By David Bautz, PhD
NASDAQ:IMNN
READ THE FULL IMNN RESEARCH REPORT
Business Update
R&D Event Highlights Company Pipeline
On September 14, 2023, Imunon, Inc. (NASDAQ:IMNN) held a virtual R&D event to highlight the company’s DNA technology and its application in infectious diseases and immuno-oncology (IO). The event featured presentations by company management as well as invited guest speakers Dr. Sallie Permar, who discussed future vaccine technology, and Dr. Patrick Ott, who discussed personalized cancer vaccines. The event can be viewed at the company’s website (link).
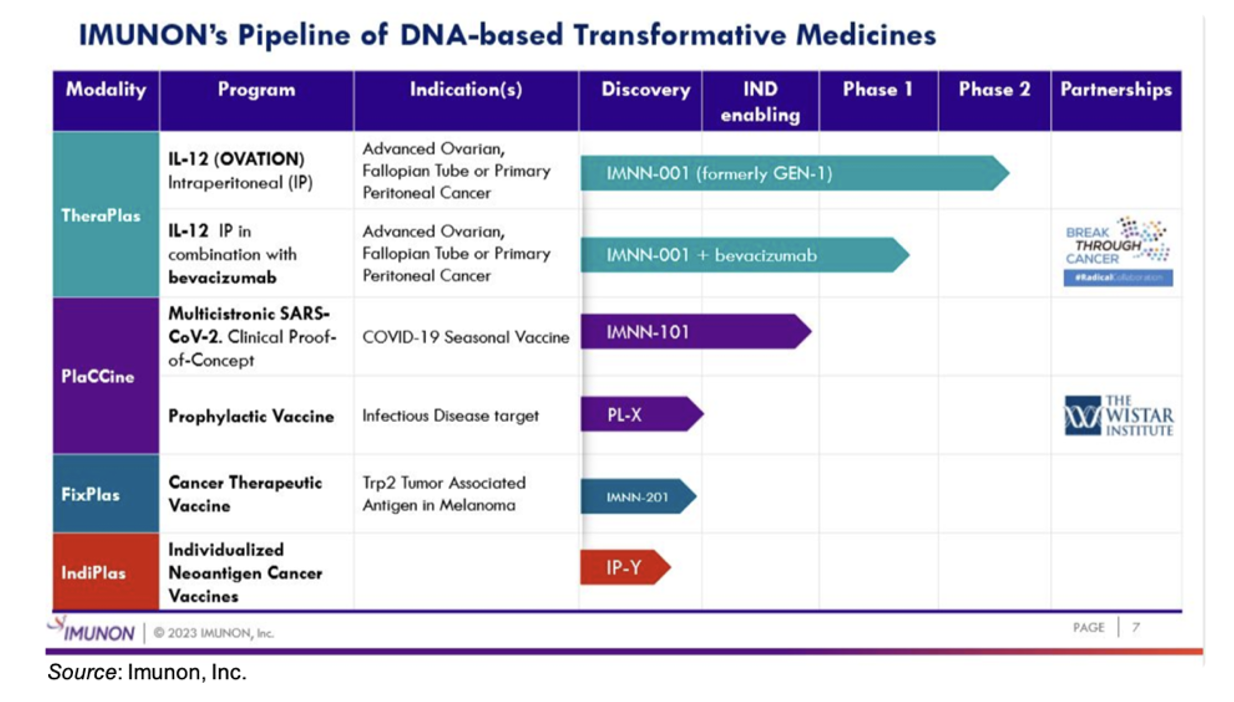
The following image gives an overview of the company’s pipeline. The TheraPlas, PlaCCine, and FixPlas technologies were all discussed during the presentation. A brief overview of the presentation is below.
