By David Bautz, PhD
NASDAQ:FBLG
READ THE FULL FBLG RESEARCH REPORT
Business Update
Phase 1/2 Trial in Diabetic Foot Ulcers to Initiate in 1Q26
FibroBiologics, Inc. (NASDAQ:FBLG) is continuing preparations for a Phase 1/2 trial of CYWC628, a fibroblast-based spheroid product candidate, in patients suffering from diabetic foot ulcers (DFUs), which we anticipate initiating in the first quarter of 2026 and completing in the third quarter of 2026. Recent activities the company has accomplished to support the upcoming clinical trial include:
- In October 2024, FibroBiologics and Charles River Laboratories announced a master services agreement to develop and manufacture the company’s therapeutic master cell bank, working cell bank, and fibroblast-based spheroids product CYWC628. In addition, the company announced the technology transfer for cGMP manufacturing was successfully completed along with feasibility studies evaluating the cell manufacturing processes. Based on the positive results of the feasibility studies, Charles River became the contract development and manufacturing organization (CDMO) to produce drug products for the upcoming Phase 1/2 for CYWC628.
- In February 2025, FibroBiologics and Charles River announced the completion of FibroBiologics’ proprietary master cell bank, which will support the upcoming clinical trial of CYWC628. In addition, the cell bank, which was manufactured in accordance with cGMP, successfully passed all required safety testing.
- In September 2024, FibroBiologics announced it had engaged Southern Star Research to provide clinical research organization (CRO) services for the company in Australia, including preparation for the planned Phase 1/2 clinical trial of CYWC628 for the treatment of DFUs.
CYWC628 is being developed for the treatment of DFUs, which cause significant morbidity for the 6.3% of diabetic adults (~33 million) that develop them. Of those, 20% will require lower extremity amputation and 10% will die within the first year of their first DFU. In addition, once a DFU forms there is a high rate of recurrence, both at one year (40%) and three years (70%).
Fibroblasts have excellent therapeutic potential in the treatment of DFUs due to the critical role they play in every stage of wound healing, including hemostasis, inflammation, proliferation, and remodeling. Importantly, fibroblasts are the key cells that secrete extracellular matrix proteins that maintain all the tissues and organs in the body.

The company does not utilize single cell fibroblasts for treatment but instead a fibroblast spheroid, which is composed of approximately 3,000 fibroblasts and is administered to the top of the wound at which time the cells migrate from the surface of the wound and release various cytokines and growth factors to initiate the wound healing process. The use of spheroids is more practical from a therapeutic perspective as they have higher viability than single cells, they don’t require pre-culturing before administration, they can be easily frozen and thawed, and they have a significantly higher potency and efficacy compared to single cells.
FibroBiologics has compiled a robust pre-clinical data set showing the efficacy of fibroblast spheroids in the treatment of wounds. For example, the following figure shows results using a diabetic mouse model in which administration of fibroblasts led to a statistically significant average 83.8% wound closure by Day 19 compared to 66.0% for Grafix™ and only 51.2% for control.
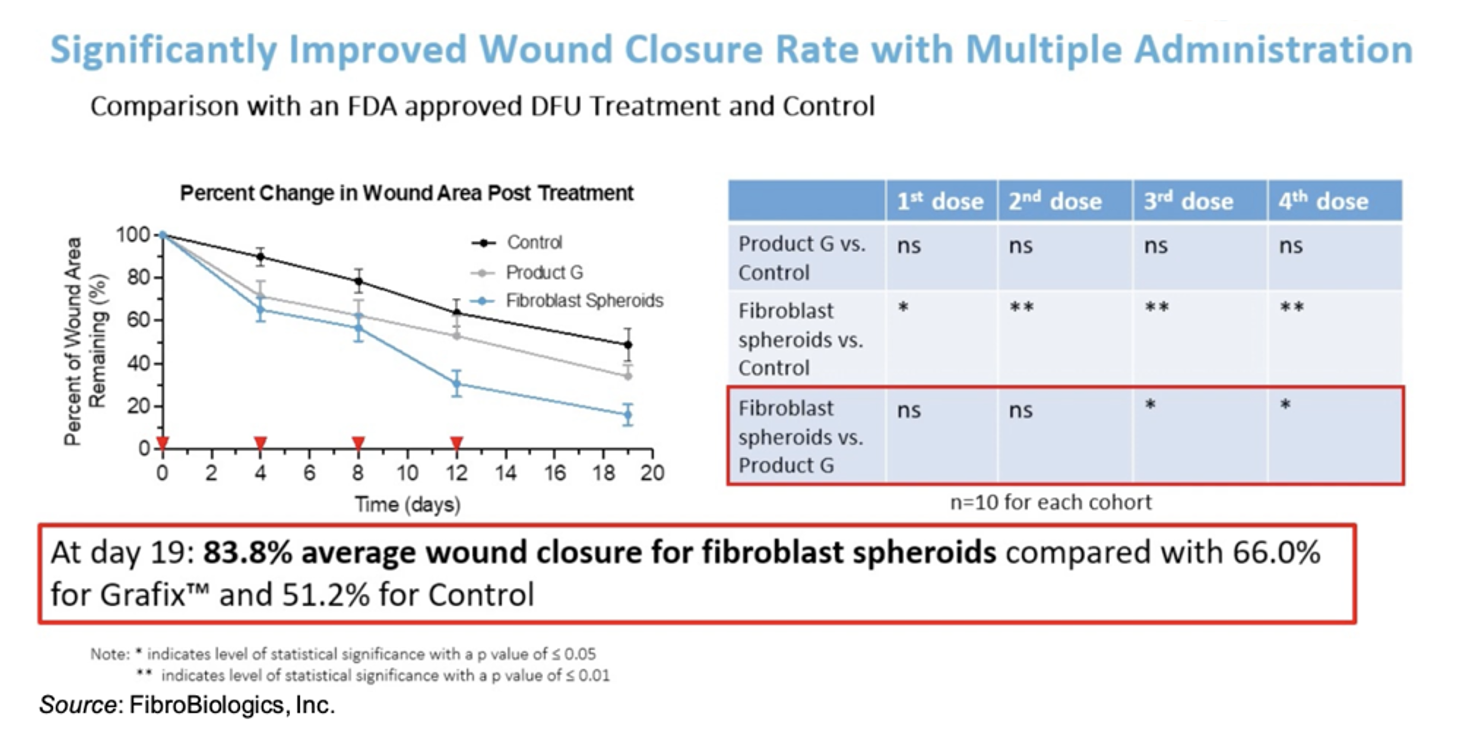
While wound healing is important, the quality of the wound healing is just as important. FibroBiologics has data on seven key biomarkers that are key to demonstrating the quality of the wound healing. Fibroblast treatment shows much better re-epithelialization, granulation, cell proliferation, neo-vascularization, recruitment and proliferation of fibroblasts, keratinocyte migration, and epithelial-mesenchymal transition. Intriguingly, even though the fibroblast spheroids are administered topically, there appears to be a systemic effect on cytokine levels, including IL-6, TNF-α, IL-1β, and IL-10.
