By John Vandermosten, CFA
NASDAQ:LPCN
READ THE FULL LPCN RESEARCH REPORT
TLANDO Licensing Agreement with Antares Pharma
On October 18th, Lipocine (NASDAQ:LPCN) announced that it had entered into a licensing agreement with Antares Pharma. The agreement is for commercialization of TLANDO in the US and includes the following terms:
➢ Up to $21 million in licensing fees;
◦ $11 million payable immediately;
◦ $10 million in future payments subject to certain conditions;
➢ Commercial sales payments based on milestones up to $160 million;
➢ Tiered royalties on net sales from mid-teens up to 20%;
➢ Antares responsible for all commercialization, post-marketing studies, and sourcing of TLANDO in US;
➢ Also included is Antares option to license TLANDO XR; if exercised
◦ Antares gains license to develop and commercialize TLANDO XR in the US;
◦ Lipocine would receive $4 million in license fees;
◦ Up to $35 million in clinical and regulatory milestone payments;
◦ Tiered royalties on net sales from mid-teens to 20%
◦ Antares responsible for all clinical development costs, regulatory filings, commercialization and post-marketing activities.
Lipocine retains all rights to rest of world and non-testosterone replacement therapy indications for both TLANDO and TLANDO XR. We update our model to reflect the terms of this arrangement and assume that Antares will exercise its option to advance TLANDO XR.
LPCN 1144 for Treatment of Non-Cirrhotic NASH
With the recent grant of Fast Track from the FDA and terms for the commercialization of TLANDO settled, LPCN 1144 moves into pole position. In support of our positive assessment of LPCN 1144, we summarize the clinical data available to date for the LiFT study in this report highlighting the safety and efficacy of the drug and providing a comparison with other NASH candidates. Now that responsibilities for commercializing TLANDO have been assigned, we expect Lipocine to shift its primary attention towards LPCN 1144 and an anticipated upcoming meeting with the FDA to determine trial design. We anticipate that a clear path forward for LPCN 1144 will be provided to stakeholders in early 2022.
Fast Track Designation
In a November 4th press release, Lipocine shared its success in obtaining the FDA’s Fast Track Designation for LPCN 1144 for treatment of non-cirrhotic NASH. The designation is requested by a sponsor company for drug candidates that treat serious conditions and fill an unmet medical need. As no other NASH treatments have been approved and results from the LiFT trial were positive, the designation is welcome and not a surprise. The status should help Lipocine more efficiently design the Phase III program with closer guidance by the regulatory agency. It will also confer several benefits including more frequent interaction with the FDA, eligibility for accelerated approval, priority review and rolling review. Rolling review will allow Lipocine to submit portions of its new drug application (NDA) as they become ready rather than waiting until the entire package is ready thereby speeding the review process.
January 2021 Topline Results from LiFT Study
Lipocine announced positive 12-week topline data from its LiFT study in January 2021. LiFT is a Phase II evaluation of its candidate LPCN 1144, an oral pro-drug of endogenous testosterone, in biopsy-confirmed, non-cirrhotic male NASH patients with F1-F3 fibrosis. Results were positive and show statistical significance in the primary endpoint of change in hepatic fat fraction, quantified via MRI-PDFF1 at week 12. Lipocine’s LPCN 1144 demonstrated robust, statistically significant efficacy in not only its primary endpoint of reduction of hepatic fat fraction, but also secondary endpoints that were published in August.
LiFT Study Design
LiFT (Liver Fat intervention with oral Testosterone) is a Phase II trial designed to evaluate LPCN 1144 oral testosterone in men with biopsy-confirmed NASH. It enrolled 56 men with confirmed NASH, randomized 1:1:1 into three arms. The arms included Treatment A, 142 mg testosterone equivalent twice daily, Treatment B, which was the same as Treatment A but with the addition of 217 mg of d-alpha tocopherol equivalent twice daily and a placebo arm with twice-daily administration. Excluding the open label extension, LiFT had a duration of 36 weeks.
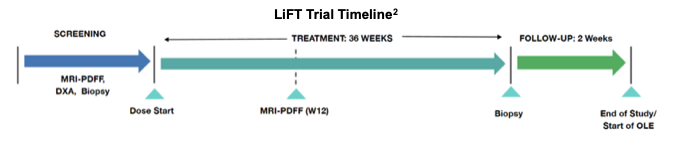
The primary endpoint for LiFT was change in hepatic fat fraction, evaluated using MRI-PDFF at week 12, topline results for which were released in January 2021. Secondary endpoints included change in NASH activity and fibrosis via liver biopsy scoring at week 36, change in hepatic fat fraction via MRI-PDFF at week 36, change in liver injury markers, anthropomorphic measurements, lipids, insulin resistance and inflammatory/fibrosis markers, and Patient Reported Outcomes (PROs) including quality of life and global impression scores (PGI). In the latest update, Lipocine obtained biopsy samples to evaluate the four markers used in NASH CRN scoring.