IMNN-101
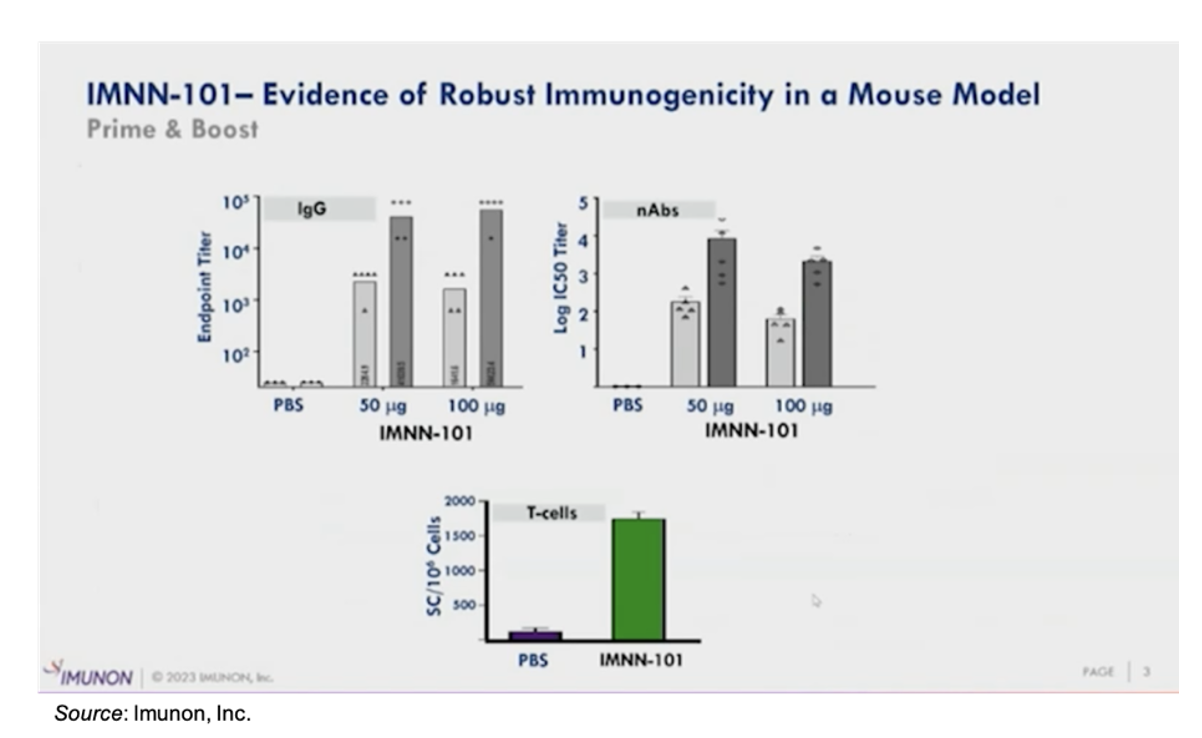
IMNN-101 is the company’s seasonal COVID-19 vaccine candidate that is derived from the PlaCCine platform. It is a DNA vaccine that addresses a number of limitations of current vaccines, including the durability of antigen expression, stability, and flexibility to adapt to mutating viruses. The company has generated pre-clinical data showing robust immunogenicity in mice and complete viral clearance in non-human primates. The following slide shows a robust IgG response, high levels of neutralizing antibody levels, and the T cell response following a prime and boost administration schedule.
