By John Vandermosten, CFA
NASDAQ:HTBX
READ THE FULL HTBX RESEARCH REPORT
Fourth Quarter and Fiscal Year 2020 Results
Heat Biologics, Inc. (NASDAQ:HTBX) reported full year 2020 results on March 25, 2021 concurrent with the submission of Form 10-K to the SEC. 2020 was a productive year for Heat, advancing its COVID vaccine to scale-up manufacturing stage, generating favorable results from multiple cohorts of the DURGA trial, launching a Phase I for PTX-35 and raising capital sufficient to accumulate a war chest of $111 million. After year end, Heat updated investors on interim survival data from its ongoing HS-110 Phase II NSCLC trial, published additional preclinical data for its COVID vaccine, commenced manufacturing process for ZVX-60, and promoted William Ostrander to Chief Financial Officer.
In the financial sphere, aside from raising almost $120 million, Heat generated $2.9 million in grant revenue and posted a loss of ($26.0) million or ($1.63) per share. $29.1 million was expended to advance clinical programs in PTX-35, HS-110 and ZVX-60.
For the fiscal year ending December 31, 2020 and versus the fiscal year ending December 31, 2019:
➢ Research & development expenses totaled $12.9 million, essentially flat compared to $13.0 million as increases in manufacturing and testing costs for HS-110 were offset by a decrease in expenses related to PTX-35 in the transition from pre-clinical development to in-human studies;
➢ General & administrative expenses rose 59% to $14.9 million from $9.4 million, driven primarily by an increase in personnel and CEO stock compensation expense;
➢ Net loss was $26.0 million, or ($1.63) per basic and diluted share, compared to $20.0 million, or ($4.21) per basic and diluted share;
As of December 31, 2020, cash, equivalents and short-term investments on the balance sheet was $111.8 million, compared to $14.7 twelve months earlier. The increase was driven by a $119.3 million contribution from financing during 2020. Cash burn for the year totaled $22.4 million compared to $13.1 million in 2019, suggesting a runway of approximately five years. In December, Heat announced a 1-for-7 reverse stock split and the disbursement of $2 million to Pelican stakeholders relating to first patient dosing of PTX-35 in a Phase I trial, a milestone stipulated in the acquisition agreement.
Corporate Milestones
➢ HS-110 interim data readout – November 2019
➢ HS-110 Phase II interim readout – 4Q:19
➢ First patient dosed in HS-130 Phase I – December 2019
➢ ASCO poster presentation – May 29, 2020
➢ COVID-19 program launched - March 2020
➢ PTX-35 IND clearance, Phase I initiation and first patient dosing – June 2020
➢ William Ostrander appointed CFO - January 2021
➢ Various coronavirus vaccine milestones – 2021
◦ Commenced manufacturing process for ZVX-60 - January 2021
◦ Investigational New Drug (IND) Application
◦ Phase I launch
➢ Positive interim survival data from HS-110 Ph2 NSCLC - February 2021
➢ Discussion with potential partners – ongoing
➢ Complete HS-130 Phase I trial – 1Q:21
➢ Complete PTX-35 Phase I trial – 1Q:21
➢ End of Phase II meeting for HS-110 – 1H:21
PTX-35
In early June 2020, Heat announced that the FDA had cleared its investigational new drug application (IND) for PTX-35 followed shortly after by the initiation of the first clinical site for the trial and the appointment of Anthony Tolcher, MD as lead investigator. By the end of June, the first patient had been treated in the Phase I trial to evaluate PTX-35. Up to 30 patients are expected to be enrolled with advanced solid tumors refractory to standard of care.
As a reminder, PTX-35 is being developed by 85% owned subsidiary, Pelican Therapeutics, and we allocate royalties accordingly. There is also a relatively complex royalty arrangement with Cancer Prevention and Research Institute of Texas (CPRIT), in return for the $15.2 million total in support that includes a repayment of 4x the original grant and a 0.5% royalty in perpetuity. There are a number of milestones owed to Pelican by Heat that we also reflect in our valuation.
