By John Vandermosten, CFA
NASDAQ:BIOR
READ THE FULL BIOR RESEARCH REPORT
Biora Therapeutics, Inc. (NASDAQ:BIOR) reported first quarter results encapsulating a period during which it has initiated its BT-600 Phase I study, added new collaborators to its BioJet program, completed several cohorts of its clinical study and improved its capital structure. Following the end of the quarter, Biora shared interim results from the BT-600 trial, raised additional capital and completed the multiple-ascending dose portion of the BT-600 trial. Other highlights include the issuance of a new patent and participation in Digestive Disease Week on May 19th. Based on the preclinical work that has been completed so far with collaborators, management is optimistic that they will see a partnership emerge this year.
First Quarter Financial and Operational Results
Biora reported first quarter 2024 results in a press release and Form 10-Q filing with the SEC on May 15th. A conference call was held to discuss results with investors following the release. For the quarter ending March 31, 2024 revenues of $542,000 were recognized. Net loss for 1Q:24 totaled ($4.2) million or ($0.14) per share. Operational expenses rose 3% to $16.1 million on higher consulting and professional fees. For the first quarter 2024 versus the same prior year period:
➢ Revenues were $542,000 up from $2,000 the prior year due to an increase in collaboration revenue;
➢ Research and development expenses totaled $7.0 million, down 3% from $7.2 million on lower consulting and professional fees and supply costs partially offset by an increase in clinical trial expenses and salaries and benefits;
➢ General & Administrative expenses were $9.1 million, up 8% from $8.4 million as a result of increased consulting and professional fees offset by a decrease in in business insurance, salaries and benefits, and facilities costs;
➢ Interest expense was ($2.8) million compared to ($2.7) million with the change attributable to a decrease in the balance of the 7.25% convertible senior notes due 2025 from note exchanges, offset by issuance of 2028 Convertible Notes;
➢ Other income was $14.1 million vs. $783,000 with the increase almost entirely related to a gain on warrant liabilities resulting from mark to market and equity share price declines;
➢ Income tax was $48,000 vs. $0;
➢ Net loss was ($4.2) million vs. ($17.4) million or ($0.14) and ($1.59) per share, respectively.
As of March 31, 2023, cash totaled $10.8 million. This amount compares to the $15.2 million cash balance held at the end of 2023. Multiple transactions took place in financing including contributions from issuance of common stock and convertible notes offset by offering costs and finance costs for insurance premiums. Debt is carried on the balance sheet at $45.3 million, which consists of short-term and long-term convertible notes. Biora also monetized its interest in Enumera Molecular which contributed an additional $3 million to the cash pile. Following the end of the quarter, Biora closed a $6 million registered direct offering.
New Patent Issue Notification
The US Patent and Trademark Office (USPTO) provided an issue notification for a new NaviCap patent to deliver a Janus kinase (JAK) inhibitor to the gastrointestinal tract. As reported, US Patent Application 17/313,339, entitled “Treatment of a Disease of the Gastrointestinal Tract with a JAK Inhibitor and Devices,” will issue as US Patent #11,857,669 on January 2, 2024 following payment of fees. JAK inhibitors such as tofacitinib present a variety of potential side effects such as infections, liver damage, blood cell count changes, blood clots, and pregnancy risks. Patients taking these medications require close monitoring. Direct delivery to the diseased tissue may allow for improved efficacy compared with systemic delivery and reduced side effects through the use of a lower dose.
BT-600 Program
IND Clearance for BT-600
Following an investigational new drug (IND) submission in September 2023 for the NaviCap BT-600 program, the FDA responded with questions in late October. The data the agency sought for the clinical trial evaluating the safety of tofacitinib delivery to the small intestine for ulcerative colitis (UC) was readily available and resubmitted to the agency a week later. On November 30, the FDA cleared Biora’s IND application for BT-600, allowing the study to begin. An announcement of the trial’s initiation was made in January. The Phase I will directly deliver tofacitinib to the colon for the treatment of moderate to severe UC. Data from the study is expected to produce data related to the pharmacokinetic and pharmacodynamic effects of BT-600. In late February, Biora announced that the single ascending dose (SAD) cohorts had been completed and that the multiple ascending dose (MAD) cohorts would begin. The MAD portion is designed to enroll 24 participants to receive BT-600 tofacitinib at 5 and 10 mg or placebo. By the end of April 2024, the MAD cohorts had been completed and management guided towards a late June report of full study data.