Patients were offered access to LPCN 1144 through an open label extension study, announced December 30, 2020. This option for patients enabled the collection of additional data for up to 72 weeks total. The open label enrollment is ongoing with data reporting expected in mid-2022.
Topline Results for Primary Endpoint
Baseline characteristics were presented that recounted the participants, completion, age, BMI, diabetes and hypertension, as well as baseline measurements of endpoint factors.
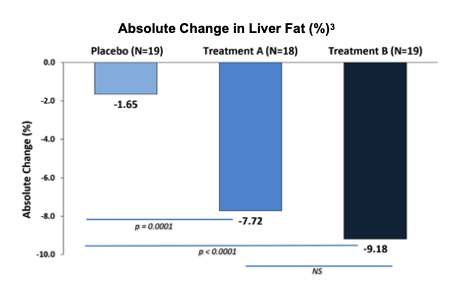
The primary endpoint for LiFT was change in hepatic fat fraction. As reported, there was a statistically significant decrease in hepatic fat, significant at the 0.01% level (p < 1%) for both Treatment A and Treatment B arms. The percentage of subjects with greater than 30% reduction in liver fat was statistically significant in excess of 1% in both arms vs placebo. Absolute and relative change in liver fat percentage in subjects with baseline liver fat in excess of 5% was statistically significant at 0.01% for both arms vs placebo.
In evaluation of liver injury markers, d-alpha tocopherol’s potentiation of LPCN 1144 was quantifiably apparent. With respect to alanine aminotransferase (ALT), both treatment arms were statistically differentiable from placebo at 1.6% and 0.01% for A and B arms, respectively. Here, Treatment B was statistically distinguishable from Treatment A arm at the 0.5% level. Evaluation of change in aspartate aminotransferase (AST) was again statistically significant for both arms at 2% and 0.01% levels for A and B arms, respectively, but here A and B arms were not statistically different, although directionally consistent. ALT and AST reduction in absolute terms were up to 22.4 U/L and 10.4 U/L, respectively. Results including d-alpha tocopherol generated substantially reduced liver injury markers ALT and AST compared with LPCN 1144 alone. The difference between the two arms was less dramatic in other measures such as hepatic fat fraction reduction. Lipocine proposed antioxidant activity of d-alpha tocopherol as a possible mechanism that impacted ALT and AST, although the exact mechanism was not understood.
Finally, longitudinal analysis of changes in the two liver injury markers showed that as early as week four, for AST, and week eight for both ALT and AST, combination LPCN 1144 and d-alpha tocopherol showed statistically significant changes. The treatment was not only successful where other NASH therapies have struggled, but also conveniently orally administered and timely in its efficacy.
Adverse events were comparable to the placebo arm with no observed tolerability issues. Three subjects in the placebo group and one in a treatment arm discontinued study due to treatment emergent adverse events (TEAE).
LiFT Liver Biopsy Results
On August 25, 2021, Lipocine released liver biopsy results for its Phase II LiFT study in NASH. As a long-term follow up to the results in January, August’s biopsy results focused on the 36-week assessment of histological change for NASH resolution and no worsening of fibrosis and body composition and liver injury markers. These are the secondary endpoints. After the post-biopsy two week follow up, subjects may join the open label extension study which will further examine safety. In total, 56 patients enrolled in the safety set, 44 provided both baseline and 36-week biopsy, and 37 produced a baseline NAS of greater than or equal to 4 with improvement in at least 1 point for both inflammation and ballooning, measures of NASH resolution.
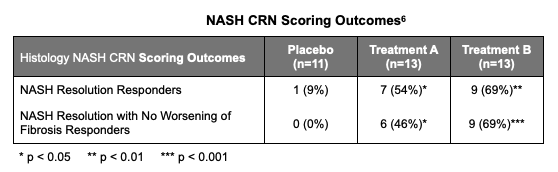
Liver biopsies were taken at baseline and at 36 weeks to measure NASH resolution and fibrosis. NASH resolution results were favorable with very good statistical significance. The FDA requires resolution of steatohepatitis on overall histopathological reading and no worsening of liver fibrosis on NASH CRN fibrosis score.4 We see that with this small sample of data, both treatment arms were able to produce substantially more responders than the placebo group, even with the added constraint of no worsening of fibrosis.
