By John Vandermosten, CFA
NASDAQ:LEXX
READ THE FULL LEXX RESEARCH REPORT
Lexaria Bioscience Corporation (NASDAQ:LEXX) announced results from its Phase Ib study evaluating multiple GLP-1 agonists in combination with DehydraTECH (DHT). The study met its primary endpoint with DHT-semaglutide reducing overall side effects by 48% and gastrointestinal (GI) side effects by 55% as compared to Rybelsus. The company also reported its fiscal year 2025 results via the filing of Lexaria’s Form 10-K. Since our previous update in early October, Lexaria has provided a strategic update, extended its active material transfer agreement (MTA) and closed a $3.5 million registered direct offering. With a set of clinical trial data in hand, Lexaria is seeking new business development activities employing an advisory firm to identify and pursue leads.
2025 Results
Lexaria reported annual results for the twelve-month period ending August 31st, 2025 through the filing of its Form 10-K. The company reported revenues of $706,000 and total operating expense of $11.9 million resulting in net loss of ($11.9) million or ($0.66) per diluted common share.
For its fiscal year and versus the comparable prior year period:
- Revenue totaled $706,000, up 52% from $464,000 due to increases in licensing revenues related to the agreement with Premier. The arrangement with Premier expired on August 31st, 2025;
- Research and development expenses totaled $8.2 million, up 249% from $2.4 million as a result of spending on the commencement and completion of the Phase Ib, 12-week chronic study investigating DHT with several GLP-1 agonists and cannabidiol;
- General and administrative expenses totaled $4.3 million up 13% from $3.9 million on account of higher wages and salaries, reduced foreign exchange and patent-related impairment losses, and greater insurance premiums. These increases were partially offset by lower consulting fees, advertising and promotion expenses, legal and professional fees, and investor relations expense;
- Other loss of ($31,000) represented unrealized loss on marketable securities related to decreases in fair value and a small contribution from interest income;
- Net loss was ($11.9) million, or ($0.66) per share, compared to net loss of ($5.8) million or ($0.47) per share.
As of August 31st, 2025, cash and marketable securities totaled $1.9 million which compares to $6.6 million at the end of fiscal year 2024. Cash burn for FY:25 was approximately ($10.5) million. Cash from financing over the same period totaled $6.0 million from equity sales. Following the end of the quarter, Lexaria executed two registered direct offerings that together raised $7.5 million gross proceeds. The capital raises provide sufficient funding to support operations during calendar year 2026.
GLP-1-H24-4 (Phase Ib) Results
GLP-1-H24-4 Trial Design
GLP-1-H24-4 was conducted with 24-25 overweight, obese, pre- or type 2 diabetic patients in each of the five study arms (n=126), of which 4 arms evaluated various DehydraTECH formulations with the 5th being the Study control arm. Arm breakdowns follow:
- Arm 1 – DHT-CBD
- Arm 2 – DHT semaglutide
- Arm 3 – DHT semaglutide + DHT-CBD
- Arm 4 – Rybelsus tablets
- Arm 5 – DHT tirzepatide
Changes in glycated hemoglobin (HbA1c) and weight were other measured endpoints in the GLP-1-H24-4 study. Lexaria extracted these same metrics from Novo Nordisk’s Pioneer studies[1],[2] as endpoints. DHT-semaglutide was able to reduce HbA1c and weight over the eight weeks as reported in the interim readout at the end of July, but at a lesser magnitude than what was achieved by Rybelsus. The primary takeaways from the July interim look are that the trend in endpoints is moving in the right direction and that reduced adverse events will allow for a greater number of patients to continue on a therapy so they can obtain its benefit. Lexaria also brought attention to the focus on adverse events with a quote[3] from Martin Holst Lange, Novo Nordisk’s Chief Scientific Officer: “We want to win the weight loss [battle] but we also want to have a gastrointestinal adverse event profile that is attractive and competitive.”
