By David Bautz, PhD
NASDAQ:TNXP
READ THE FULL TNXP RESEARCH REPORT
Business Update
Phase 2 Trial of TNX-102 SL Initiated in Long COVID
On August 22, 2022, Tonix (NASDAQ:TNXP) announced the initiation of the PREVAIL Phase 2 clinical trial of TNX-102 SL in patients with Long COVID (NCT05472090), a heterogeneous condition that involves nociplastic pain following infection with and recovery from SARS-CoV-2, the virus that causes COVID-19 (Bierle et al., 2021). Interestingly, a number of Long COVID symptoms (multi-site pain, fatigue, sleep disorders, and cognitive dysfunction) overlap with key symptoms of fibromyalgia, and it is these patients that the company will be focusing on.
Research continues to advance in the study of musculoskeletal pain, and recently a new descriptor has been proposed to describe pain that is neither nociceptive (due to the activation of nociceptors from actual or threatened damage to non-neural tissue) or neuropathic (caused by a lesion or disease of the somatosensory nervous system) (Trouvin et al., 2019). Nociplastic pain is used to describe pain that arises from altered pain sensation despite no clear evidence of actual or threatened tissue damage, or for disease or lesion of the somatosensory system. Patients suffering from fibromyalgia-like Long COVID fit into the description of those suffering from nociplastic pain.
Central sensitization (amplified neural signaling within the central nervous system that causes hypersensitivity to pain) is a complementary explanation why patients suffer from chronic non-specific pain, particularly in conditions such as fibromyalgia, chronic fatigue syndrome, and irritable bowel syndrome (Woolf et al., 2011). The Central Sensitization Inventory (CSI) is a screening tool to identify patients with central sensitization (Neblett et al., 2013). A survey of 491 Long COVID patients showed that 70% had symptoms of central sensitization (CSI≥40/100) and 65% had severe central sensitization symptoms (Goudman et al., 2021).
Approximately 50% of patients who contract SARS-CoV-2 experience Long COVID symptoms up to a month after infection. Even after six months, 30% of patients continue to experience Long COVID symptoms. Patients who are hospitalized and those with severe symptoms following SARS-CoV-2 infection are most likely to suffer from Long COVID (Hirschtick et al., 2021). A recent report from the Brookings Institute reported that approximately two to four million Americans weren’t working due to the effects of Long COVID, which translates to approximately $170 billion a year in lost wages.
To better understand the potential size of the Long COVID market, a retrospective, observational database study was performed to identify the number of Long COVID patients from approximately 75 million patients from a network of inpatient and outpatient electronic medical records from 48 U.S. healthcare organizations. The results showed that of 1 million patients diagnosed with COVID, approximately 52,000 had Long COVID symptoms that lasted between 3 and 6 months. In addition, of the patients with Long COVID, 41% had multi-site pain. These patients took a variety of medications, include benzodiazepines, opioids, and antidepressants. The rate of opioid use was 50% for patients that had multi-site pain and insomnia, with or without fatigue. This is especially concerning since approximately 25% of patients prescribed opioids long term will struggle with opioid addiction (U.S. Department of Labor). The results of this study clearly show that there is a large patient population suffering from Long COVID that need additional treatment options.
The Phase 2 PREVAIL study is a 14-week, double blind, randomized, multicenter, placebo controlled trial to evaluate the efficacy and safety of TNX-102 SL in patients with multi-site pain associated with post-acute SARS-CoV-2 infection (PASC). We anticipate approximately 470 patients being enrolled into the trial with the primary efficacy endpoint being the change from baseline in the weekly average of daily self-reported worst pain intensity scores at the Week 14 timepoint. Key secondary endpoints include change from baseline in self-reported scores for sleep disturbance, fatigue, and cognitive function. We expect an interim analysis to be performed after the first 50% of enrolled patients have completed the study, with the results of that analysis guiding possible sample size re-estimation or to stop the study early for efficacy. The outcome of the interim analysis is expected in the first half of 2023.
