By David Bautz, PhD
NASDAQ:KMDA
READ THE FULL KMDA RESEARCH REPORT
Financial Update
On May 24, 2023, Kamada Ltd. (NASDAQ:KMDA) announced financial results for the first quarter of 2023. Kamada reported revenues of $30.7 million, which was a 9% increase over the first quarter of 2022. The revenue consisted of $24.1 million from proprietary products and $6.6 million from the distribution business. Gross profit and gross margins were $11.8 million and 39%, respectively, in the first quarter of 2023, compared to $11.3 million and 40%, respectively, in the first quarter of 2022. The cost of goods sold for the proprietary products in the first quarter of 2023 included $1.3 million in depreciation expenses associated with intangible assets generated through the IgG products acquisition. Excluding such intangible assets depreciation would have resulted in gross profit and gross margins of $13.2 million and 43%, respectively.
Operating expenses in the first quarter of 2023 (which includes R&D, Sales & Marketing [S&M], G&A, and other expenses) totaled $11.6 million compared to $11.1 million in the first quarter of 2022. During the first quarter of 2023, Kamada conducted a planned workforce reduction at its Israeli plant to optimize staff level to its capacity needs. This resulted in an expense of $0.6 million for excess severance remuneration. The downsizing is expected to result in an annualized reduction of approximately 6% in the overall Israeli labor costs. Net loss for the first quarter of 2023 was $1.8 million, or $0.04 per share, which was in line with a net loss of $1.8 million, or $0.04 per share, in the first quarter of 2022. Excluding depreciation expenses of intangible assets and finance expenses of the contingent consideration and other assumed long-term liabilities associated with the acquired IgG products, Kamada would have recorded net income of $1.7 million, or $0.04 per share, in the first quarter of 2023 compared to $1.9 million, or $0.04 per share, for the first quarter of 2022. Adjusted EBITDA, as shown in the following table, was $3.8 million in the first quarter of 2023 compared to $3.3 million in the first quarter of 2022.
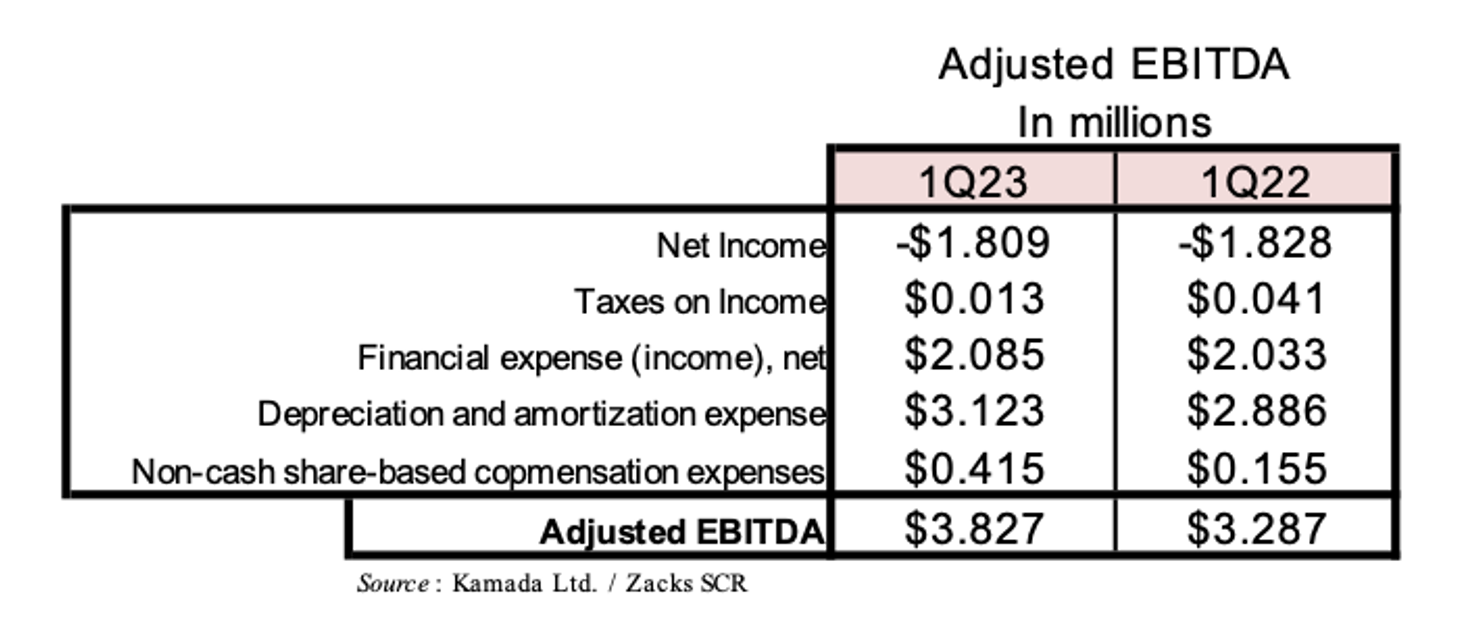
Kamada exited the first quarter of 2023 with $27.1 million in cash, cash equivalents, and short-term investments. This does not include the anticipated net proceeds from the recently announced $60 million financing, which we expect to close in the second half of 2023 (discussed below).
