By John Vandermosten, CFA
NASDAQ:LNTH
READ THE FULL LNTH RESEARCH REPORT
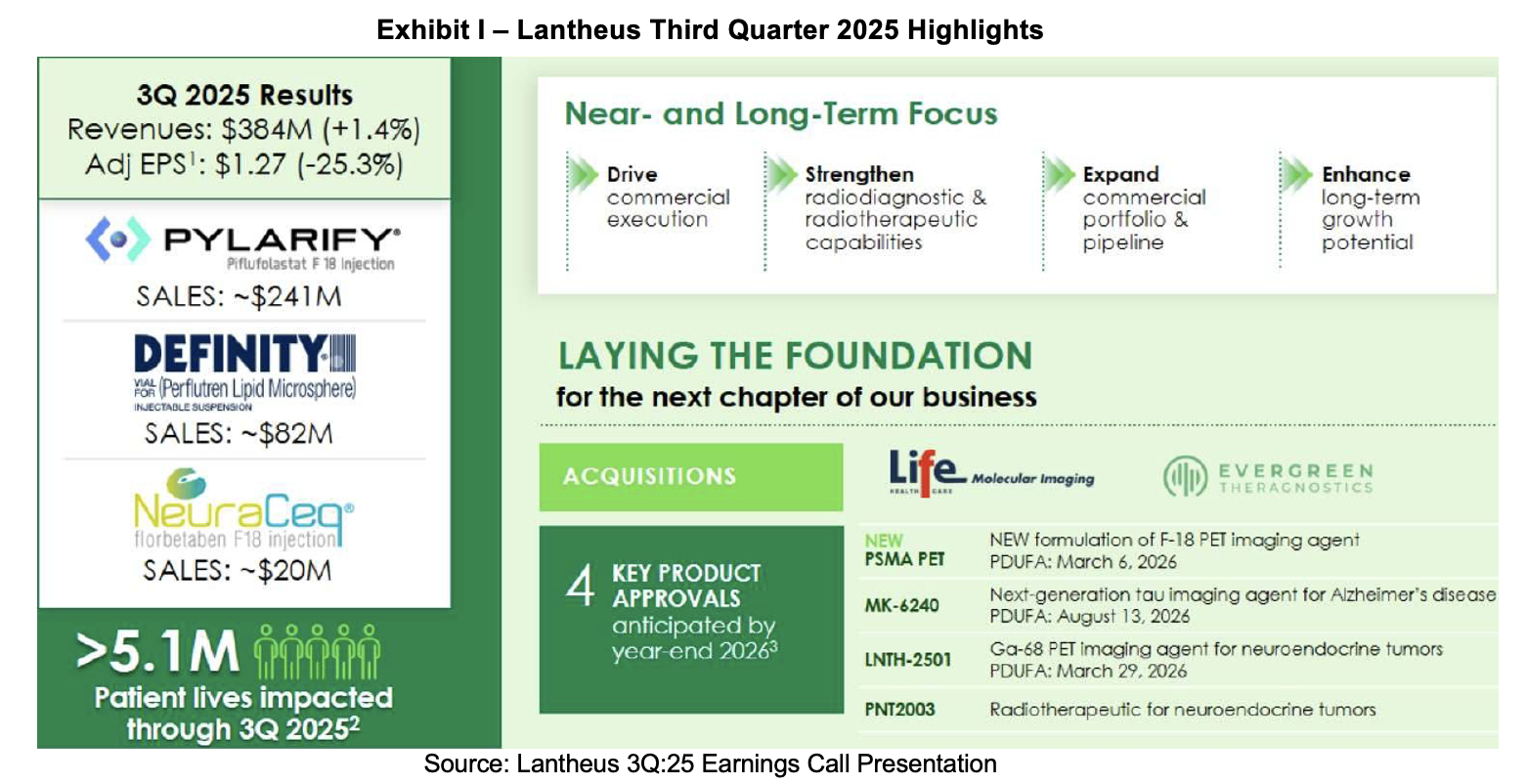
Lantheus Holdings, Inc. (NASDAQ:LNTH) reported third quarter 2025 financial and operational results on November 6th, producing sales of $384 million, a 1% increase compared to 3Q:24 levels. Pylarify and Strategic Partnerships saw a year over year decline which was offset by an increase in revenue from Definity, TechneLite and Other Precision Diagnostics sales. NeuraCeq revenues were recognized for the first time in 3Q:25, adding $20 million in sales. Free cash flow generation was $95 million in 3Q:25; however, the cash balance decreased sequentially due to payments for the Life Molecular acquisition and repurchase of common stock. Other notable events since our previous update include participation at several investment conferences, a deal with GE HealthCare for commercialization of Pylarify in Japan, FDA acceptance of MK-6240 for Tau imaging, grant of target action date for LNTH-2501 and CEO Brian Markison’s retirement announcement.