Scoring
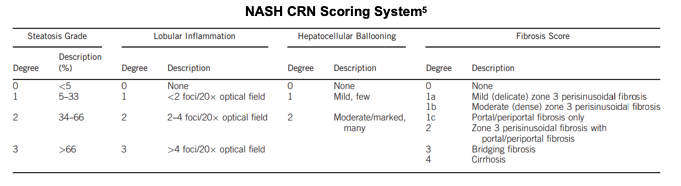
A standard scoring system was used for the LiFT study evaluation. The same system has been used in other trials and is the standard accepted by the FDA to evaluate results. Below, we summarize the components of the scoring system which includes measurements of steatosis grade, inflammation, ballooning and fibrosis.
When evaluated using CRN, LPCN 1144 was superior to placebo at p-values of 5% for Treatment arm A, and 1% and 0.1% level in Treatment B, respectively.
The FDA requires histologic support of resolution of NASH with no worsening of fibrosis and/or resolution of fibrosis with no worsening in NASH and surrogate markers of efficacy for approval. To score fibrotic development, NASH CRN is used as a surrogate, compared to baseline. Each of the readings were performed on the same slides and pathologists were blinded as to the timing of the reads (baseline and end of study) and treatment group to reduce bias.
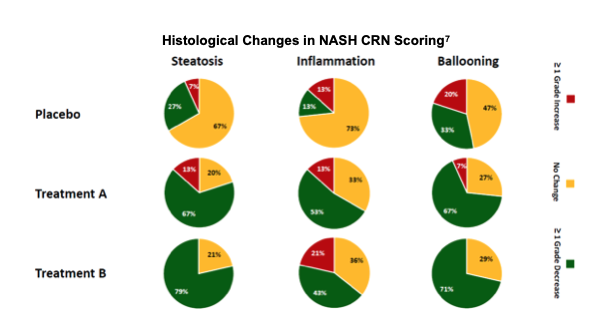
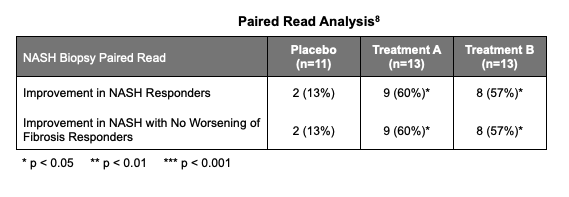
Other approaches to assess resolution of NASH include paired read analysis, where the baseline biopsy vs. the 36 week biopsy slides are evaluated side by side. The paired read results concurred with the NASH CRN findings.
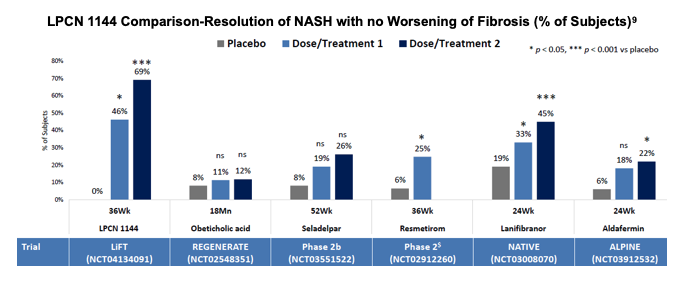
Lipocine compiled a comparison of results from other clinical trials and the proportion of the population in the treatment arm that was able to resolve NASH. The comparator trials are different in terms of trial design, size and patient populations and that there have been no head to head trials among the NASH candidates. Results from the comparison are favorable for LPCN 1144 and demonstrate a substantial improvement over placebo.
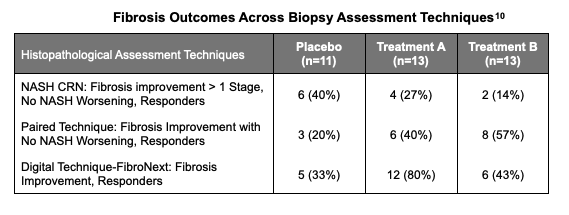
In summary, the combination of NASH measures in the restricted patient population warrant confirmation in a larger study. Of note, the placebo group appeared to outperform the treated arms on a measure of fibrosis improvement of > 1 Stage with no NASH worsening on number of responders.
The follow up also assessed changes in liver injury markers, ALT and AST, and body composition, that were directionally favorable, but with varying degrees of statistical significance.