COVID-19 Program
The global threat of coronavirus has changed the landscape for many biotechnology research and development companies. The spread of the virus may delay trial progression and the availability of drug product but it also has incentivized programs that many companies, including Heat Biologics, have developed in infectious disease. Heat’s wholly-owned subsidiary, Zolovax, has been focused on developing medicines and vaccines for infectious diseases using the glycoprotein platform, gp96. Previous research has been conducted for simian immuno-deficiency virus, malaria and Zika. In March 2020, Heat entered into a research agreement with the University of Miami (UM) to sponsor new research and development of a SARS-CoV-2 (COVID-19) vaccine. The agreement granted Zolovax exclusive worldwide rights to research, develop and make use of UM technology until last-patent expiry, or 15 years from first sale, in exchange for upfront fees, annual payments and royalties tied to percentage of net sales of licensed product.
The vaccine incorporates multiple SARS-CoV-2 antigens using the gp96 platform. The approach is expected to in¬duce long-term immunity and provide protection against future infections. As no viral vector is used, Heat’s corona¬virus vaccine avoids anti-vector immunity and viral activation whilst activating T and B cells with high immunogenici¬ty. The activation of T and B cells drives induction of mucosal immunity and long-term memory response. In March 2020, the company filed multiple provisional patent applications for its technology that treat and prevent infection from the SARS-CoV-2 virus. Heat’s approach may also be appropriate in combination with other vaccines that acti¬vate the humoral immune system on account of its complementary stimulation of the adaptive immune system.
In late July 2020, Heat confirmed successful pre-clinical testing of the vaccine, demonstrating immunogenicity in an animal models. The vaccine was shown to expand human-HLA (human leukocyte antigen)-restricted T cells against immunodominant epitopes of SARS-CoV-2 Spike protein. In mid-August, Heat provided an update on the coronavirus program providing additional preclinical data for Heat’s gp96-based vaccine. In an animal model, the COVID-19 vaccine stimulated expansion of both killer CD8+ T cells and helper CD4+ T cells. These immune cells destroy virally infected cells and assist in producing antibodies specific to the virus. Both the CD8+ and CD4+ cells release cytokines that augment the immune response and migrate to the lungs and airways where the SARS-CoV-2 infection resides. Heat was involved in two COVID-19 publications last summer including a white paper on “The Importance of T-Cell Immunity in SARS-CoV-2 Infection” and the preclinical COVID-19 vaccine results in a paper entitled “Induction of SARS-CoV-2 protein S-specific CD8+ T cells in the lungs of gp96-Ig-S vaccinated mice.”
On December 16, 2020, Heat announced the completion of the ZVX-60 vaccine cell line for COVID-19. Data from University of Miami, confirmed expression of gp96, OX40L and spike protein, a milestone in preparation for manufacturing and clinical vetting of the vaccine. Shortly thereafter, Heat commenced the manufacturing process for the vaccine, announcing Waisman Biomanufacturing as its manufacturing partner on January 19, 2021. The ZVX-60 cell line, with its recently confirmed expression, was transferred to Waisman to initiate the manufacturing process.
Additional preclinical COVID-19 vaccine results were published in the journal Frontiers in Immunology. The publication highlighted additional data sets around memory T cells and built on previously reported data. The new data demonstrated polyfunctional, antiviral cytokine releasing CD8+ and CD4+ T cell memory responses specific to the SARS-CoV-2 spike protein, specifically in the lungs and spleen of immunized animals at 30 days post vaccination. In the lungs of mice, memory CD8+ T cell response was observed 60 days post vaccination, indicating a durable immune response attributed to lung tissue-resident memory T cells that are required for clearing viral infections of the lungs.
Heat has made the case for using its approach, if ultimately approved, along with other vaccines to improve re¬sistance to COVID-19. Many other vaccines that are being developed for the virus stimulate an antibody response and if used in conjunction with Heat’s gp96 approach, it would add T cell immunity to improve protection.
Positive Interim Data for Phase II HS-110 in NSCLC
Heat announced positive interim survival data for its Phase II trial of HS-110 (viagenpumatucel-L) in combination with leading checkpoint blockade inhibitors (CBIs). The trial was not a comparator trial; however, similarities in design with past trials enable comparison of competitors’ performance with HS-110.
Heat is investigating HS-110 in non-small cell lung cancer (NSCLC) and is currently conducting a Phase II trial. The Phase II DURGA trial (NCT02439450) features two treatment settings, as first line maintenance and second line or greater therapy.