BT-600 Trial Interim Readout – Single Ascending Dose
The BT-600 trial, listed under the NCT06275464 identifier, was launched in January as a randomized, double-blind, placebo-controlled Phase I study to evaluate the safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of BT-600 when administered in single and multiple ascending doses to healthy participants. The NaviCap device used a proprietary formulation of tofacitinib to deliver either 5 mg, 10 mg or placebo to healthy patients.
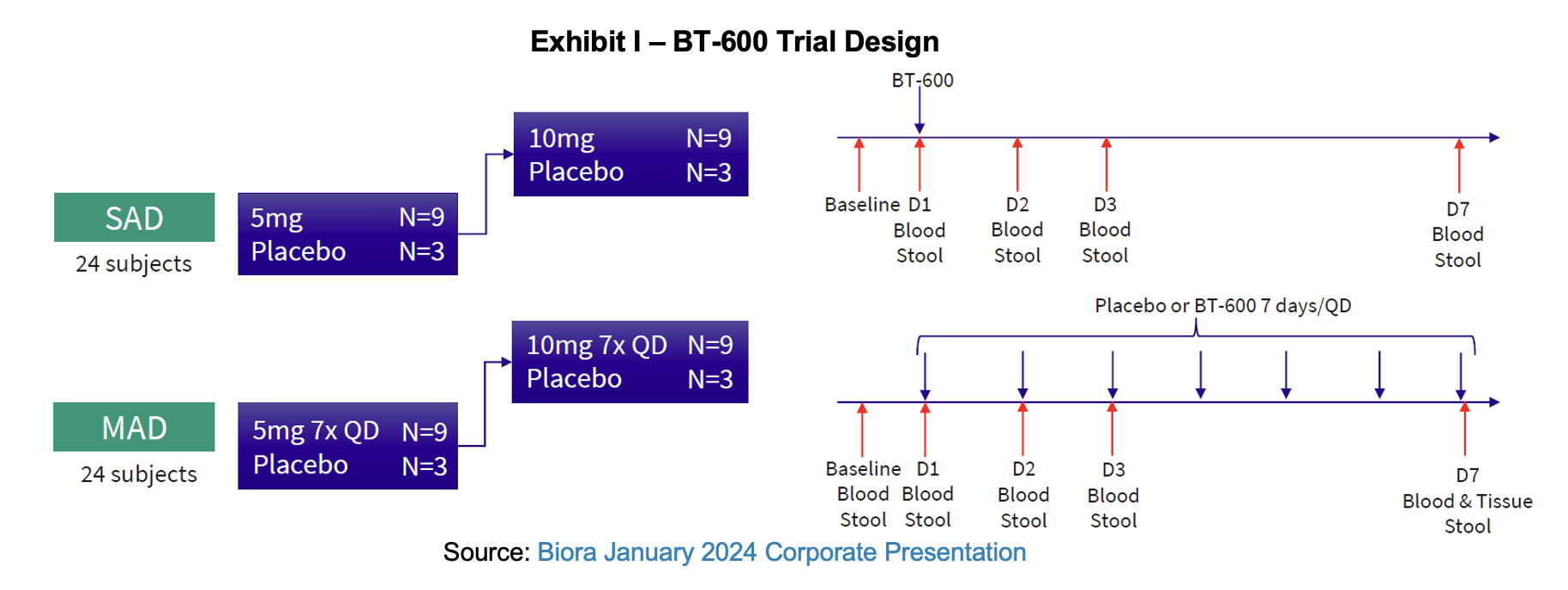
The single dose data is consistent with PK and safety data generated in previous studies. The product was deemed to be well tolerated and functioned as intended and designed with drug release as predicted. Devices carrying drug demonstrated corresponding systemic absorption and delivery to the colonic tissue. Measurable tofacitinib in the blood was observed at approximately 6 hours and maximum concentration at 8 hours. This is in contrast to the conventionally orally delivered tofacitinib which reaches its maximum concentration within the first hour. Based on measurements taken during the study, delivery to the colon vs. the upper gastrointestinal tract was confirmed. Dose proportional PK was observed between the 5 mg and 10 mg doses. NaviCap’s goal is to achieve higher concentrations of drug in the target tissue and lower levels systemically throughout the body compared with conventional administration. Further studies later this year are expected to extract biopsies to confirm tissue concentration.
