By David Bautz, PhD
NASDAQ:BCLI
READ THE FULL BCLI RESEARCH REPORT
Business Update
Citizen’s Petition Filed Requesting FDA Approve NurOwn™
On July 3, 2025, a Citizen’s Petition was filed with the U.S. Food and Drug Administration in which the petitioners, a group consisting mostly of amyotrophic lateral sclerosis (ALS) patients who have previously received NurOwn treatment, are requesting 1) that the agency invite BrainStorm Cell Therapeutics, Inc. (NASDAQ:BCLI) to re-submit its Biologics License Application (BLA) for NurOwn, and 2) request that the FDA exert its “regulatory flexibility” and grant Accelerated Approval or new Conditional Approval of NurOwn for the treatment of ALS with a Phase 4 confirmatory study with requirement of patients’ participation in a biorepository with a natural history and exposome database. The full 300+ page petition has been assigned Docket ID FDA-2025-P-2109 and can be accessed here.
We spoke with BrainStorm’s management and they confirmed that the company was not involved in crafting the petition and they only recently learned about it. Based on how large and extensive the document is we estimate it has likely been in production for at least a year.
The petition includes all of the data that has been published on NurOwn along with new data compiled from patients that participated in the Expanded Access Program (EAP), including:
- 100% Five-year survival compared to an expected 20% five-year survival rate based on ALS natural history data
- Overall survival (OS) improved by 5.5 months compared to matched controls as of 2022
- Tracheostomy-free survival that was as much as five years longer than median ALS natural history
- Extended periods of Progression-free survival (PFS)
- Long-term slowing of ALS progression by as much as 85%
- Significant impact on respiratory function as measured by delayed time-to-event for non-invasive ventilation (NIV)
While a full summary of the petitioner’s arguments is beyond the scope of this report, we will highlight what we believe to be the most salient data points below.
Expanded Access Program Data
Since the Advisory Committee meeting for NurOwn in September 2023, the petitioners have compiled data for the ten participants in the EAP using multiple data sources. The following table summarizes the Overall Survival (OS) data for those ten participants.
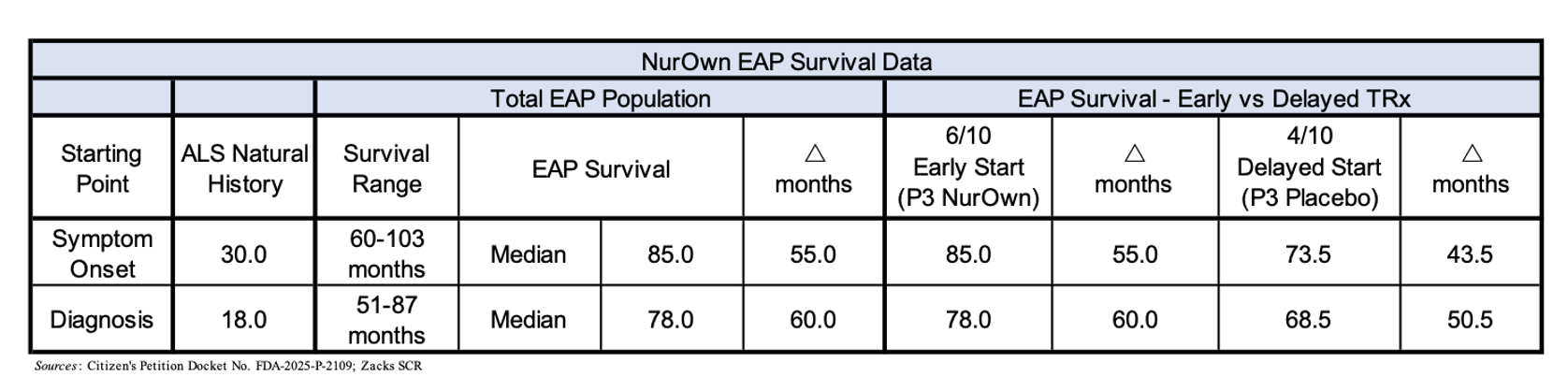
Included in the table is the expected median survival data for an ALS patient from the first symptom onset (30 months) and diagnosis (18 months) (Traxinger et al., 2013; Knibb et al., 2016). The median overall survival for the ten participants in the EAP was 85.0 months from the first symptom onset and 78.0 months from diagnosis, both of which are immense improvements over what would be expected in a group of ALS patients. In addition, all 10 patients lived for longer than five years.
To corroborate the survival data, we performed an examination of various publicly available information sources, which shows that life expectancy for those diagnosed with ALS is typically 2-5 years from when symptoms first appear, however there is a wide degree of variability:
- Typical survival times from onset of symptoms appears to be influenced by ethnicity and can range from 24 months for those from Northern Europe to 48 months to those from Central Asia (Marin et al., 2016).
- The Centers for Disease Control estimates that “most people with ALS live on average between 2 to 5 years after symptoms develop” (CDC National ALS Registry).
- The National Institute of Neurological Disorders and Stroke (NINDS) states that “most people with ALS die…usually within three to five years of symptoms first appearing. But about 1 in 10 people survive for 10 years or more.” (NINDS ALS).
Specifically examining the rate of patients that survive greater than five years shows that:
- A 2012 review cited multiple ALS studies with a median survival of 20-36 months for population-based studies, five-year survival rates ranging from 10-20%, and 5%-10% of ALS patients surviving for more than 10 years (Chio et al., 2009).
- A study out of Olmsted County, Minnesota found that of the 94 patients that were diagnosed with ALS between 1925 and 2004, 14% survived for five years or more following symptomatic onset while the mean survival for all 94 patients was 2.95 years (Mateen et al., 2010).