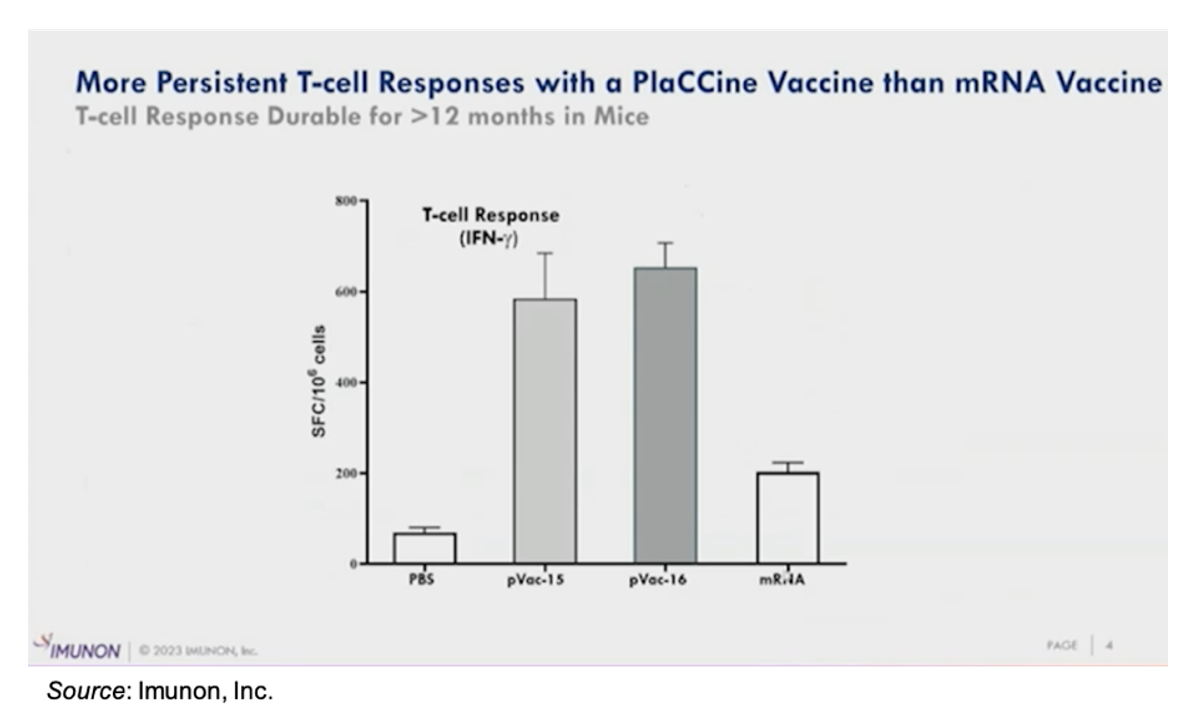
In contrast to the currently approved mRNA vaccines, the following slide shows that the T cell response for the company’s DNA vaccines is more durable. pVac-15 and pVac-16 are two different formulations of IMNN-101 that target different variants of the SARS-CoV-2 virus. Both formulations result in a more robust T cell response than one of the commercially available mRNA Covid-19 vaccines 12 months following administration. This may be due to the persistence of the DNA vaccine compared to mRNA, which results in production of spike protein antigen and its exposure to the immune system for an extended period of time.