The use of fibroblast spheroids for wound healing can be thought of as a platform technology. In addition to the treatment of DFUs, fibroblast spheroids could also be used for the treatment of burns and surgical wounds.
In regards to safety, the company has performed a number of experiments to examine any potential adverse events associated with fibroblast-based therapy. The cells do not graft into tissue. Following application, the cells stay on the surface of the wound and initiate the healing process before gradually dying off within four days of treatment. In addition, there is no impact on CBC, WBC, liver function, or kidney function, thus showing that administration of fibroblasts appears to be quite safe.
Confirms Ability to Manufacture CybroCell™ for Degenerative Disc Disease and Cartilage Repair Programs
On July 9, 2026, FibroBiologics announced an advancement in its cartilage repair program. The company successfully differentiated CYWC628 spheroids (which are used in its wound healing program) into chondrocytes, which are the specialized cells responsible for cartilage formation. This means that the company can now utilize the current CYWC628 master cell bank for manufacturing CybroCell, the investigational cell therapy that is being developed for the treatment of degenerative disc disease (DDD). The ability to use the CYWC628 master cell bank will help reduce development and manufacturing time and costs.
Files Patent for Methods of Generating Multipotent Cells from Donor Tissue
On July 28, 2025, FibroBiologics announced the filing of a patent application with the United State Patent and Trademark Office that covers methods for generating multipotent cells from fibroblasts isolated from donor tissue for clinical applications. A multipotent cell is one that can differentiate into multiple specialized cell types. Fibroblasts are considered multipotent since they can be differentiated into multiple cell types, including chondrocytes, osteocytes, hepatocytes, and cardiomyocytes. The methods covered in the patent give the company the ability to obtain large quantities of stable and scalable multipotent cells from donor-derived fibroblasts.
Financial Update
On July 31, 2025, FibroBiologics announced financial results for the second quarter of 2025. As expected, the company did not report any revenues for the three months ending June 30, 2025. Research and development expenses were approximately $2.0 million in the second quarter of 2025, compared to approximately $1.0 million in the second quarter of 2024. The increase was primarily due to increased CRO costs and personnel related expenses. General and administrative expenses were approximately $2.4 million in the second quarter of 2025, compared to approximately $2.2 million in the second quarter of 2024. The increase was primarily due to additional personnel and professional fees.
The company exited the first quarter of 2025 with approximately $8.9 million in cash and cash equivalents. During the second quarter of 2025, the company closed on the third $5 million tranche of the previously announced Standby Equity Purchase Agreement with YA II PN, Ltd., an investment fund managed by Yorkville Advisors Global, LP. FibroBiologics can sell an additional $10 million in common stock to Yorkville while the promissory notes remain outstanding. As of July 30, 2025, FibroBiologics had approximately 41.9 million common shares outstanding and, when factoring in stock options, a fully diluted share count of approximately 47.8 million.
Conclusion
FibroBiologics has strengthened its balance sheet with the closing of the third $5M tranche from the $25M financing facility, which will provide the company with the capital necessary to conduct its Phase 1/2 clinical trial in DFUs. The trial is currently slated to begin in the first quarter of 2026 and conclude in the third quarter of 2026. In addition, the company will be concluding the pre-IND studies of CPYS317 for the management of psoriasis before the end of 2025. With no changes to our model our valuation remains at $11 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.