By John Vandermosten, CFA
NASDAQ:LEXX
READ THE FULL LEXX RESEARCH REPORT
As we step into fall, Lexaria Bioscience Corporation (NASDAQ:LEXX) continues to work on multiple GLP-1 trials. Most notably, it reported the last patient-last visit for the Phase Ib study designated GLP-1-H24-4 and reported data from its rodent biodistribution study. It also closed a $4.0 million capital raise earlier this week. Activity in the obesity and GLP-1 agonist space has been robust in 2025 with a number of deals and new trial data announced. In this report we will bring investors up to date on recent activity and update near term milestones.
Lexaria View on GLP-1 Agonists
In the fall of 2023, Lexaria began its efforts to evaluate members of the GLP-1 agonist class formulated with DehydraTECH to determine if the new formulation could improve bioavailability, cost-effectiveness, tolerability, weight-loss potential, management of diabetes and other health conditions. Since then, the company has launched five human studies along with other non-human work to answer this question. Over the last two years, Lexaria has reported the data from these trials showing reduced levels of side effects, comparable levels of glucose reduction and insulin increase, less semaglutide necessary than in Rybelsus to achieve similar effects. In an August 6th press release, management penned a missive reviewing GLP-1 agonist market activity highlighting the primary players (Eli Lilly and Novo Nordisk), the number of drugs in development (39) and number of sponsors advancing them (34). The note from management also addressed the dealmaking environment around the class, underlining the collaboration between Novo Nordisk and Septerna, Roche’s asset acquisition from Zealand Pharma, Regeneron’s licensing deal with Hansoh Pharmaceuticals and Merck and Cyprumed, which we discussed in our previous report.
In late August, Eli Lilly announced data from its Phase III trial evaluating its oral GLP-1 receptor agonist, orforglipron. The study met all of its primary and secondary endpoints. With the completion of the study, Lilly has all the clinical data necessary to submit orforglipron for approval globally. Availability of an oral for Eli Lilly could raise the stakes for having an oral version of this class of drug and benefit Lexaria as others find other oral solutions for GLP-1 agonist delivery.
Lexaria’s data comparing Rybelsus, tirzepatide, semaglutide and liraglutide with DHT-formulated versions of these compounds now spans five human studies. With this trove of supportive information, the company seeks the interest of a major player in the industry who recognizes the value of an oral version of these injectable drugs.
GLP-1 Agonist Rodent Biodistribution Study
In November 2024, Lexaria announced that it had signed a contract for a GLP-1 agonist biodistribution study with an unidentified contract research organization. The research fluorescently tagged DHT formulated semaglutide and tracked its biodistribution in rodents. Understanding the distribution of the drug in subject tissue will help researchers understand the binding properties and receptors that are targeted by the drug. It can also help understand in which areas or tissues the drug concentrates and potentially lead to unwanted side effects. The work will also help researchers understand how orally delivered DHT-semaglutide differs from the infused formulation of the drug. Last month, Lexaria announced results from this study in a press release.
Study Design
Details were tracked via fluorescent imaging detection to show how and where the semaglutide distributes and localizes following oral ingestion by Sprague-Dawley rats. After the initial evaluation, the animals were euthanized and various key tissues were examined including those from the brain, pancreas, lung, kidney, liver and heart for more detailed fluorescent imaging detection showing very specific tissue localization patterns and concentrations. The analysis includes DHT and non-DHT formulated Rybelsus orally administered product.
Efforts also included measurement of certain GLP-1 receptor specific antibodies detectable through an immunofluorescence methodology. This was done to allow the analytical laboratory to confirm the extent of GLP-1 receptor binding of the two formulations in the tissue samples taken from the animals, providing a detailed measure of fluorescence distribution and localization patterns. The fluorescent tagging of the two variations of semaglutide (Rybelsus and the DHT formulation) will show the biodistribution differences between the two.
Study Results
A September 19th press release shared results from the study. It noted that DHT-fluorescently tagged semaglutide (FTS) demonstrated a predominantly higher apparent trend in brain biodistribution than the Rybelsus equivalent composition and all study controls. The determination was made based on fluorescent signal intensity using whole brain imaging. Below, the DHT performance can be seen in the middle light blue, aqua and turquoise bars in the center of the exhibit.
