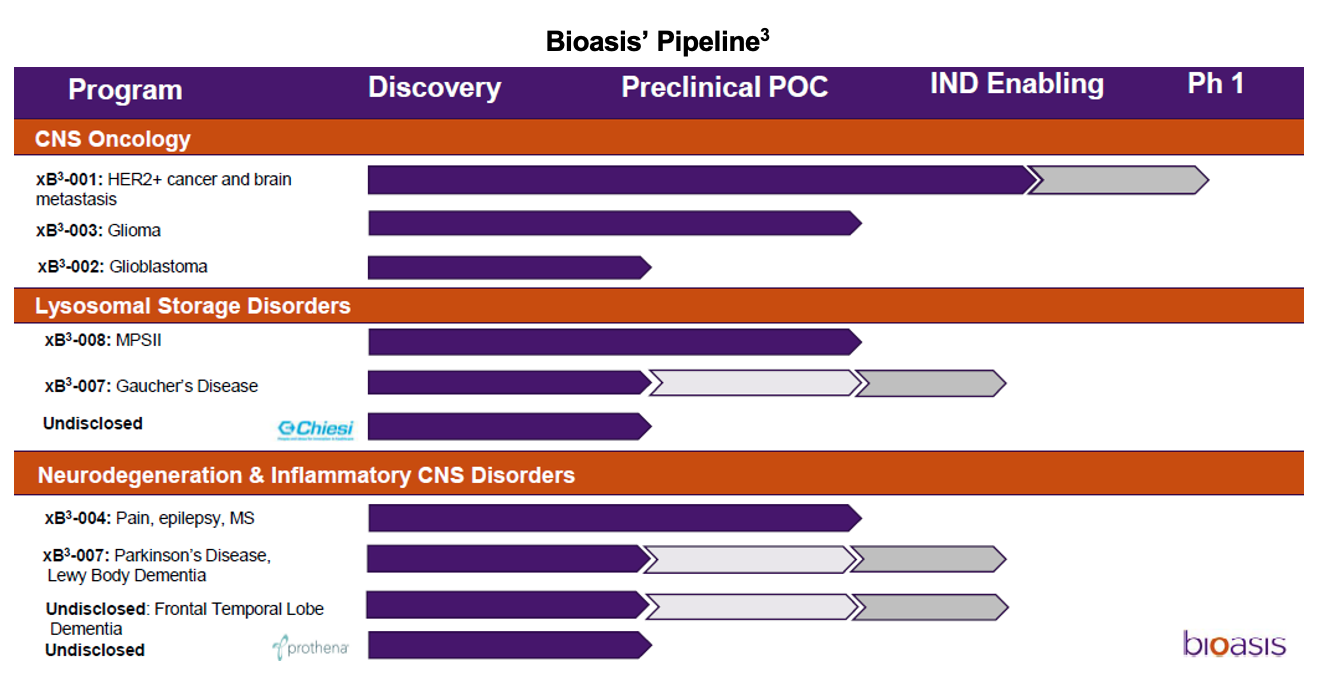
In addition to its internal programs, Bioasis also has cultivated partnerships with other drug development companies that wish to leverage xB3’s ability to cross the BBB. Bioasis is working with three collaborators including Chiesi Group, Prothena Corp. and an unidentified pharmaceutical partner pursuing several lysosomal storage disease and neurodegenerative targets.
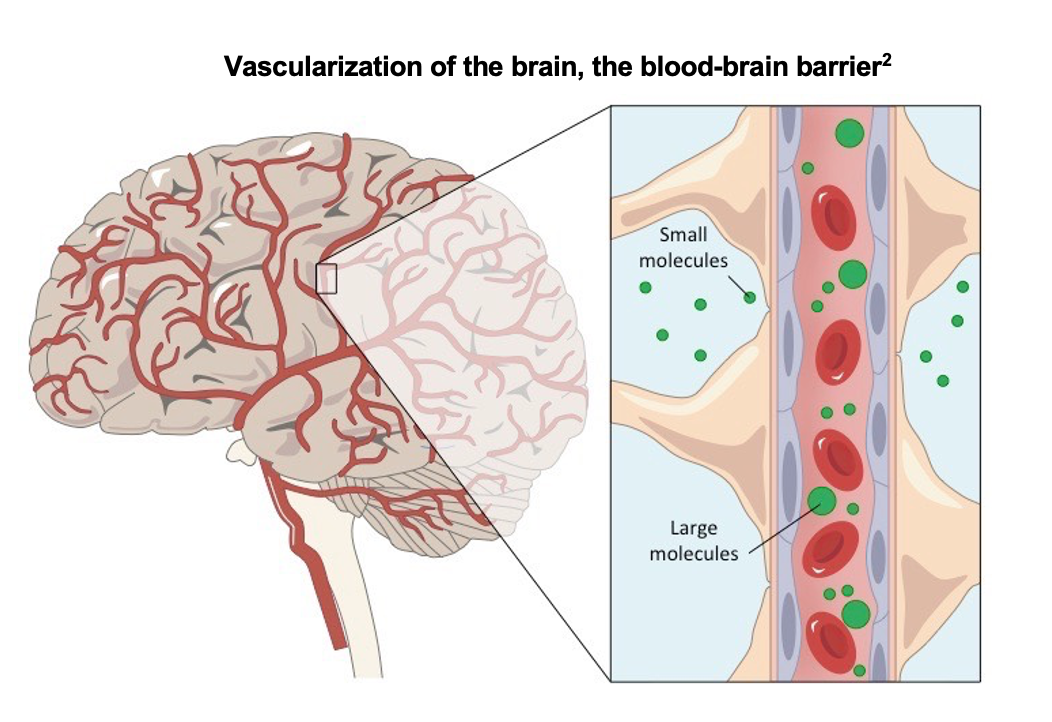
The blood brain barrier prevents free access to the brain from the bloodstream. Only select solutes such as water, gasses, lipid soluble substances and other substances transported via endocytosis or transcytosis are allowed through. While the BBB prevents many harmful substances such as toxins and pathogens from crossing into the brain parenchyma it also blocks helpful therapies that may address a multitude of brain diseases including cancer, neurodegenerative and lysosomal storage diseases among numerous others.
