By David Bautz, PhD
NASDAQ:CVKD
READ THE FULL CVKD RESEARCH REPORT
Business Update
Acquires Portfolio of Factor XIa Inhibitors
On September 15, 2025, Cadrenal Therapeutics, Inc. (NASDAQ:CVKD) announced it had acquired a portfolio of Factor XIa inhibitors from eXIthera Pharmaceuticals. Included in the acquired assets is frunexian, a Phase 2-ready asset that is being developed as an acute anticoagulant therapy for prevention of coagulation where medical devices play a significant role such as in cardiopulmonary bypass and catheter thrombosis. The portfolio also includes EP-7327, an oral Factor XIa inhibitor for the prevention and treatment of major thrombotic conditions.
Anticoagulation therapy is utilized for the prevention and treatment of thrombosis without disrupting hemostasis. Current anticoagulants target FXa or thrombin, which are enzymes in the extrinsic pathway of coagulation. However, inhibition of Factor XIa is a new area of focus since epidemiological and animal data support the concept that Factor XIa is essential for thrombosis but not required for hemostasis. Individuals with congenital FXI deficiency do not have an increased risk for spontaneous bleeding, while at the same time these patients are at a lower risk for venous thromboembolism (VTE) and ischemic stroke. Conversely, patients with elevated FXI levels are at a higher risk for VTE and stroke.
Frunexian is a small-molecule inhibitor of Factor XIa and is designed for intravenous administration. While there are a number of other FXIa inhibitors in development (as shown in the following table), frunexian is the only one we are aware of that is being developed in the acute care setting.
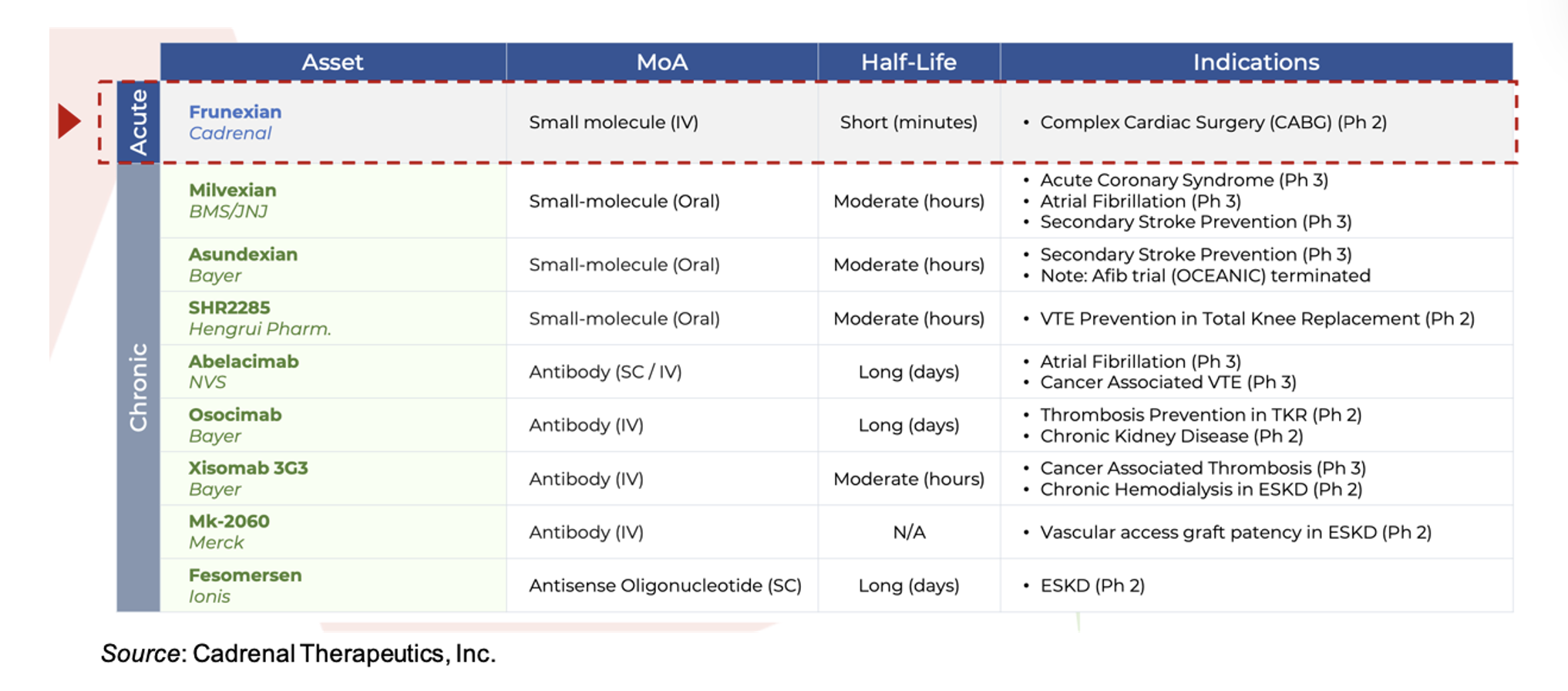
Frunexian has been successfully tested in two Phase 1 clinical trials in healthy volunteers. The results showed that the drug was well tolerated at doses up to 2.25 mg/kg/h, and there were no reports of serious adverse events. Steady-state plasma levels of the drug were rapidly achieved, and importantly, the drug was quickly cleared following cessation of dosing, an important factor for use in the acute care setting. The company has additional work to perform regarding the manufacturing of frunexian before initiating the clinical trial plan, which may initially focus on complex cardiac surgery.
Clinical Trial Planned in ESKD w/ Atrial Fibrillation Transitioning to Dialysis
In August 2025, Cadrenal announced plans for a clinical trial for tecarfarin in patients with end-stage kidney disease (ESKD) that are transitioning to dialysis. We anticipate enrollment initiating in the first quarter of 2026 and topline data could be available approximately one year following the start of the trial.