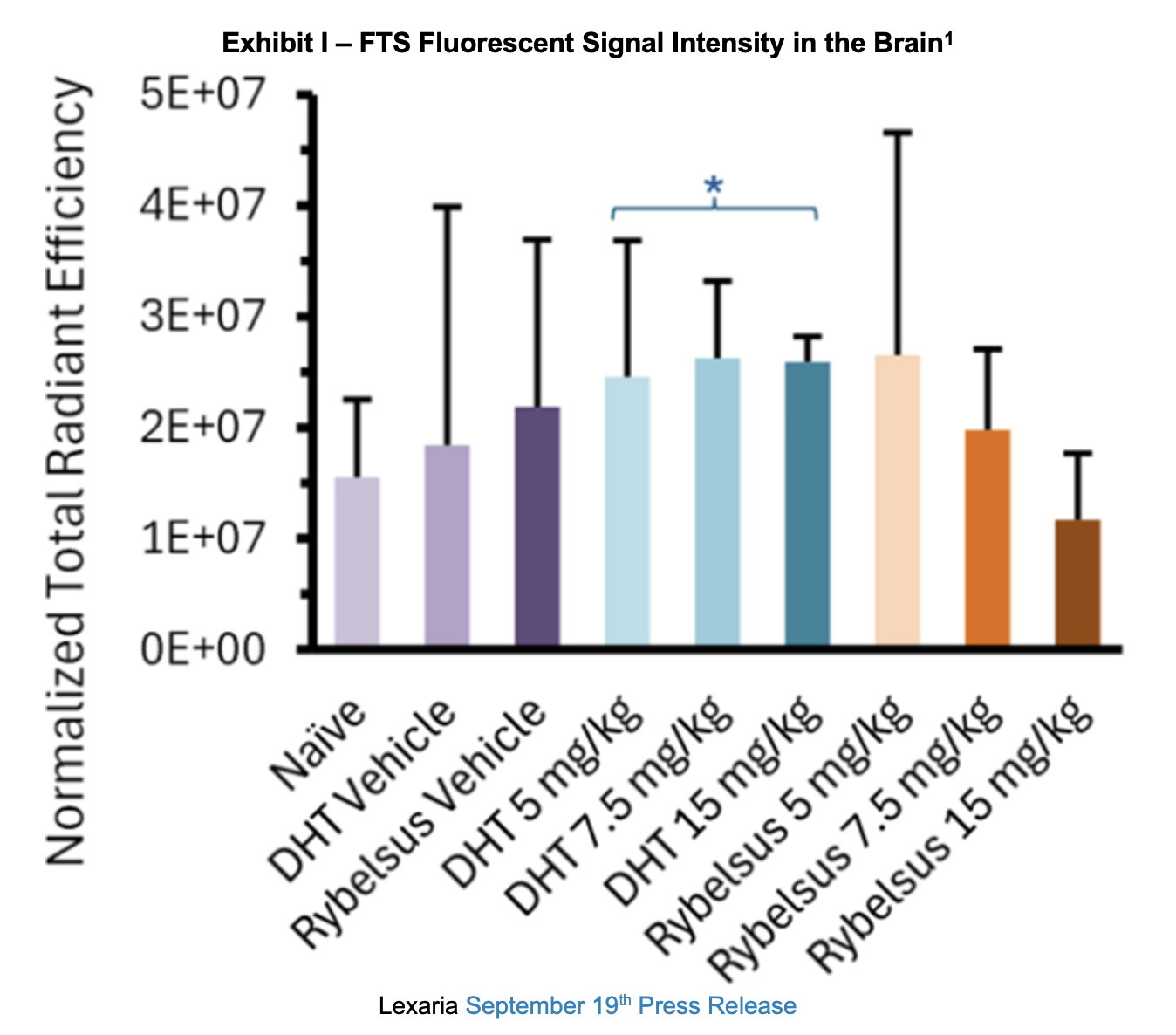
Following the whole-organ imaging of the rat model, the brain was further sectioned via sagittal slices (2-3 mm thickness) into two and then four pieces to better visualize the brain regions in which semaglutide is binding and stimulating a response. These regions of interest include the brainstem, known for direct semaglutide interaction; the paraventricular nucleus of the hypothalamus, involved in energy homeostasis; and the circumventricular organs, which lack a blood-brain barrier, such as the area postrema, subfornical organ and median eminence. Upon measurement, investigators noted that all three DHT doses tested displayed fluorescence above that of the naïve and vehicle groups, while only the highest dosage (15mg/kg) of the Rybelsus equivalent composition surpassed the naïve and vehicle groups.