For the remainder of 2023, we continue to model for the company to have total revenues of $142 million, which is at the mid-point of the company’s reiterated guidance of $138 million to $146 million. This would be a $13 million (10%) increase over 2022 revenues. We model for EBITDA of $24 million, which is also at the mid-point of the company’s guidance of $22 million to $26 million. This would be an approximately $6 million (35%) increase over EBITDA in 2022. We view the large increase in EBITDA along with the topline revenue growth as indicative of strong business fundamentals.
$60 Million Financing with FIMI Opportunity Funds
On May 24, 2023, Kamada announced a share purchase agreement with FIMI Opportunity Funds, the leading private equity firm in Israel and a major shareholder of Kamada. The purchase agreement calls for Kamada to issue 12.6 million shares to FIMI at a price of $4.75 per share (which represents the average closing price of the company’s shares on NASDAQ during the 20 trading days prior to the date of the purchase agreement). Upon closing of the transaction, FIMI is expected to beneficially own approximately 38% of Kamada’s ordinary shares and will become a controlling shareholder of the company, within the meaning of the Israeli Companies Law, 1999.
This is an exciting development for the company and shows the confidence that FIMI has with the business. The transaction will help to strengthen the company’s balance sheet and will allow for increased financial flexibility through accelerating the growth of the existing business while also allowing for the pursuit of additional business development opportunities.
Business Update
Phase 3 InnovAATe Trial Continues Enrolling Patients
Kamada initiated the Phase 3 InnovAATe trial of inhaled alpha-1 antitrypsin (AAT) in December 2019 (NCT04204252). It is a randomized, double blind, placebo controlled pivotal Phase 3 trial designed to assess the efficacy and safety of inhaled AAT in patients with AATD and moderate lung disease. Up to 250 patients will be randomized 1:1 to receive either 80 mg inhaled AAT or placebo daily for two years. The primary endpoint of the trial is lung function as measured by FEV1. Secondary endpoints include lung density changes as measured by CT densitometry, along with other parameters of disease severity such as pulmonary function, exacerbation rate, and six-minute walk test.
Enrollment rates were very slow through 2021 due to the COVID-19 pandemic, however in 2022 the company opened up additional sites in Europe and there are currently 7 sites recruiting patients. Thus far, 60 patients have been enrolled in the trial and the independent data safety monitoring board (DSMB) recently recommended that the trial continue without modification for the fifth time since the study initiated.
Kamada's Inhaled AAT, if approved, will be competing in a market which is expected to be approximately $1.5 billion by 2028 (EvaluatePharma).
Conclusion
Kamada’s business continues to perform very well and we are encouraged that the full year 2023 revenue guidance has remained the same. The $60 million financing is an interesting development, and we will be curious to see how Kamada utilizes the proceeds from the private placement. With revenue and profitability numbers mostly in line with our expectations we have not made any adjustments to our model and our valuation remains at $14.00 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.