On August 14th, 2025 Lexaria announced that the last patient-last visit had been completed. Study work accelerates with full sample and data analyses underway with the goal of reporting data prior to the end of 2025. The company’s CRO is managing the laboratory analysis phase of the work and management is blinded until the work has been completed.
December 2025 Final Primary and Secondary Endpoint Results
Lexaria reported results from its fourth human diabetes weight loss study designated GLP-1-H24-4 study on December 23rd, 2025. The primary endpoint of the study was the assessment of DHT-formulation impacts upon safety and tolerability based on the incidence of treatment emergent adverse events. On this metric the DHT-semaglutide arm generated fewer adverse events (AEs), fewer GI AEs and reduced nausea, vomiting and diarrhea compared with the Rybelsus control arm. The study generated a 47.9% reduction in the total quantity of AEs observed in the DHT-semaglutide arm vs. the Rybelsus arm. The study also showed a statistically significant (p-value <0.05), 54.9% reduction in GI-related AEs from DHT-semaglutide vs. Rybelsus.
Exhibit I – Summary of Adverse Events for Study GLP-1-H24-4
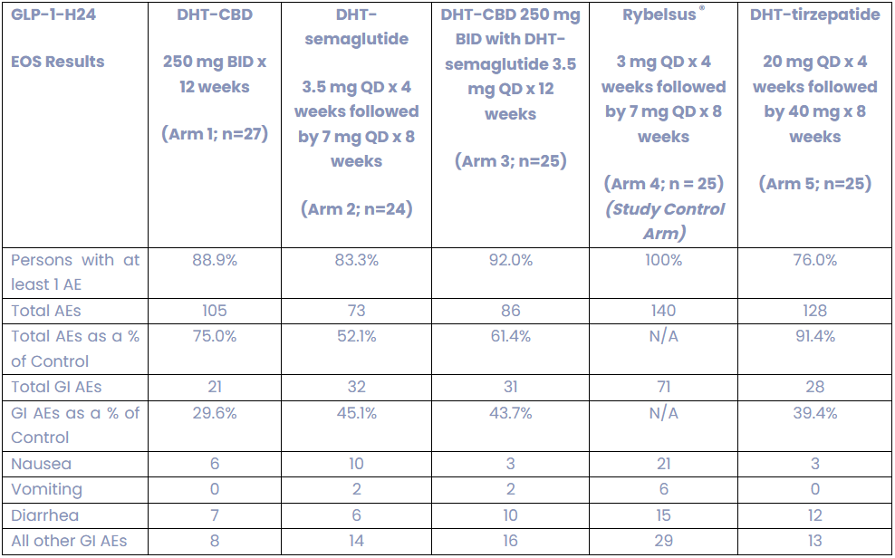
Source: Lexaria December 23rd, 2025 Press Release
The study also evaluated HbA1c levels and body weight as secondary efficacy endpoints. On this metric, the DHT-semaglutide arm only showed modest reductions relative to the Rybelsus arms. Other arms in the study including the cannabidiol arms and DHT-tirzepatide moved in the wrong direction for HbA1c or were modest compared with the Rybelsus control arm. We note that this was a small study not designed for statistically significant efficacy results. Novo Nordisk’s PIONEER I and II study, Rybelsus produced weight loss of 3.8 to 4.4 kg after 26 weeks using 14 mg of the drug.[4][5] This compares to the 5.0 to 5.3 kg loss observed in the Rybelsus arm in Lexaria’s Phase Ib study using 3 mg and later 7 mg of Rybelsus, suggesting that there may be features of this population that make it different from the enrolled population in the PIONEER studies and the impact of small cohort sizes. Lexaria expects to release the data for other secondary endpoints in the future.