Based upon the above data, it appears very unlikely that 10/10 ALS patients in the EAP surviving for greater than five years occurred by chance. These real-world data represent a clinically meaningful observation and make a compelling argument that NurOwn is having a positive impact on the EAP patient’s disease trajectory.
In addition to the impressive OS data, the data for “tracheostomy-free” survival (e.g., the time until a patient is put on a breathing machine through a tracheostomy) is perhaps even more impressive. The OS data shown above includes the fact that all 10 participants in the EAP were living “trach-free” a minimum of five years from symptom onset. Four of the EAP participants have passed away after hitting the five-year survival threshold, however even those individuals lived a median of 5.5 years trach-free from symptom onset, which is nearly three years longer than the expected median trach-free survival.
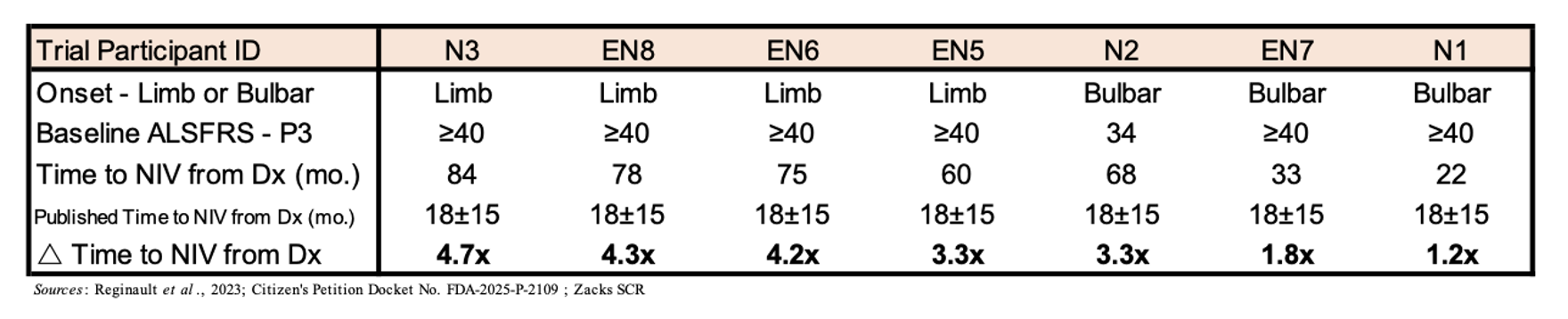
The leading cause of death in ALS patients is respiratory failure, which is caused by the loss of motor neurons in the phrenic nerve, which innervates the diaphragm and is critical for respiratory function. Thus, the ability to avoid assisted breathing, whether in the form of non-invasive ventilation (NIV) or tracheostomy, improves both the patient’s quality of life and potentially their OS. In the table below are data compiled for seven EAP participants showing the time to NIV from diagnosis. While there is limited data from large studies regarding the median time to NIV, a 2023 study from France showed that the mean time from diagnosis to NIV initiation in a cohort of 265 ALS patients (half with bulbar onset disease) was 15±18 months (Reginault et al., 2023). Thus, the fact that initiation of NIV for the EAP patients is orders of magnitude greater than that reported in the literature is evidence that NurOwn treatment may be having a positive impact on patients’ respiratory function.
Multiple Pathways to Approval
The petitioners believe there are multiple pathways by which NurOwn could be approved if the agency decided to take a new look at the “totality of the evidence”, particularly the new data from the EAP that was not evaluated as part of the Advisory Committee meeting in September 2023.
- First, the new survival data likely meets the agency’s “substantial evidence” threshold. While there were only 10 participants in the EAP, the quality of the data is such that the petitioners believe it overrides the fact that the population was small.
- Second, if it doesn’t meet the “substantial evidence” threshold, the petitioners believe the Phase 3 data along with the EAP data likely meet the “reasonable likelihood” threshold for accelerated approval. The unprecedented “time to tracheostomy” and “time to NIV” are very likely to reasonably predict a clinically meaningful outcome on mortality, due to the fact that respiratory function is the biggest survival predictor in ALS.
- Third, the biomarker data previously disclosed by the company (see here) likely supports the use of the accelerated approval pathway. The biomarker data demonstrate target engagement across various disease pathways, including neuroinflammation, neurodegeneration, and neuroprotection.
- Lastly, new FDA Commissioner Dr. Martin Makary has discussed his intention to initiate a “new pathway” for approving rare disease drugs, with approval based on a “plausible mechanism”. This pathway would be reserved for rare or incurable diseases that affect a small number of individuals (e.g., ALS) and drugs approved under this pathway would include a patient surveillance system. While the specifics of that pathway are still being codified, the petitioners believe NurOwn would fit with the vision for that program and BrainStorm already has everything in place to begin a Phase 4 post-marketing study through its preparation for a Phase 3b trial.
Conclusion
The Citizen’s Petition requesting that the FDA use its “regulatory flexibility” to approve NurOwn is an interesting development for BrainStorm and provides the company with an immense opportunity. Having reviewed the entire document, we believe the petitioners make a compelling argument for why NurOwn should be conditionally approved while a Phase 4 study is conducted. At this point, BrainStorm is still planning to conduct a Phase 3b trial, as per the SPA with the FDA, and according to its latest conference call all that is left to do is obtain the necessary funding before that trial can be initiated. Thus, if conditional approval were granted the company would be ready to initiate a confirmatory Phase 4 trial almost immediately.
How the FDA will respond to this petition is unknowable at this point, however based on our reading of the petition we believe there is a high likelihood that the company will be invited to resubmit the BLA for NurOwn, including the previously undisclosed EAP data. Based on this, we have adjusted our model by increasing the probability of approval for NurOwn, which has increased our valuation to $17 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.