By John Vandermosten, CFA
NASDAQ:ACHV
READ THE FULL ACHV RESEARCH REPORT
Achieve Life Sciences, Inc. (NASDAQ:ACHV) reported its second quarter 2025 results and highlighted several milestones achieved since the last quarterly update. This includes forging a partnership with Omnicom for launching cytisinicline, submitting the New Drug Application (NDA), generating necessary safety exposure data for the NDA and publishing ORCA-3 results in the Journal of the American Medical Association (JAMA).
During its second quarter earnings conference call, management elaborated on the ORCA-OL trial and commercialization preparation efforts. As a follow up to the trial, a survey was given to participants who reported very high levels of satisfaction and success with cytisinicline. On the commercialization front, new members have been added to the team to help with the initiative emphasizing availability, access and awareness. Looking ahead, management expects to have the NDA submission accepted by September and to remit ORCA-OL safety data in October.
Financial and Operational Results
Achieve’s second quarter 2025 financial and operational results were provided in a press release, filing of Form 10-Q and a webcast which provided an opportunity for analysts to ask questions. No revenues were reported in 2Q:25. Operating expense was $12.6 million producing a net loss of ($12.7) million or ($0.37) per share. For the quarter ending June 30th, 2025 and versus the same comparable prior year quarter:
- Research & development expense totaled $6.7 million, up 31% from $5.1 million, due to the initiation and enrollment of the ORCA-OL safety trial in May 2024;
- General & administrative expense was $5.8 million, up 82% from $3.2 million on higher employee expenses and commercial launch preparation;
- Net other expense was ($155,000) vs. ($30,000) as interest income offset interest expense and minor miscellaneous items including other expense and change in fair value comprised the remainder;
- Net loss was ($12.7) million vs. ($8.5) million or ($0.37) and ($0.25) per share, respectively.
As of June 30th, 2025, cash and equivalents totaled $55.4 million. This compares to a $34.4 million balance held at the end of 2024. Achieve carries convertible debt of $9.9 million on the balance sheet which includes accrued interest. In July 2024, the company refinanced its debt agreement with Silicon Valley Bank (SVB), extending maturity until the end of 2027. Achieve is eligible to draw an additional $5 million from the facility following FDA acceptance of its NDA, which we expect in September. Cash used in operations during 2Q:25 was ($9.1) million versus ($4.9) million in the same prior year period. On the last day of the second quarter, Achieve closed a capital raise which generated net proceeds of $41.3 million. Following the end of the reporting period, on July 25th, 2025, the underwriters exercised their option to sell 1.4 million additional shares at $3.00. We estimate this generated just under $4 million in net proceeds that add to the June 30 cash balance.
Public Offering
On June 30th, 2025 Achieve closed its $45 million capital raise. 15 million shares were issued at $3.00, each of which included an attached warrant exercisable at $3.00 per share. An additional 1,766,666 warrants were issued upon the partial exercise by the underwriters of their option to purchase additional shares, bringing total issued warrants to approximately 16.8 million. Net proceeds from the capital raise are estimated to be $41.3 million as disclosed in the June 27th Form 8-K filing. On July 25th, 2025, the underwriters exercised their option to purchase an additional 1,419,896 shares at $3.00 per share.
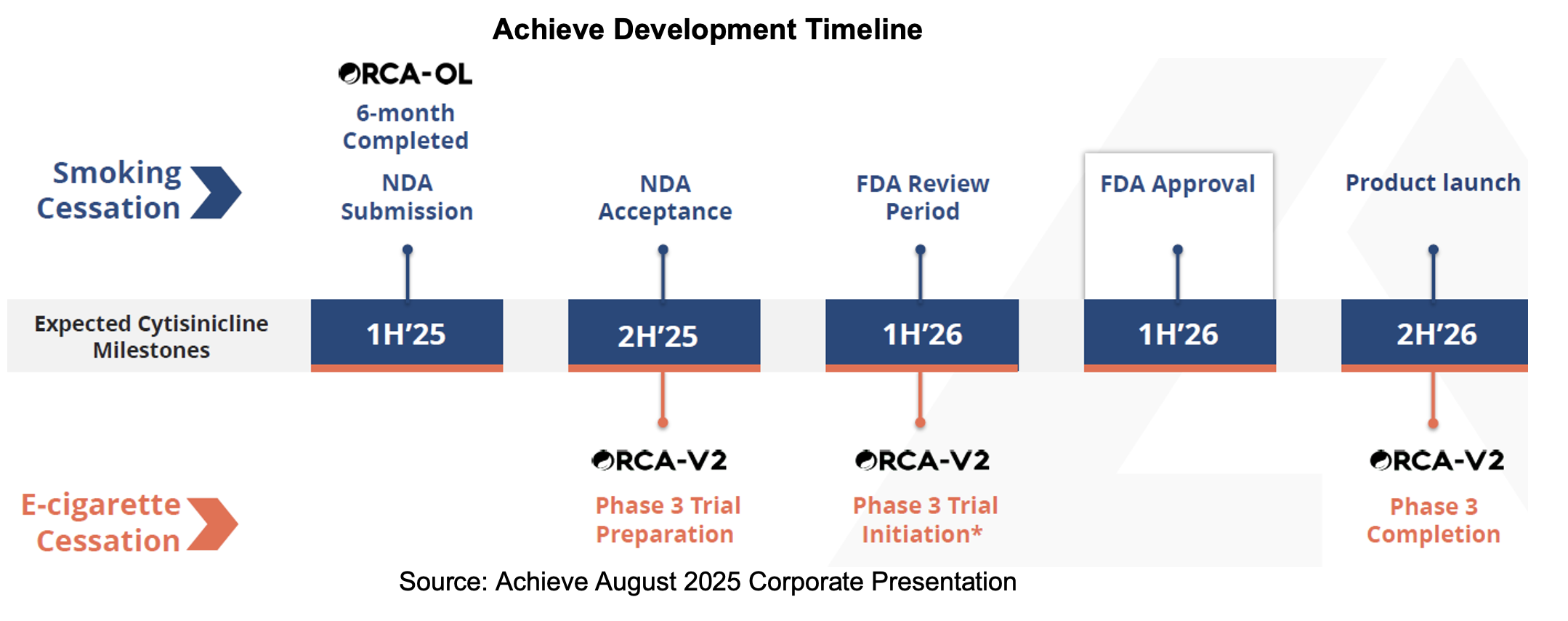
NDA Submission
Achieve announced its NDA submission of cytisinicline for smoking cessation in a June 26th press release. The company conducted two Phase III studies, an open label safety study and other studies that evaluated over 2,000 participants with the results demonstrating the safety, efficacy and tolerability of cytisinicline. As per management guidance, we expect to see acceptance of the NDA in September and one year ORCA-OL safety data to be shared with the agency in October.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.