Exhibit II – Summary of Adverse Events for Study GLP-1-H24-4
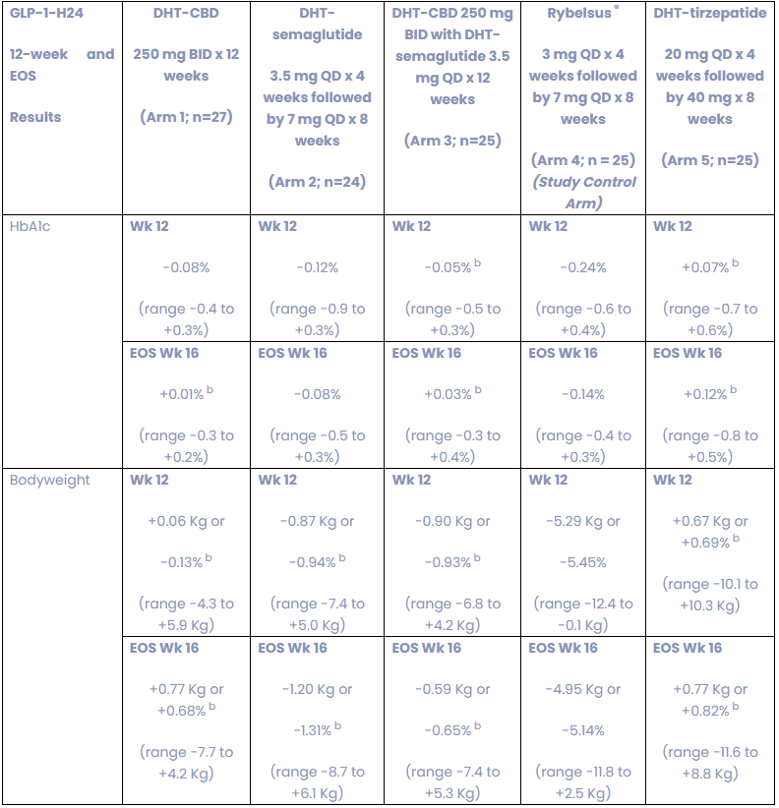
Source: Lexaria December 23rd, 2025 Press Release
Now that the Phase Ib data is available, Lexaria will share the information with its undisclosed MTA partner which has conducted other work on the DHT formulation. Lexaria recently announced that it had extended the MTA agreement so that the partner may review the full dataset from the study.
Initial conclusions from the data presented by Lexaria for the Phase Ib study are that DHT-semaglutide shows a better adverse event profile compared to that of Rybelsus. Looking back to previous results where DHT was used with Rybelsus and its sodium caprylate (SNAC) technology rather than pure semaglutide, results from study #2 showed that the DHT-Rybelsus combination produced 18.8% better absorption than Rybelsus. These results suggest that future studies may evaluate DHT-Rybelsus instead of DHT-semaglutide.
GLP-1-H24-4 Interim Readout
Our report reviewing 3Q:25 results provided a summary of adverse events from the interim readout of GLP-1-H24-4. Lexaria’s press release documented at least one adverse event (AE) for each of the 25 subjects in the Rybelsus arm. Five subjects in the DHT arm (5/24) experienced no AEs (referenced as a 20.8% reduction in Lexaria’s press release). A study cited by Lexaria (Bergmann, et al. 2022) found just under 90% of semaglutide patients in the study experienced an AE. The press release compares this to DHT-semaglutide’s AE rate of 79.2%. However, the comparison must be placed in the context of the Lexaria data at the 8-week mark and including 24 people compared to the greater than 1,000 subjects assessed for injected semaglutide. Lexaria reviewed several tirzepatide studies and found a similar incidence of AEs as they did for semaglutide in a meta-analysis (Mishra et al. 2023). The study noted a positive correlation between dose level and incidence of AEs. Another remarkable takeaway from the meta-analysis is the high rate of GI-related AEs which comprised up to 50% of the total AEs for injectable tirzepatide. Lexaria compared this hurdle to the 22% rate achieved with DHT-semaglutide in the 8-week study.