HS-110 Phase II Structure (1)
The treatment setting in first line maintenance is divided into two arms, HS-110 and pembrolizumab (KEYTRUDA) with and without pemetrexed (ALIMTA), a chemotherapy agent. The second arm features HS-110 and nivolumab (Opdivo). Pembrolizumab and nivolumab are monoclonal antibodies targeting programmed death receptor-1 (PD-1) and are known as checkpoint inhibitors. Checkpoint inhibitors interfere with the ability of tumor cells to defy immune response, allowing the body to attack the tumors. Finally, the cohorts within the nivolumab arm, cohorts A and B, evaluate the combination of HS-110 and nivolumab in both checkpoint inhibitor naïve and checkpoint inhibitor progressor subjects. The study is intended as proof of concept in a defined patient population. Enrollment has completed with 122 patients.
Interim data was positive and compared favorably to historical data. The CheckMate 057 study was conducted in 2015 by Bristol Myers Squibb to evaluate Opdivo in non-squamous NSCLC. CheckMate 057 was a Phase III trial evaluating nivolumab versus docetaxel in patients with previously-treated non-squamous NSCLC. Patients were randomized 1:1 to nivolumab and docetaxel, with primary endpoint of overall survival (OS). Secondary endpoints included progression-free survival (PFS) and safety. Results showed that median OS was 12.2 months in the nivolumab only group versus docetaxel at 9.4 months (2). Median PFS was 2.3 months and 4.2 months for nivo-lumab and docetaxel, respectively. This compares to cohort A of Heat’s HS-110 trial in an almost completely non-squamous population (94%) that had median OS of 24.6 months and median PFS of 1.84 months. The interim data supports the trial’s objective of clinical proof-of-concept and interim data compared favorably to historic data.
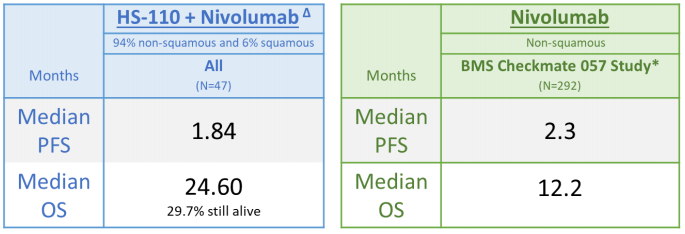
Cohort A Versus Historical Data (3)

Overall Survival Curve, Cohort A (4)
Results from Cohort B, checkpoint inhibitor progressors, were also compared to historical data. Constantini et al. 2018 (5) and Schvartsman et al. 2017 investigated third-line or greater chemotherapy in CBI-failed subjects. Median OS ranged between 6.8 months and 9 months and median PFS ranged from 2.7 to 4.7 months in the studies. This compared to cohort B data with median OS of 11.9 and median PFS of 2.76, exceeding the upper end of median OS in the chemotherapies studied by almost three months. Heat’s Phase II DURGA trial lacks a control arm and there are limitations to comparing it with other trials; however, Heat’s interim results cast a favorable light on its candidate.
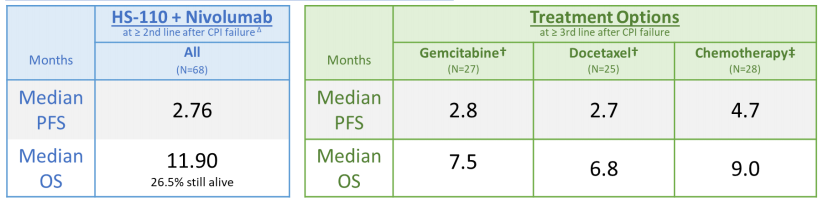
Cohort B Versus Historical Data (6)

Overall Survival Curve, Cohort B (7)
HS-110 has a favorable safety profile with approximately 200 patients receiving the drug and no treatment-related serious adverse reactions. For additional detail on results for HS-110, please refer to our previous article.