TNX-801 Phase 1 Trial to Initiate 1H23
In July 2022, Tonix Pharmaceuticals Holding Corp. announced a collaboration with the Kenya Medical Research Institute (KEMRI) to plan, seek regulatory approval for, and conduct a Phase 1 clinical trial in Kenya to develop TNX-801, a live form of the horsepox virus, as a vaccine to prevent monkeypox and smallpox. We anticipate the study initiating in the first half of 2023.
The World Health Organization (WHO) has declared monkeypox a global health emergency. More than 40,000 cases of the disease have been reported in 94 countries around the world. While the exact cause of the current outbreak is unknown, it has thrust the potential need for a monkeypox vaccine into the spotlight.
In June 2022, Tonix announced a poster presentation at the 4th Symposium of the Canadian Society for Virology that described results from animals vaccinated with TNX-801 to protect against monkeypox. A copy of the poster presentation can be found here. All animals (n=8) vaccinated with TNX-801 were fully protected with sterilizing immunity (e.g., no lesions developed) from an intra-tracheal challenge with monkeypox. The vaccinations with TNX-801 were well tolerated as exhibited by stable weights and body temperature for all treated animals.
Tonix recently announced that the U.S. Patent and Trademark Office (USPTO) issued U.S. Patent 11,345,896, titled “Synthetic Chimeric Poxviruses”, that includes claims covering synthetic horsepox virus and for the company’s recombinant pox virus platform to protect against other pathogens, including SARS-CoV-2. The patent should provide market exclusivity in the U.S. until 2037, excluding any patent term extensions or patent term adjustments.
Update on Phase 3 RESILIENT Trial for TNX-102 SL in Fibromyalgia
In April 2022, Tonix initiated the RESILIENT study, a randomized, double blind, placebo controlled potentially pivotal Phase 3 trial of TNX-102 SL for the treatment of fibromyalgia. It is expected to enroll approximately 470 participants in the U.S. The results of a planned interim analysis are expected in the second quarter of 2023.
In March 2022, Tonix announced that, as expected based on a previously reported pre-specified interim analysis, TNX-102 SL did not achieve the primary endpoint of reducing fibromyalgia (FM) daily pain at Week 14 in the Phase 3 RALLY trial (P=0.115).
In December 2020, Tonix announced positive topline results for the Phase 3 RELIEF study of TNX-102 SL 5.6 mg (primary endpoint, P=0.010). Thus, since Tonix already has already conducted one positive trial in fibromyalgia, positive results from the RESILIENT study could put the company in position to file a new drug application (NDA) for TNX-102 SL for the treatment of fibromyalgia.
Phase 2 Study of TNX-1900 for Chronic Migraine to Initiate 4Q22
In June 2022, Tonix announced the publication of a paper in Pharmaceutics that describes the enhancing effect of magnesium on the activity of intranasal oxytocin in an animal model of craniofacial pain (Bharadwaj et al., 2022). TNX-1900 is based on the magnesium enhanced formulation of intranasal oxytocin to prevent migraines in chronic migraineurs. The technology is also being utilized in TNX-2900, which is being developed to treat over-eating in adolescent and young adult patients with Prader-Willi syndrome. We anticipate the company initiating a clinical trial of TNX-1900 for the prevention of migraine headache in chronic migraineurs in the fourth quarter of 2022.
Migraine
A migraine is a headache that can cause severe pain, can last up to 72 hours, has a pulsing quality, and may be associated with nausea, vomiting, phonophobia, or photophobia. Approximately one billion individuals in the world suffer from migraine headaches (GBD 2016 Headache Collaborators). In the U.S., migraines affect approximately 72 million individuals (Gooch et al., 2017). Chronic migraines, which are defined as having a headache at least 15 times a month for three months, are less common (1-5% of migraine sufferers). While not a fatal condition, migraines still contribute to a significant decrease in quality of life and the total estimated cost of all migraine headaches is approximately $78 billion per year (Gooch et al., 2017).