The xB3 platform is based on a twelve amino acid peptide that activates receptor mediated transcytosis to carry molecules across the BBB. Based on the naturally occurring, iron-binding protein melanotransferrin, the sequence can be linked to a variety of payloads including antibodies, enzymes, siRNA and small molecules. While still in the preclinical stage, Bioasis’ platform has demonstrated the ability to efficiently and effectively transport a variety of therapeutics across the BBB with a more diversified payload capability as compared to competing technologies.
Highlights of the platform include:
‣ Small peptide convenient to manufacture, easy to manipulate and lower in cost vs. predecessor technology
‣ Allows for rapid and reproducible synthesis of xB3 based therapeutics
‣ Therapeutic agents transported more readily across the blood-brain barrier vs. previous technology
‣ Preclinical collaborations demonstrated xB3 does not affect the pharmacokinetics of its therapeutic payload
‣ xB3 linked to payloads has demonstrated therapeutic efficacy in rodent disease models
‣ xB3 has been demonstrated to be safe in a wide range of doses in rodent models
Lead Candidates
The two leading internally-developed candidates are designated xB3-001 and xB3-007 which are intended to treat Human Epidermal growth factor Receptor 2-positive (HER2+) breast cancer that has metastasized to the brain and Gaucher Disease (GD). xB3-001 conjugates Bioasis’ amino acid sequence to Herceptin, which is approved for use in HER2+ breast cancer, while xB3-007 links the sequence to Cerezyme, which is approved to treat Type 1 GD.
Bioasis has received non-dilutive funding from interested partners that wish to evaluate the abilities of the xB3 platform with their own products. Further, the company has an agreement in place with XOMA (US) LLC (XOMA), a royalty aggregator that provides funding in return for future interests in developed products.
Bioasis is a preclinical company with several candidates in early stages of development. We anticipate that xB3-001 and xB3-007 will enter the clinic in 2022 followed by the other candidates in the pipeline in subsequent years, some of which will be advanced by partners.
At the end of its first fiscal quarter on May 31, 2020, Bioasis held approximately CAD $190,000 in cash that was augmented by another USD $3 million (~CAD $4 million) following the end of the first quarter representing the upfront payment from the Chiesi agreement. Other non-dilutive funds may be obtained from XOMA in return for royalty and milestone interests in the Chiesi agreement and future commercialization efforts. Additional capital will be required prior to entry in the clinic. The company currently holds no substantive debt following the June 2020 repayment of its 7.5% debentures. We expect Bioasis to consume CAD ~$750,000 in cash per quarter until clinical trials start next year. Note that financial statement items are denominated in Canadian dollars and our target price is denominated in US dollars.
Summary
Bioasis’ innovative approach to crossing the BBB addresses many of the shortcomings faced by therapies developed to treat diseases of the brain. The xB3 technology has attracted attention from a variety of stakeholders, including development partners and investors providing capital, supporting internal development of the product. While the company is in an early stage of development, data generated to date is highly supportive of the amino acid chain’s ability to transport molecules across the BBB. With many approved and in development products that may address brain diseases but cannot cross the BBB on their own, we see many opportunities for future development and ultimate approval of medicines attached to the xB3 platform.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks provides and Zacks receives quarterly payments totaling a maximum fee of $40,000 annually for these services. Full Disclaimer HERE.
________________________
1. Share prices are in US Dollars and financial statement items are in Canadian Dollars.
2. Source: Bioasis July 2020 Corporate Presentation
3. Source: July 2020 Corporate Slide Deck