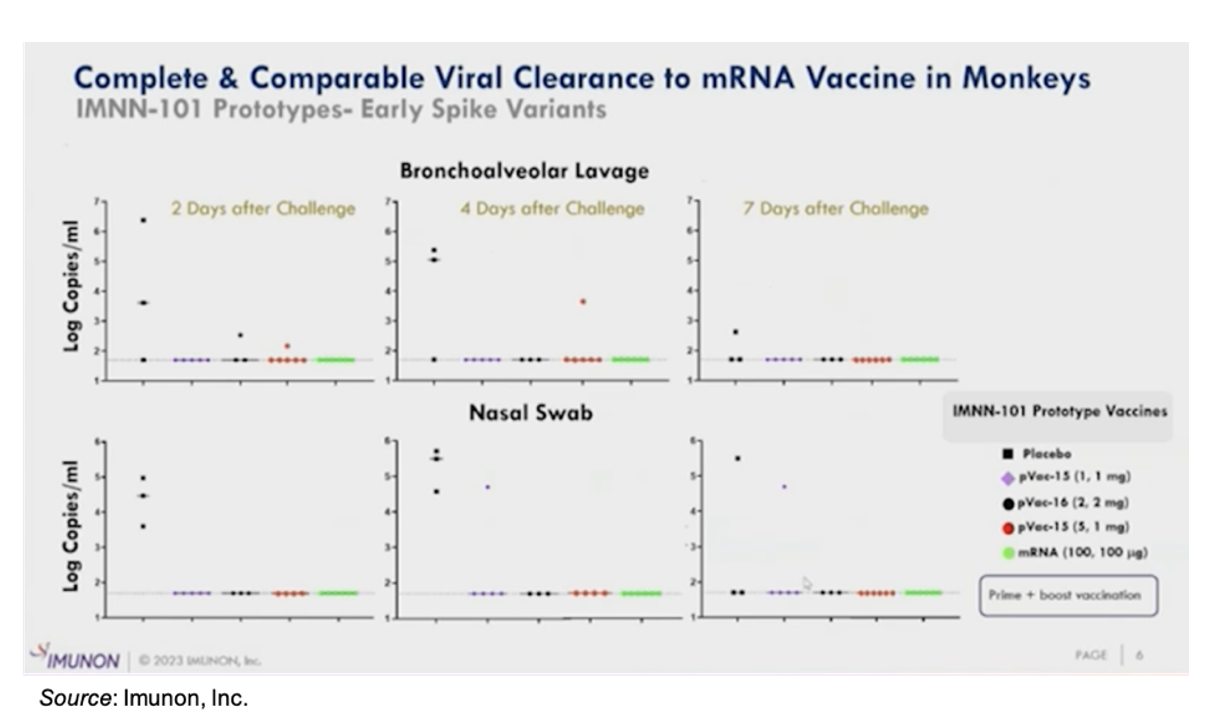
IMNN-101 offers complete viral clearance in non-human primates that is comparable to one of the commercially available mRNA vaccines. The following figure shows viral copies found in bronchoalveolar lavage and nasal swab following immunization with different formulation of IMNN-101. Except for a few rare exceptions, there was no detectable virus found in animals immunized with IMNN-101 following viral challenge.
Lastly, in contrast to mRNA vaccines that must be stored at -80ºC, the PlaCCine vaccines can be stored at 4ºC for an extended period of time (at least 12 months) with little effect on IgG response, as shown in the