Migraine treatments are classified as abortive (intended to stop a migraine once it begins) or prophylactic (decrease the frequency and/or severity of migraines). Abortive treatments include triptans and ditans (which specifically target serotonin receptors), over the counter medications (that typically include some combination of a nonsteroidal anti-inflammatory drug (NSAID), acetaminophen, and/or caffeine), ergots, and calcitonin gene-related peptide (CGRP) antagonists. By 2026, the market for migraine treatments is estimated to be approximately $12 billion (EvaluatePharma).
Antibodies that target the interaction of CGRP with its receptor represent the newest class of prophylactic treatments and include Aimovig (erenumab), which targets the CGRP receptor, and Ajovy (fremanezumab), Emgality (galcanezumab), and Vyepti (eptinezumab), which each target CGRP. These drugs generated over $1 billion in sales in 2021, and are estimated to generate 2026 sales of $2.2 billion (EvaluatePharma). A summary of their results in Phase 3 clinical trials is shown in the following table.
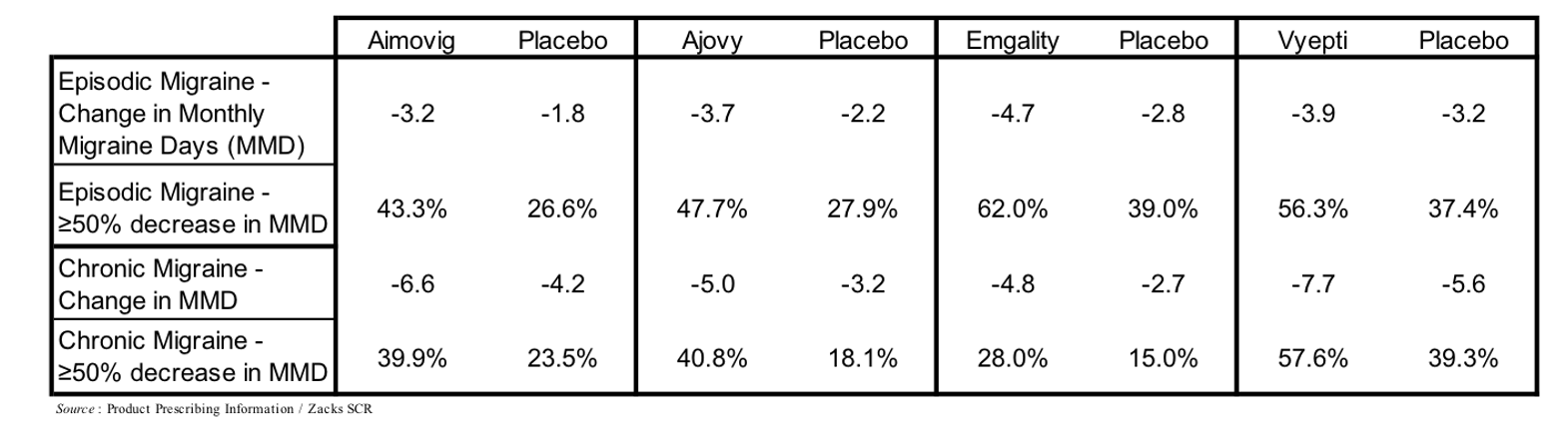
Each of the CGRP-directed antibody treatments are administered as subcutaneous injections or infusions on a monthly or once every three months schedule. Side effects associated with CGRP antibodies are generally mild or moderate, with redness and/or pain at injection site the most common, although due to a number of core systems that CGRP is involved in there are a number of theoretical long-term adverse effects that may arise once the data (≥10 years) is available, including CGRP’s role in hypertension, wound healing, and insulin levels (Robbins, 2020).
Oxytocin in Migraine
While the exact cause of migraines remains to be elucidated, it appears to be associated with the hypothalamus and its connections with the spinal trigeminal nuclei and sensory trigeminovascular system, thus neuromodulators that target the trigeminal pathway could prove effective in migraines.