SAD Interim Results from BT-600
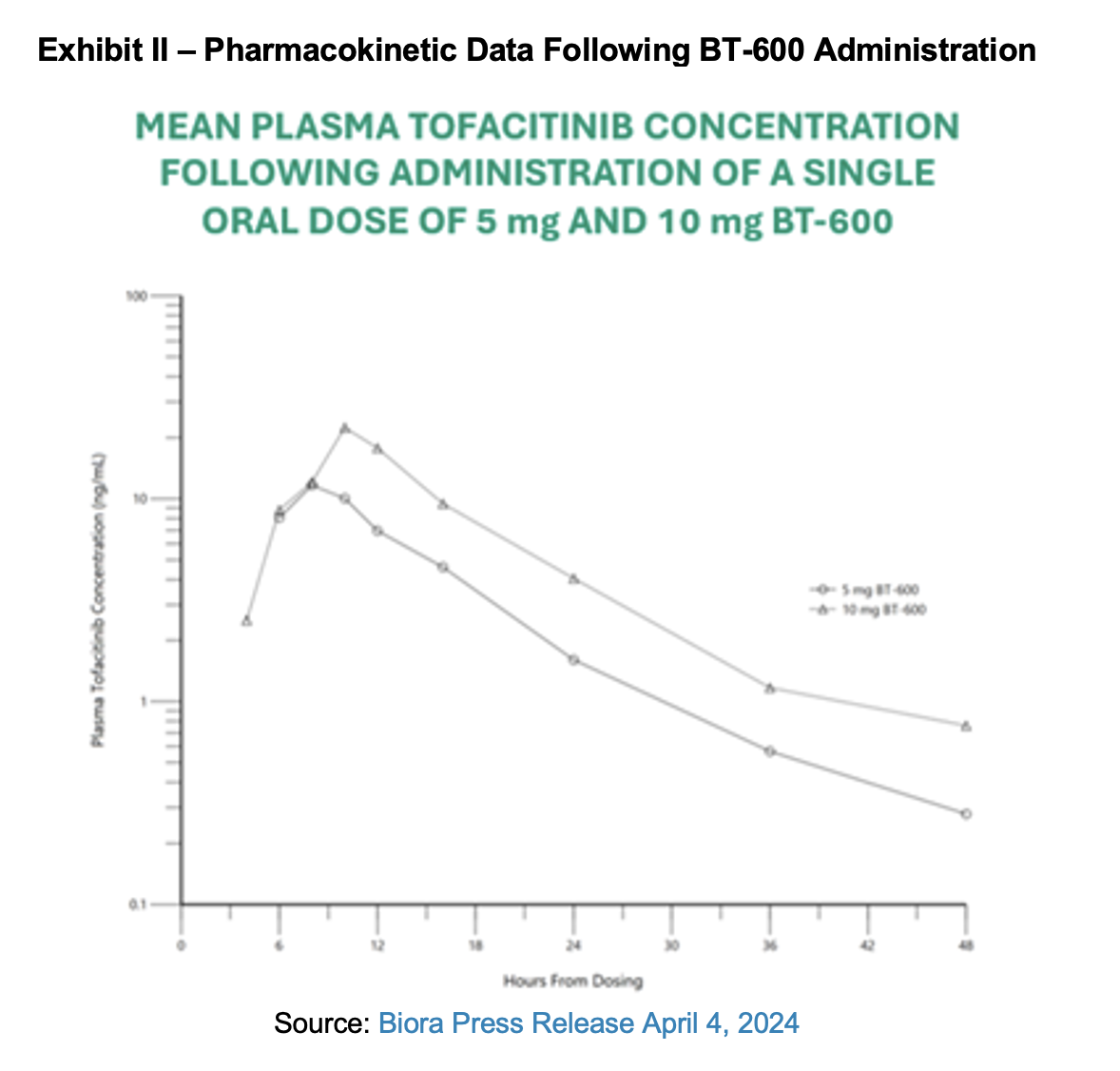
Biora provided additional data for its BT-600 study in an April 4th press release. Clinical data indicated that all pharmacokinetic endpoints were met by all study participants. The drug-device combination was well tolerated with no serious adverse events and all devices performed as intended. All participants showed systemic drug absorption. Six hours after administration, tofacitinib was detected in subject plasma and TMAX was achieved 8-10 hours after first administration of BT-600.1 Tofacitinib was measured in fecal samples of all subjects, further confirming delivery of drug to the colon.