following figure. This is a considerable advantage, particularly for those areas that may not have the infrastructure to support ultra-low temperature storage.
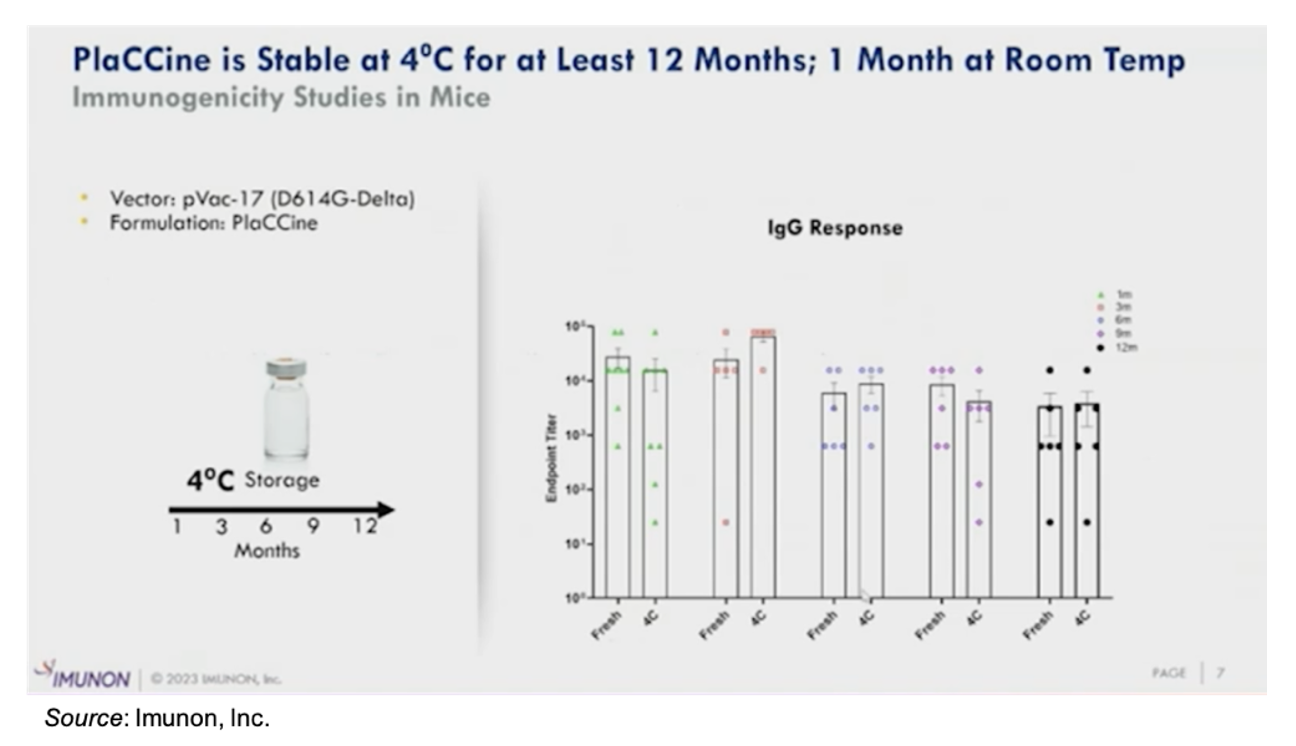
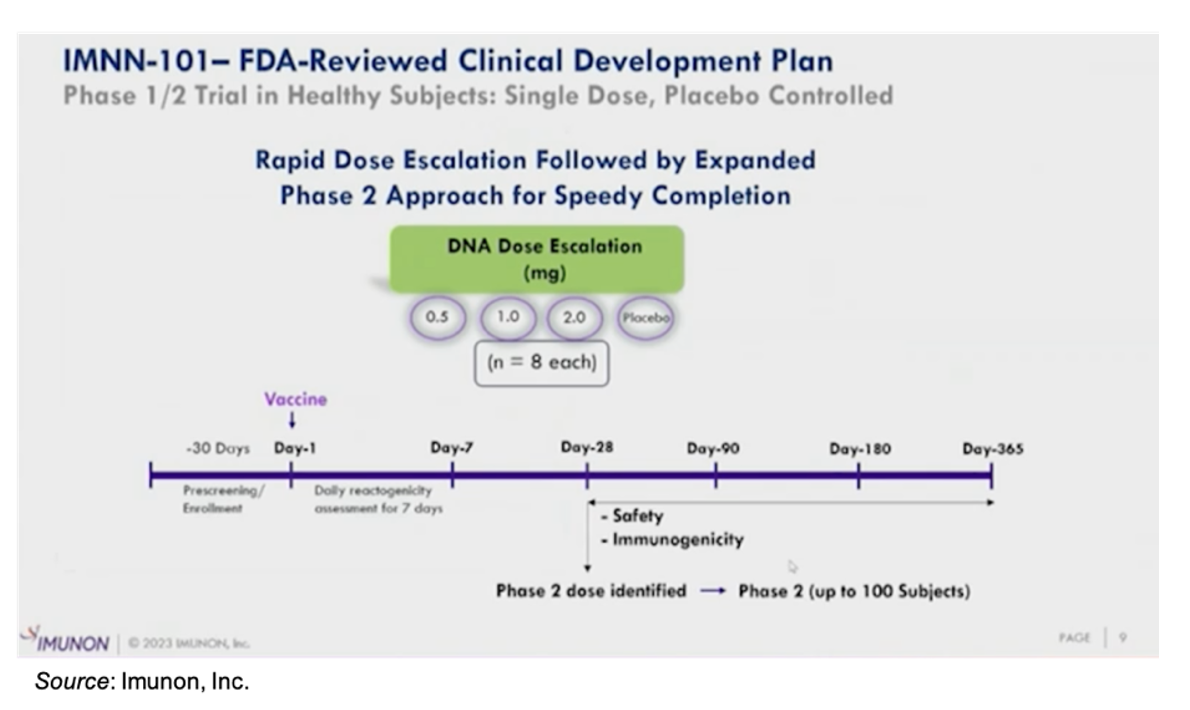
IND-enabling studies for the company’s seasonal COVID-19 vaccine will commence in the fourth quarter of 2023. We anticipate an IND filing in the first quarter of 2024 and a Phase 1/2 clinical trial to commence shortly thereafter. The following slide gives an overview of the proposed Phase 1/2 trial, which will include a Phase 1 dose escalation portion such that a dose can be rapidly identified to advance into a Phase 2 trial.