Oxytocin is a nine amino acid peptide hormone that is involved in a wide array of biological processes, including learning and memory, anxiety, addiction, feeding behavior, maternal behavior, and processing of social information (Cilz et al., 2019). A review of the available literature showed that in 29/33 animal studies oxytocin increased “pain” tolerance (Rash et al., 2014), which suggests it may act as an analgesic. Circumstantial evidence shows that oxytocin may be an effective migraine treatment based on the fact that increased oxytocin levels in pregnancy and during breastfeeding correlates with decreased migraine occurrence (Hoshiyama et al., 2012) and some women who suffer from migraines report that sex (which results in a surge in oxytocin levels) provides at least temporary relief from the condition (Hambach et al., 2013).
While the potential for oxytocin as a treatment for migraine headaches appears promising, getting oxytocin to the trigeminal system is challenging due to the fact that: 1) oxytocin is a small peptide that is broken down almost immediately in the gastrointestinal system; and 2) its half-life in the blood stream is very short (3-5 min) with only limited ability to cross the blood-brain barrier, thus eliminating the availability of oral or parenteral administration. But delivered intranasally, oxytocin permeates the nasal mucosa and is transported along the trigeminal nerve pathway to the trigeminal ganglion where it can inhibit release along trigeminal neurons that synapse within the lining of the dura mater and inhibit CGRP release. Additionally, the most direct way to bypass the blood brain barrier and gain access to the CNS could be nasal administration along olfactory and trigeminal pathways to the brain (Dhuria et al., 2010).
Multiple preclinical studies have examined the potential for oxytocin as a migraine treatment. Oxytocin receptors are present in rat trigeminal neurons, and the vast majority of neurons that express oxytocin receptors also express CGRP (Tzabazis et al., 2016). Intranasal administration of radiolabeled oxytocin in rats results in a very high concentration of oxytocin in all three branches of the trigeminal nerve and trigeminal ganglion (Tzabazis et al., 2017). Administration of nitroglycerin triggers migraine headaches in patients (Sances et al., 2004), thus intraperitoneal injection of nitroglycerin is utilized in a rat model of migraine (Ma et al., 2008). This model results in expression of C-fos in multiple trigeminal neurons (a marker for trigeminal nerve activation), however pre-treatment with intranasal oxytocin markedly reduces the expression of C-fos, thus pointing to a potential role of oxytocin in preventing pain transmission.
A Phase 2 clinical trial of intranasal oxytocin was previously conducted by Trigemina, Inc., from which Tonix acquired TNX-1900 in June 2020. This was a double blind, placebo controlled trial in 218 mostly female migraine sufferers (143 on oxytocin; 75 on placebo) and was conducted in Chile, Australia, and New Zealand (NCT01839149). The trial consisted of a 28-day “run-in” period to establish a baseline of migraine days followed by 56 days of “as needed” dosing with either intranasal oxytocin or placebo. Results showed that while intranasal oxytocin was well tolerated, the study did not meet the primary endpoint of a reduction in migraine headache days from baseline. This was mostly due to an extremely high placebo response rate at the clinical sites in Chile, which was 74%, while subjects from New Zealand and Australia experienced a normal placebo response and showed a statistically significant difference between active and placebo groups (Tzabazis et al., 2017).
Based on results of preclinical studies demonstrating enhanced analgesic activity of oxytocin mediated by the oxytocin receptor in the presence of magnesium, Trigemina developed a proprietary, potentiated formulation of oxytocin containing magnesium. Tonix acquired the intellectual property for this formulation and plans to advance it first in the study of chronic migraine.
To follow up on the results of the prior Phase 2 trial, we anticipate Tonix conducting a similarly designed Phase 2 trial of TNX-1900, which is the magnesium-potentiated formulation of intranasal oxytocin, for the prophylactic treatment of chronic migraine. We expect the trial to be similarly designed to the previous Phase 2 trial with a 28-day baseline period followed by 84 days of dosing. We anticipate the trial initiating in the fourth quarter of 2022.