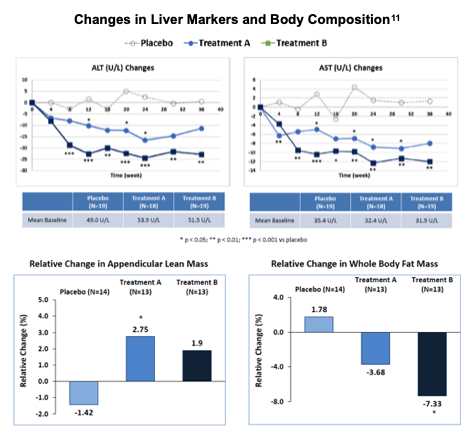
Safety results from the trial through week 36 were favorable with frequency and severity of treatment emergent adverse effects (TEAEs) for both treatment arms comparable with placebo. Drug related TEAEs were mild to moderate. Four subjects discontinued participation from the trial in the placebo arm and one discontinued in the treatment arms. Cardiovascular events were balanced among the groups and there were no reported cases of hepatocellular carcinoma or drug induced liver injury. Other measures were also mild to moderate with little difference between the control and active groups.
Next Steps
Now that selected 36-week biopsy data has been presented to stakeholders and with the Fast Track Designation in its back pocket, Lipocine’s next steps are to schedule an End-of-Phase II meeting with the FDA, prepare a presentation for a scientific and medical conference and complete the extension study. Results from the Phase II are positive and can lead down several pathways depending upon the FDA’s guidance following the anticipated meeting. A Phase IIb study may be launched, or if data appears to provide sufficient proof of concept, a Phase III may be more appropriate. We think it is likely that Lipocine will seek a partner to advance LPCN 1144 into a registrational study.
Milestones
➢ NDA filed for TLANDO – February 2020
➢ IND clearance for Phase II study of LPCN 1148 – May 2020
➢ Tentative approval of TLANDO – December 2020
➢ LiFT Study
◦ Primary endpoint – January 2021
◦ Last patient, last visit for biopsy data – June 2021
◦ Presentation of 36-week, biopsy data to investors – August 2021
◦ End of Phase II meeting with FDA – 2H:21
◦ Presentation of study data at conference – 4Q:21
➢ Abstract presentation at EASL – June 2021
➢ TLANDO licensing agreement – October 2021
➢ Grant of Fast Track Designation for LPCN 1144 – November 2021
➢ Topline announcement for PK study for LPCN 1154 – 4Q:21/1Q:22
➢ Launch of Phase IIa proof of concept PPD trial – 4Q:21
➢ Male cirrhosis trial first subject dosed for LPCN 1148 – 4Q:21
➢ Results from LiFT Extension Study – Mid-2022
➢ TLANDO eligible for final approval and marketing – 2Q:22
Valuation
We adjust our valuation to consider the terms of the licensing for TLANDO with Antares Pharma. We also adjust our expectations for sales reflecting the competitive environment and recent sales trends from Jatenzo and previously approved products in the TRT space. While TLANDO has a better side effect profile compared with Jatenzo and does not require titration, the slow start to Jatenzo’s sales12 temper our estimates for TLANDO’s future potential. We also reviewed Antares’ Xyosted which was initially launched in early 2019 but is unlikely to generate revenues over $100 million in 2021. We forecast TLANDO revenues similar to the average profile of earlier testosterone products such as Androgel or Testim and a peak of about $600 million in product sales several years down the road.13 We give credit to TLANDO XR and anticipate that it will be successfully developed and take over from TLANDO several years down the road assuming it is approved. We also reduce our previous royalty estimate from 35% to 25%, which includes the base royalty of up to 20% and an additional amount to reflect future milestones.
The brightest spot in Lipocine’s portfolio is LPCN 1144, which has been generating Phase II data all year and was recently granted Fast Track authorization. The positive results are sufficient for Lipocine to obtain an attractive partner, or, using the proceeds from the Antares deal, possibly fund a pivotal study themselves. We apply a 40% probability of approval to the candidate and anticipate product sales in excess of $1 billion if it is successfully commercialized as there are no alternatives available and there have been several failures by competitors.
The remaining candidates have shown early promise but will likely need partners to take them across the finish line. We apply a 10% likelihood of commercial success to these programs. LPCN 1148 in cirrhosis management is a relatively small market with a prevalence of about 650,000. As the population is not explicitly defined and data is scarce, we estimate a low single digit penetration into the market. Despite the low penetration, pricing is expected to be high given the cost savings a successful drug could generate in this category. We apply a probability of success of 10% given the early stage and uncertainty of ultimate commercialization.