Appointment of William L. Ostrander to Chief Financial Officer
Heat promoted William Ostrander to the position of Chief Financial Officer, which went into effect January 4, 2021, and was announced a week later on the 11th. Mr. Ostrander joined Heat in September 2019 as Vice President of Finance bringing two decades of experience to the role. Ostrander held leadership positions at Liquidia Technologies, serving as Director of Finance & Controller then Executive Director of Finance between 2014 and 2019, at KBI Biopharma for seven years as Senior Director, Finance & Accounting, and at Lexis Nexis, Seisint, and Boca Research in Finance and Accounting roles.
Pipeline
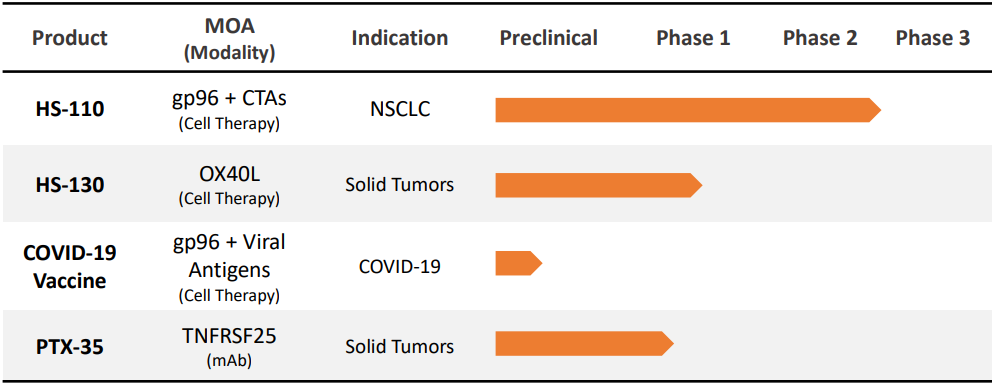
Heat Biologics Product Pipeline (8)
Summary
Heat Biologics has made substantial progress this year with the entry of HS-130 and PTX-35 into the clinic as well as continued advancement of the Phase II HS-110 program. Substantial preclinical work has also taken place for a coronavirus vaccine which may enter the clinic this year. Manufacturing milestones have been achieved, and an IND and Phase I study are in the works. Year to date, Heat has been opportunistic in raising capital and has accumulated over $117 million on the balance sheet that should sustain operations for the next several years.
Heat Biologics’ interim data from the DURGA trial reflect results from a combination approach of HS-110 with CBIs. The results compare favorably to historical data with CBIs alone. This is an early confirmation of the opportunity for Heat’s gp96/CTA technology. The interim data show HS-110’s potential as a second line or later therapy, in conjunction with an anti-PD-1, in either checkpoint inhibitor naïve or progressor patients. Based on this interim data, it appears that HS-110 and nivolumab could be superior to CBI or chemotherapy alone. As Heat completes the Phase II trial, we look forward to topline and final analysis from the study, which is expected to be presented at a major oncology conference yet to be disclosed. Heat management is in the process of arranging an End-of-Phase II meeting with the FDA where trial design for the anticipated Phase III will be a primary point of discussion. Management is also in talks with key opinion leaders (KOLs) regarding the optimal regulatory path to pursue for the candidate. HS-110 may be pursued as a frontline treatment for NSCLC or as second line in checkpoint inhibitor-progressed patients.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks provides and Zacks receives quarterly payments totaling a maximum fee of $40,000 annually for these services. Full Disclaimer HERE.
________________________
1. HS-110: Heat Biologics, Inc. (HTBX)
2. Borghaei, H., Paz-Ares, L., Horn, L., Spigel, D. R., Steins, M., Ready, N. E. & Brahmer, J. R. (2015). Nivolumab versus docetaxel in advanced non-squamous non–small-cell lung cancer. New England Journal of Medicine, 373(17), 1627-1639.
3. Heat Biologics Corporate Presentation February 2021
4. Source: Heat Biologics March 2021 Corporate Presentation
5. Costantini, A., Corny, J., Fallet, V., Renet, S., Friard, S., Chouaid, C. & Cadranel, J. (2018). Hyper-progressive disease in patients with advanced non-small cell lung cancer (NSCLC) treated with nivolumab (nivo).
6. Heat Biologics Corporate Presentation February 2021
7. Source: Heat Biologics March 2021 Corporate Presentation.
8. Source: Heat Biologics March 2021 Corporate Presentation.