Developing TNX-601 ER as Abuse Deterrent, Extended-Release Formulation of Tianeptine Oxalate
In July 2022, Tonix announced the development of TNX-601 ER (tianeptine oxalate extended-release tablets) as a naloxone-free formulation of TNX-601, which is designed to be an abuse deterrent treatment for major depressive disorder (MDD), posttraumatic stress disorder, and neurocognitive dysfunction associated with corticosteroid use. Tonix had previously been developing a naloxone-containing tablet, TNX-601 CR, for MDD that was designed to mitigate the risk of parenteral abuse. Tianeptine has weak μ-opioid receptor agonist properties and has been associated with misuse at much higher doses than those reported to be effective in the treatment of MDD (Lauhan et al., 2018).
Tianeptine sodium immediate release tablets have been approved in Europe and many countries in Asia and Latin America for the treatment of MDD for over three decades, however no tianeptine-containing product has been approved by the FDA. The efficacy of tianeptine sodium in comparable to both selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants, while the incidence of sexual dysfunction (which is very high for both SSRIs and tricyclic antidepressants) is lower.
• In a study that compared tianeptine and escitalopram (an SSRI) in patients with MDD, treatment with tianeptine led to more improvements in neurocognitive functions compared with escitalopram (Jin Jeon et al., 2014).
• An eight-week study of tianeptine in elderly patients with MDD utilized escitalopram as an active comparator (Emsley et al., 2018). Results showed that tianeptine improved depressive symptoms and it was well tolerated, with only minimal differences in tolerability from placebo.
• A study examining the prevalence of sexual dysfunction in patients with MDD revealed that treatment with tianeptine was associated with a lower level of sexual dysfunction compared to SSRIs or tricyclic antidepressants (Bonierbale et al., 2003).
• Tianeptine is not subject to first-pass hepatic metabolism, thus there is a decreased likelihood of drug-drug interactions that are common with other MDD treatments (Wagstaff et al., 2001). In addition, its side effect profile is similar to that of SSRIs and minimal when compared to tricyclic antidepressants, including anticholinergic effects.
TNX-601 ER is designed for once-daily dosing, which is in contrast to the three time per day dosing that is required for tianeptine sodium available in Europe and other jurisdictions, which should help to increase patient adherence. We anticipate Tonix initiating a Phase 2 trial of TNX-601 ER in the first quarter of 2023, pending clearance of the Investigational New Drug (IND) application by the U.S. FDA.
Financial Update
On August 8, 2022, Tonix announced financial results for the second quarter of 2022. As expected, the company did not report any revenues for the second quarter of 2022. Net loss available to common shareholders for the second quarter of 2022 was $27.4 million, or $1.22 per share, compared to a net loss available to common shareholders of $23.6 million, or $2.25 per share, for the second quarter of 2021. The weighted average common shares outstanding for the second quarter of 2022 were approximately 22.4 million compared to approximately 10.5 million in the second quarter of 2021.
R&D expenses for the second quarter of 2022 were $16.6 million, compared to $18.1 million for the second quarter of 2021. The decrease was primarily due to decreased non-clinical expenses partially offset by an increase in employee-related expenses. G&A expenses for the second quarter of 2022 were $6.8 million, compared to $5.4 million for the second quarter of 2021. The increase was primarily due to employee-related expenses.
As of June 30, 2022, Tonix had approximately $145.5 million in cash and cash equivalents. As of August 8, 2022, the company had approximately 43.1 million common shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 45.5 million.
Conclusion
The monkeypox outbreak has brought renewed interest in TNX-801 and we look forward to updates from the company on its development. Tonix has a large number of programs that will be entering the clinic over the next 6 to 12 months, and we are particularly excited about the Long COVID program since the company will be focusing on those patients whose symptoms overlap with fibromyalgia. The next major inflection point that investors should be aware of is likely the interim analysis for the Phase 3 RESILIENT study in fibromyalgia, with those results expected in the second quarter of 2023. After accounting for the company’s recent capital raises through the ATM and purchase agreement, our valuation has decreased to $8 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.