Along with its 3Q:25 report, the company narrowed its revenue and earnings guidance. Flow through from 340B pricing[1] resets will continue to pressure Pylarify revenues in the fourth quarter despite an anticipated low single digit volume increase. Market share losses in the first half of the year have shown signs of reversing and customers are switching back to Pylarify due to its perceived superior imaging capabilities. To fight the pricing pressures and to provide an improved formulation of Pylarify, Lantheus is preparing to launch its new F-18 PSMA PET formulation. It uses an improved manufacturing process and produces an increased batch size. This is expected to increase production efficiency, supply resilience and enable increased patient access. Assuming an FDA approval, the new agent could be available for sale by 2H:26.
Lantheus’ leading growth franchise is in the neuroimaging space. For the first time, there are revenues from this category that come from the acquisition of NeuraCeq, a β-amyloid targeting imaging agent. New PET Manufacturing Facilities (PMFs) supporting NeuraCeq have been activated bringing the total to 20. Six more are planned for 2026. Another asset in this class, MK-6240, an F-18 labeled Tau-targeting molecule has an assigned target action date in August 2026. The new drug application is supported by two Phase III trials which met sensitivity and specificity endpoints. These assets are augmented by other neurology pipeline candidates including LNTH-2620 and NAV-4694.
Lantheus’ growth strategy for NeuraCeq will expand geographic coverage to ensure broad access across leading Alzheimer’s centers and community practices, improve availability and scheduling flexibility, and leverage revised appropriate use criteria and updated benefit manager guidelines, which recommend repeat scanning. The recent FDA approval of blood-based biomarkers is an important advancement, enabling earlier identification of patients. Lantheus believes that blood-based biomarkers will complement, not replace, PET imaging to visualize and quantify disease.
We think the management team has correctly identified neuroimaging as a material growth area and its investment into four assets in this space along with the acquisition of a commercialization team provides the company a head start when it obtains approval for its neuro-imaging assets. Beyond the anticipated launch of several new products over the next 18 months, the company has relationships with other R&D companies providing an early look at innovative radiopharmaceuticals moving to approval. Strong cash flow provides the funds needed to advance Lantheus’ pipeline in a financial environment where other development companies are having trouble raising capital and established companies are facing patent cliffs.
3Q:25 Financial and Operational Results
Lantheus announced third quarter 2025 results in a November 6th press release followed by a conference call before the market open. A slide deck was provided to guide investors through the event. The company subsequently filed Form 10-Q with the SEC. Revenues for the quarter totaled $384 million up 1% vs. prior year levels producing GAAP earnings of $0.41 per share and adjusted earnings of $1.27 per share.
For the quarter ending September 30th, 2025 relative to the same prior year period:
- Net sales were $384 million up 1% from $378 million recorded in the same prior year period. The increase was driven by the addition of NeuraCeq revenues and a rise in Definity and TechneLite revenues. Other Precision Diagnostics were essentially flat. Pylarify fell 7.4% due to an estimated 10.7% price decline offset by a 3.3% increase in volumes. Strategic Partnerships and Other fell by 10.1% with the change attributable to the absence of milestones recognized in 3Q:24;
- Cost of goods sold rose 18% to $162 million and gross margin was impacted by a decrease in Pylarify price partially offset by higher margin contributions from NeuraCeq and contract manufacturing services. A batch failure, and error and omissions charges were other headwinds negatively impacting cost of goods sold. Product gross profit margin fell to 56.3% from 62.4%[2];
- Sales and marketing expenses were $48.8 million, up 12% from $43.7 million on account of the increased costs in connection with sales of NeuraCeq and provision of contract manufacturing services and employee-related costs related to the acquisitions of Life Molecular and Evergreen;
- General and administrative expenses were $81.9 million vs. $40.5 million, increasing 102%. The change was attributable to the acquisitions of Life Molecular and Evergreen;
- Research and development expenses were $48.0 million, up 99% from $24.1 million. The increase was related to additional costs from the Life Molecular and Evergreen acquisitions. R&D was 13% of revenues;
- Interest expense was $5.0 million, slightly increased from prior year levels of $4.9 million and related to the 2.625% convertible senior notes due in 2027 and cost of maintaining access to the 2022 revolving facility;
- Investment in equity securities was a net unrealized gain of $1.2 million related to the carrying value of Perspective and Radiopharm’s common stock;
- Other income was ($2.6) million vs. ($10.0) million due to lower contributions from cash balance interest;
- Income tax of $14.6 million represents a 34.5% tax rate with state income taxes, nondeductible stock compensation and non-deductible acquisition-related costs contributing to the difference between the reported rate and the U.S. statutory rate of 21%. This was partially offset by tax credits;
- GAAP net income was $27.8 million or $0.41 per diluted share. Adjusted net income as presented by Lantheus was $85.7 million or $1.27 per diluted share. The difference between the two is explained in part by the removal of stock and incentive plan compensation, strategic collaboration and license costs, amortization of acquired intangible assets, acquisition, integration and divestiture-related costs partially offset by unrealized gain in investment securities and income tax effect of non-GAAP adjustments.