Exhibit III – Lexaria’s GLP-1 Agonist Human Studies
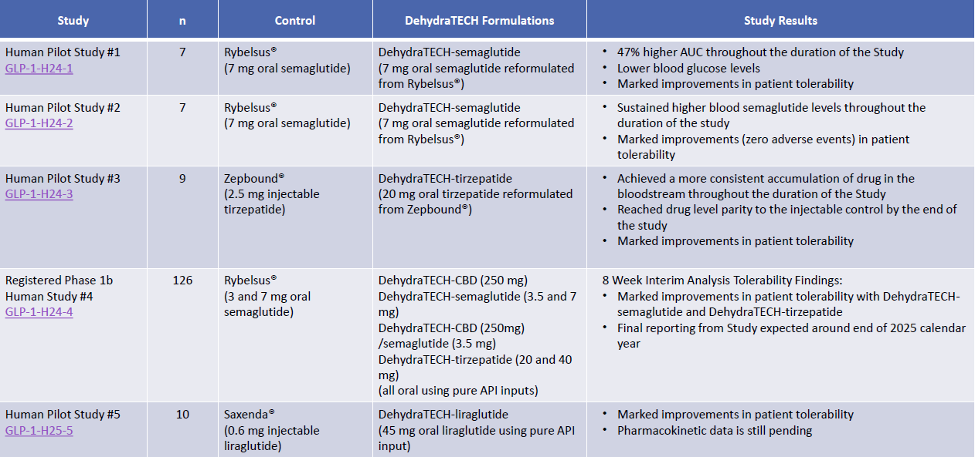
Source: Lexaria Bioscience September 2025 Corporate Presentation
Capital Raises
September $4.0 Million Raise
On September 26th, Lexaria announced a $4.0 million registered direct offering priced at the market. 2,666,667 shares of Lexaria common stock were offered at $1.50 per share along with the same number of warrants with a $1.37 exercise price. Gross proceeds are estimated to be approximately $4.0 million and we forecast net proceeds to be about $3.63 million after deducting the placement agent fees and other offering expenses. The offering closed on September 29th.
December $3.5 Million Raise
On December 15th Lexaria issued a press release announcing pricing of a registered direct offering to raise $3.5 million. The agreement calls for the sale of 2,661,600 shares of common stock at $1.315 per share. A warrant is attached to each share with a five-year life and an exercise price of $1.19 per share. The direct offering closed the following day. We expect net proceeds to be about $3.2 million. H.C. Wainwright acted as the exclusive placement agent for the offering.
Summary
Lexaria closes out 2025 with the report of results from its largest study to date, the 5-arm Phase Ib study of DHT-formulated GLP-1 agonists seeking to evaluate relative safety vs Rybelsus. The study met its primary endpoint of reduced side effects vs the Rybelsus control arm; however, the efficacy results were less convincing. The trial was not designed to show efficacy, so while the relative weight loss and reduction in HbA1c are disappointing, they are not conclusive. Lexaria reports its fiscal year 2025 results with R&D expenses up sharply on the funding of the GLP-1 agonist trials that have been active this year. Post fiscal year end, Lexaria raised an additional $7.5 million gross that should fund the company for 2026. Now that Lexaria has additional data to share and is working with an advisory firm, we anticipate further conversations with partners that may lead to collaborations. DehydraTECH offers improved speed of onset, better bioavailability, reduced adverse events and potentially a favorable regulatory pathway via the 505(b)(2) regulatory pathway. The reduced level of adverse events, especially GI tolerability, as shown in all of Lexaria’s human studies is a particularly attractive feature.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] Aroda, V.R. et al. A new era for oral peptides: SNAC and the development of oral semaglutide for the treatment of type 2 diabetes. Reviews in Endocrine and Metabolic Disorders. October 2022.
[2] Husain, M. et al. Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine. June 2019.
[3]Novo Nordisk R&D Investor Event, June 22, 2025
[4] Rodbard, H.W., et al. Efficacy of Oral Semaglutide: Overview of the PIONEER Clinical Trial Program and Implications for Managed Care. American Journal of Managed Care. December 13th, 2020.
[5] The Rybelsus control arm, as indicated in the exhibit, used significantly lower amounts of 3 mg for four weeks followed by 7 mg for eight weeks. Lexaria provides additional summarized data in its press release about the performance of Rybelsus.