Lexaria believes that findings from the study suggest that the DHT-FTS composition, absent all of the Rybelsus composition excipients, may enable unique delivery and distribution enhancements in brain tissue possibly supporting improved pharmacodynamic performance. Complementary biodistribution benefits may be derived by using a similar DehydraTECH semaglutide composition combined with the Rybelsus excipients. Future testing to show this would be required to measure this and potential safety and efficacy improvements.
This type of study helps researchers further understand the pharmacokinetics and mechanism of action of DHT-formulated semaglutide and will be part of the conversations with large pharma partners.
GLP-1-H24-4 Interim Results (Fourth Study)
At the end of July, Lexaria provided an interim look into its GLP-1-H24-4 study highlighting the reduction in side effects for the DHT formulated GLP-1s vs. approved versions of the drug. At eight weeks, the DHT-semaglutide arm saw a 36.5% reduction in overall side effects and 43.5% lower gastrointestinal (GI) side effects compared to results in the Rybelsus arm. Our report reviewing 3Q:25 results provided a summary of adverse events from the trial.
Lexaria’s press release documented at least one adverse event (AE) for each of the 25 subjects in the Rybelsus arm. Five subjects in the DHT arm (5/24) experienced no AEs (referenced as a 20.8% reduction in Lexaria’s press release). A study cited by Lexaria (Bergmann, et al. 2022) found just under 90% of semaglutide patients in the study experienced an AE. The press release compares this to DHT-semaglutide’s AE rate of 79.2%. However, the comparison must be placed in the context of the Lexaria data at the 8-week mark and including 24 people compared to the greater than 1,000 subjects assessed for injected semaglutide. Lexaria reviewed several tirzepatide studies and found a similar incidence of AEs as they did for semaglutide in a meta-analysis (Mishra et al. 2023). The study noted a positive correlation between dose level and incidence of AEs. Another remarkable takeaway from the meta-analysis is the high rate of GI-related AEs which comprised up to 50% of the total AEs for injectable tirzepatide. Lexaria compared this hurdle to the 22% rate achieved with DHT-semaglutide in the 8-week study.
GLP-1-H24-4 was conducted with 24-25 overweight, obese, pre- or type 2 diabetic patients in each of the five study arms (n=126), of which 4 arms evaluated various DehydraTECH formulations with the 5th being the Study control arm. Arm 1 evaluated DHT-CBD, Arm 2, DHT-semaglutide and Arm 3, DHT-semaglutide and DHT-CBD. Arm 4 evaluated Rybelsus tablets and Arm 5 examined DHT-tirzepatide.
Changes in glycated hemoglobin (HbA1c) and weight were other measured endpoints in the GLP-1-H24-4 study. Lexaria extracted these same metrics from Novo Nordisk’s Pioneer studies[2],[3]. DHT-semaglutide was able to reduce HbA1c and weight over the eight weeks, but at a lesser magnitude than what was achieved by Rybelsus. While DHT-semaglutide achieved lower levels of weight loss and HbA1c reduction compared to Rybelsus, it also is associated with reduced side effects, especially those that are GI-related. We think that 8-week data is insufficient for making significant comparisons. The primary takeaways are that the trend in endpoints is moving in the right direction and that reduced adverse events will allow for a greater number of patients to continue on a therapy so they can obtain its benefit. Lexaria also brought attention to the focus on adverse events with a quote[4] from Martin Hoist Lange, who was promoted to Chief Scientific Officer of Novo Nordisk earlier this week: “We want to win the weight loss [battle] but we also want to have a gastrointestinal adverse event profile that is attractive and competitive.”
On August 14th, 2025 Lexaria announced that the last patient-last visit had been completed. Study work accelerates with full sample and data analyses underway with the goal of reporting data prior to the end of 2025. The company’s CRO is managing the laboratory analysis phase of the work and management is blinded until the work has been completed.

$4.0 Million Offering
On September 26th, Lexaria announced a $4.0 million registered direct offering priced at the market. 2,666,667 shares of Lexaria common stock were offered at $1.50 per share along with the same number of warrants with a $1.37 exercise price. Gross proceeds are estimated to be approximately $4.0 million and we forecast net proceeds to be about $3.6 million after deducting the placement agent fees and other offering expenses. The offering closed on September 29th.