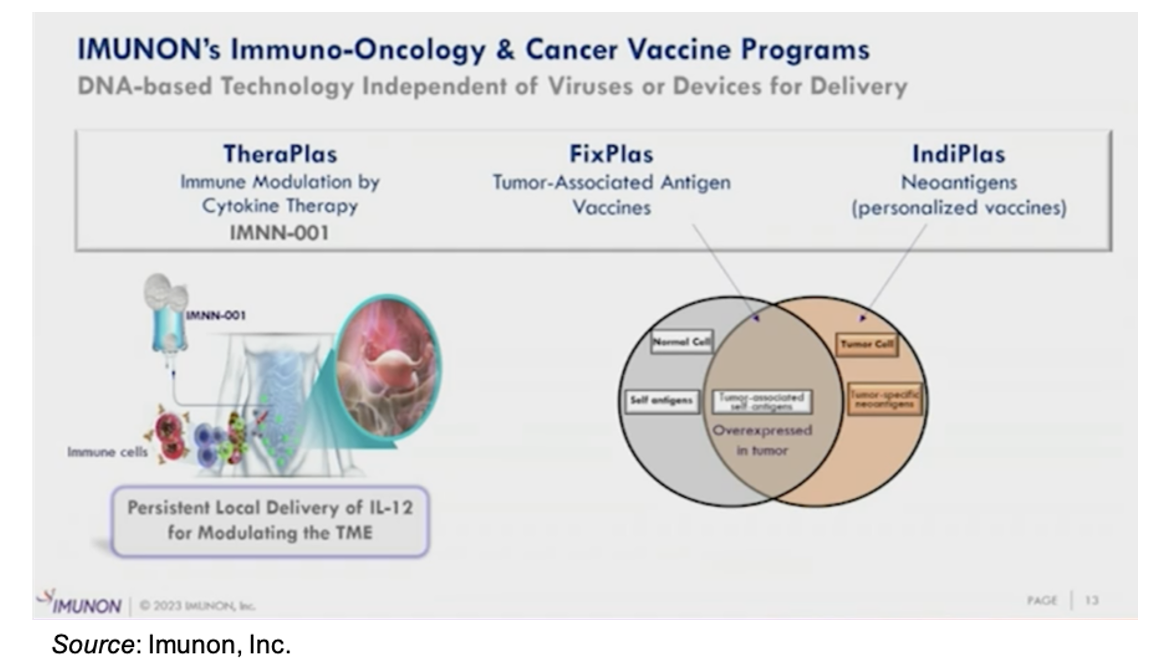
Immuno-Oncology (IO) Technology
Imunon’s IO technology consists of three separate platforms: TheraPlas, which involves immune modulation by cytokine therapy; FixPlas, which is a cancer vaccine that targets tumor associated antigens; and IndiPlas, which is a personalized cancer vaccine that targets neoantigens.
IMNN-001 is the lead development compound from the TheraPlas platform. It consists of a DNA plasmid for the safe and effective delivery of the cytokine Interleukin (IL)-12. IMNN-001 has completed five clinical trials and is currently being tested in the Phase 2 OVATION trial in patients with ovarian cancer along with a Phase 1/2 clinical trial in combination with bevacizumab in ovarian cancer patients. We anticipate a data read out for the OVATION trial in the second quarter of 2024.