LPCN 1107 has completed Phase II studies but has been on hold pending funding to advance the program and clarification on the FDA’s stance on the efficacy of Makena (hydroxyprogesterone caproate). The injectable drug was approved for pre term birth but as a condition of approval, the sponsor was required to conduct a post approval confirmatory trial. The trial ultimately did not demonstrate an effect of Makena on the surrogate end point of preterm birth and the FDA is determining whether or not the drug should remain available.14 Due to uncertainty regarding the ultimate disposition of the product, we apply a low probability of 15% for this Phase III ready asset. Other assumptions for LPCN 1107 include completed clinical trials and FDA approval by 2026 leading to first sales in 2027. We anticipate pricing of $6,000 per course of treatment and penetration peaking in the mid-teens in the fifth year after launch.
Post-partum depression is another early-stage endeavor with an addressable market of about 600,000. Treatment in this category is not chronic and we assume from 3-4 months of therapy in this population over the post-partum period. Pricing for branded product in this space averages approximately $800 per month. Our estimates for penetration for LPCN 1154 are from 1-2% given the number of anti-depressants available and the uncertainty of efficacy at this stage of development. Probability of success is estimated at 10% as the candidate is at an early stage and management has not clarified its pathway forward.
For all of Lipocine’s programs, except for TLANDO, we assume a 35% royalty, which represents all of the economic value the company receives from upfronts, milestones and royalties. We believe that the highly efficacious data and lack of other treatments for NASH along with a stronger financial position from TLANDO contributions will give Lipocine a firm position to negotiate a deal or develop subsequent products on its own.
Summary
Lipocine’s licensing agreement with Antares Pharma for the US market marks the end of the research and development stage for TLANDO and passes on the mantle of commercialization to Antares. The deal represents $11 million in immediate upfronts and up to an additional $10 million in licensing fees, as well as commercial sales milestone payments up to $160 million, with tiered royalties ranging from mid-teens to 20%. The deal also includes the option for Antares to secure rights to TLANDO XR for up to $4 million as well as future milestones and royalties.
As TLANDO’s future becomes more evident, attention turns to the other assets in the company’s portfolio, particularly LPCN 1144. Lipocine’s Phase II biopsy data for LPCN 1144 yielded positive results, with agreement across multiple measures of NASH and statistical significance in a small patient group, as well as positive liver marker, body composition and safety results. While fibrosis improved more in the placebo arm vs. the treatment group under the NASH CRN guidelines, other techniques for measuring fibrosis showed favorable conclusions, indicating that a larger study is needed to validate the results. A trial in a larger patient population is expected, which will be under consideration as Lipocine prepares to meet with the FDA. Interactions with the agency will be enhanced with the recent grant of Fast Track and we expect to hear additional details regarding the design and scope of the anticipated Phase III trial after the meetings. We update our valuation to reflect the current portfolio of programs, size of likely end markets and the reasonable expectations for their success.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
1. Magnetic Resonance imaging Proton Density Fat Fraction
2. Source: Lipocine August 25 LiFT Key Topline Results Corporate Presentation
3. Lipocine March 2021 corporate presentation
4. FDA Draft Guidance. Noncirrhotic Nonalcoholic Steatohepatitis With Liver Fibrosis: Developing Drugs for Treatment. December 2018.
5. Puri, P, Sanyal, A, Nonalcoholic Fatty Liver Disease: Definitions, Risk Factors, and Workup. Clinical Liver Disease, Vol. 1, No. 4, August 2012
6. Source: Company press releases and Zacks analyst work.
7. Source: Lipocine August 25 LiFT Key Topline Results Corporate Presentation
8. Source: Lipocine August 25 LiFT Key Topline Results Corporate Presentation
9. Source: Lipocine August 25 LiFT Key Topline Results Corporate Presentation
10. Source: Lipocine August 25 LiFT Key Topline Results Corporate Presentation
11. Source: Lipocine August 25 LiFT Key Topline Results Corporate Presentation
12. Sales for Jatenzo were $5.1 million for the first six months of 2021 as reported in the company’s SEC filings.
13. Note that revenue numbers we present in our model represent royalty revenues, not product revenues which will appear on Antares’ income statement.
14. For a discussion of the details regarding Makena’s approval and post-approval study please see: Withdrawing Approval of Makena — A Proposal from the FDA Center for Drug Evaluation and Research.