Importantly, delivery of tofacitinib with BT-600 resulted in a 65% to 75% reduction in systemic absorption of the Janus kinase (JAK) inhibitor. Maximum plasma concentration was 26 ng/mL for BT-600 at the 10 mg dose of tofacitinib compared with 88 ng/mL for 10 mg of conventionally administered oral tofacitinib.
The MAD portion of the study began after the SAD and was conducted to confirm study results. Patients received one daily dose of 5 mg, 10 mg or placebo. The dose will be one-half the amount administered as per the drug label for tofacitinib. The MAD portion of the trial evaluated daily doses of BT-600 for seven days at 5 mg and 10 mg tofacitinib or placebo, in healthy adult participants. Results are expected to be shared with stakeholders in June 2024.
In the second half of the year, another study is planned that will evaluate the use of NaviCap tofacitinib in UC patients to further confirm plasma and tissue PK and PD and to further inform subsequent trials.
BioJet Collaboration
Biora announced a new collaboration for its BioJet Systemic Oral Delivery platform last December. The new agreement provides funding to test the BioJet platform’s ability to achieve bioavailability through oral delivery of the collaborator’s molecules to the small intestine in animal models. Based on our conversations with management, we believe that several of the company’s other large pharma relationships are nearing the end of their collaborations and possibly close to securing a partnership which may include upfront payments. BioJet allows for large molecule drugs to be delivered orally, bringing a number of benefits including administration convenience and daily dosing which contrast with the more frequently used infusion method.
A January 2nd press release announced that BioJet has achieved an important bioavailability hurdle in a collaboration with AstraZeneca, mentioning the collaborator’s name for the first time. The preclinical study generated greater than 25% bioavailability compared to subcutaneous delivery with a less than 50% coefficient of variation. In this study, the BioJet device was administered to a porcine model in comparison with subcutaneous administration. BioJet devices were delivered endoscopically and released for autonomous activation. These results build upon other preclinical work evaluating a variety of molecules in animal models. The latest data from BioJet will be presented in Boston at the Next Gen Peptide Formulation & Delivery Summit in June 2024.
BioJet Patent
On April 8th, Biora announced that the US Patent and Trademark Office (USPTO) had issued US Patent #11,938,295 titled Ingestible Device for Delivery of Therapeutic Agent to the Gastrointestinal (GI) Tract. The patent makes claims to cover jet parameters for liquid jet delivery of drug to the GI tract, stomach or small intestine. The device described in the application is configured to provide at least three different modes of direct delivery of therapeutic agents to the GI tract of a subject, referred to herein as trans-epithelial, epithelial, and topical delivery. Direct delivery, as used in the patent application, refers to a force-driven delivery mechanism. The patent is anticipated to expire at the end of 2040, unless extensions are granted.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
1. Literature regarding oral administration of tofacitinib shows maximum concentration is achieved at 0.5 to 1.0 hours after administration.