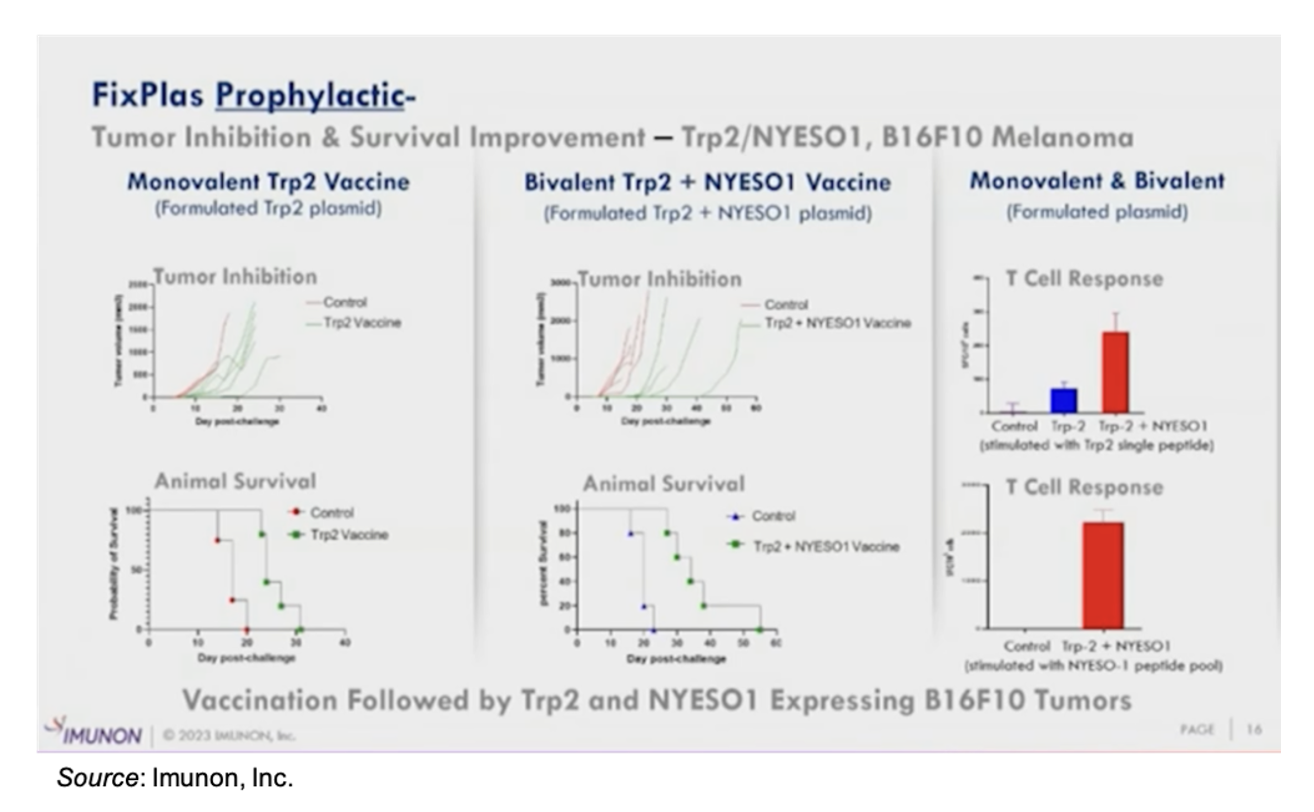
The FixPlas platform consists of a DNA vaccine that is designed to target tumor-associated antigens (TAAs) and can be utilized in both monovalent and bivalent format. The company has completed pre-clinical studies examining the FixPlas technology as a prophylactic in a mouse model of melanoma and is currently evaluating it as a therapeutic. The following slide shows results from the prophylactic study, in which the TAA Trp2 was evaluated both as a monovalent vaccine as well as a bivalent vaccine in combination with NYESO1. The results showed extended survival for animals immunized with the vaccine along with a strong T cell response.
The development plan for FixPlas will involve the selection of the disease target in the fourth quarter of 2023 followed by additional preclinical studies in the second and third quarter of 2024. We anticipate IND-enabling studies to initiate in the third quarter of 2024 with the current plan calling for a Phase 1 clinical trial to initiate in the first quarter of 2025.
Conclusion
We invite investors to watch the full R&D day presentation, as it gives a very good overview of the technologies that Imunon is developing along with anticipated timelines. We look forward to additional updates from the company as it advances both IMNN-101 and the FixPlas technology toward clinical trials. Our valuation currently stands at $15 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.