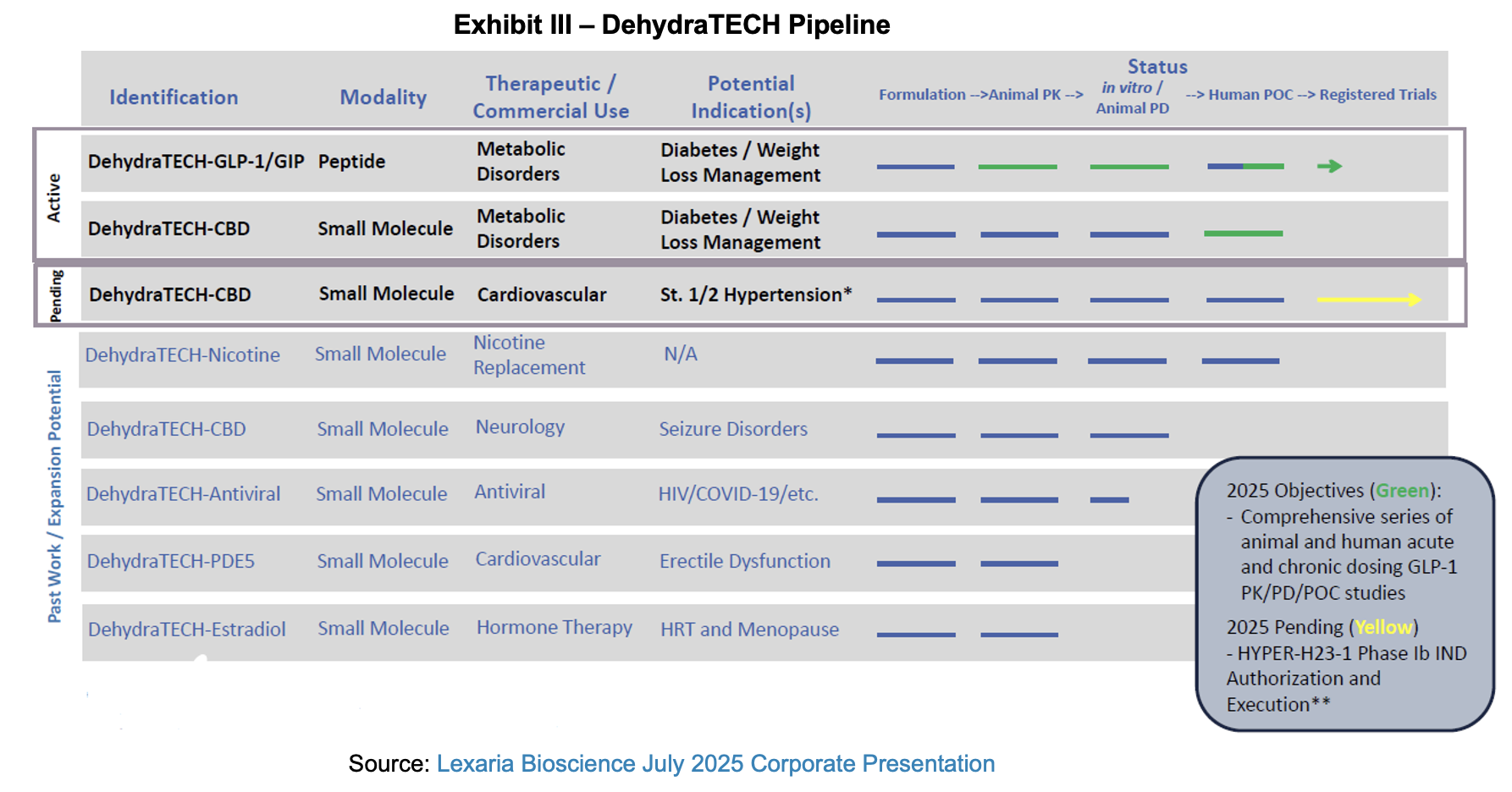
Pipeline
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] Total radiant efficiency (TRE) is a measure of the summation of flux (i.e., measured fluorescence) within a given tissue region of interest (ROI). For the brain analyses in this study, an ROI was drawn around the excised brain tissue samples to quantify the total fluorescence within that region in each case. To account for the variability in ROI size, the TRE was divided by the area of the ROI to "normalize" the value. In short, Normalized TRE = TRE/ROI area.
[2] Aroda, V.R. et al. A new era for oral peptides: SNAC and the development of oral semaglutide for the treatment of type 2 diabetes. Reviews in Endocrine and Metabolic Disorders. October 2022.
[3] Husain, M. et al. Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine. June 2019.
[4] Novo Nordisk R&D Investor Event, June 22, 2025