Patients with chronic kidney disease (CKD) have a high risk for cardiovascular events: approximately half of CKD stage 4 and 5 (ESKD) have cardiovascular disease (CVD) (Stevens et al., 2007), and cardiovascular mortality is one of the leading causes of death in patients with advanced CKD (Webster et al., 2016). There is conflicting data available as to whether current anticoagulation therapy options (both warfarin and DOACs) are effective at preventing CV events in these patients. The RENAL-AF trial compared apixaban to warfarin in dialysis patients with atrial fibrillation (AF), however the study had inadequate power to draw any conclusion regarding rates of major or clinically relevant nonmajor bleeding, although it was noted that bleeding events were much more common than stroke events in that population (Pokorney et al., 2022). Pooled observation data suggest that DOACs may have similar stroke prevention and potentially lower major bleeding vs. warfarin, however the evidence is low-quality and heterogeneous (Shen et al., 2023). The bottom line is that a new treatment option is necessary for dialysis patients with ESKD. Tecarfarin activity is not affected in ESKD patients, thus making it a suitable compound to test as an anticoagulant therapy in that population.
Financial Update
On August 11, 2025, Cadrenal announced financial results for the second quarter of 2025. As expected, the company did not record any revenues for the three months ending June 30, 2025. R&D expenses in the second quarter of 2025 were $1.1 million compared to $1.3 million in the second quarter of 2024. The decrease was primarily due to decreased consulting expenses partially offset by an increase in CMC costs. G&A expenses were $2.7 million in the second quarter of 2025 compared to $1.2 million in the second quarter of 2024. The increase was primarily due to increased public company expenses and non-cash stock-based compensation
As of June 30, 2025, Cadrenal had approximately $5.6 million in cash and cash equivalents. While the company has sufficient capital to fund operations for the next 12 months, it will need to raise additional capital to conduct the planned clinical trials for tecarfarin and frunexian. The company currently has approximately 2.0 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 3.1 million.
Conclusion
Cadrenal has now enhanced its pipeline with the acquisition of frunexian and the other Factor XIa inhibitors and we look forward to additional information regarding their development. The shift to focusing on ESKD patients for tecarfarin is important as there is a significant need for effective anticoagulant therapy for those patients and we believe positive results could also serve to de-risk the development of tecarfarin in other indications such as in LVAD patients. Before incorporating frunexian into our model we will wait and see what development path the company decides to pursue with it, thus our valuation remains at $30 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.