On September 30th, 2025, Lantheus held $382 million in cash and equivalents compared to $913 million at the end of 2024. Free cash flow for 3Q:25 was $95 million vs. $112 million in the same prior year quarter. Material items contributing to the quarter over quarter decline in the cash balance include a $100 million repurchase of Lantheus stock and a $309 million payment for Life Molecular.[3] Following the results of the third quarter, Lantheus narrowed its 2025 guidance. The lower end of the 2025 revenue range was increased to $1.49 billion from $1.475 billion and the high end remained at $1.51 billion. The earnings per share range remained constant at $5.50 per share while the high end of EPS guidance declined to $5.65 from $5.70. There was no update to previous free cash flow guidance provided in the 4Q:24 slide deck, however our estimate for free cash flow is around $200 million in 2025.
New Pylarify Formulation NDA Submission
In early August, Lantheus announced that the FDA had accepted its new drug application (NDA) submission of a new formulation of its F-18 prostate-specific membrane antigen (PSMA) imaging agent through its affiliate, Aphelion. The agency has set the applications’ target action date on March 6th, 2026. The new formulation increases batch size allowing wider effective distribution. The higher radioactive concentration will allow the product to reach new geographic locations. Based on management comments, we anticipate approval by March 2026, coding coverage in the following months and a transitional pass through (TPT) payment application by June 2026. The company estimates that TPT pricing could be available between October 2026 to January 2027. In parallel, Lantheus will need to modify its PET manufacturing facilities (PMFs) and have them approved to manufacture the new formulation.
GE HealthCare-Pylarify Arrangement in Japan
In September 2025, Lantheus entered into an exclusive licensing agreement with GE HealthCare (NASDAQ:GEHC) granting it rights to develop, manufacture, and commercialize Pylarify in Japan. Details of the arrangement were included in a September 24th press release. Lantheus will transfer regulatory dossiers, manufacturing competencies and technical support for GE HealthCare’s development of the imaging agent in Japan. GE HealthCare’s recent acquisition of Nihon Medi-Physics (NMP) is expected to support the research and development and manufacturing of the product. In return for access to Pylarify, GE HealthCare will pay Lantheus an upfront license fee, development milestones and tiered royalties based on product sales.
Earlier this year, GE HealthCare acquired full ownership of NMP from Sumitomo Chemical (NMP). NMP’s product line is positioned to serve neurology, cardiology, oncology and other diagnostic domains with imaging agents. It is also developing diagnostic and therapeutic radionuclides. NMP has experience developing and manufacturing proprietary and in-licensed radiopharmaceuticals used in single photon emission computed tomography (SPECT) and positron emission tomography (PET) molecular imaging procedures. Products in NMP’s portfolio include Vizamyl Injection (Flutemetamol (18F) Injection), used in the Alzheimer’s pathway; DaTSCAN Injection (Ioflupane (¹²³I) injection) used to evaluate patients with suspected Parkinson’s Disease or dementia with Lewy Bodies; and Myoview (Technetium (99mTc) Tetrofosmin), used in SPECT myocardial perfusion imaging for the evaluation of known or suspected coronary artery disease.
MK-6240 Progress
In late April, Lantheus announced results from its two pivotal studies in MK-6240 for Alzheimer’s disease (AD) and verified that the radiodiagnostic had met both of its primary endpoints in two pivotal studies. MK-6240 is an F-18 PET radiodiagnostic that images tau proteins in the form of neurofibrillary tangles (NFTs). The data from the studies was compiled in a new drug application (NDA) to the FDA, which has been submitted and now accepted by the regulatory agency. The FDA assigned a target action date of August 13th, 2026.
MK-6240 is being evaluated in clinical trials to guide therapies targeting tau tangles.. This asset belongs to Lantheus’ AD franchise, which also includes LNTH-2620 (Tau), NAV-4694 (β amyloid) and NeuraCeq (β amyloid). Lantheus has identified over 100 disease modifying therapies in development, which can benefit from imaging using the Tau and β amyloid imaging diagnostics. It believes that if approved, MK-6240 would complement beta-amyloid PET imaging and emerging blood-based diagnostics. Lantheus anticipates a substantial growth opportunity in this space which it estimates has a total addressable market size of $1.5 billion in 2030 and $2.5 billion by mid-2030.
MK-6240 is designed to target aggregated tau protein in the form of neurofibrillary tangles, a key hallmark of several neurodegenerative diseases, including AD. MK-6240 has demonstrated a high affinity for tau and limited off-target binding in both preclinical and clinical studies. The asset was acquired by Lantheus in 2023 and is being used in nearly 100 active clinical trials. The radiopharmaceutical has the potential to support earlier disease detection, patient staging, therapy selection and monitoring, and may help enable tau to serve as a surrogate endpoint for treatment efficacy.
LNTH-2501 Granted Target Action Date
LNTH-2501, branded Octevy and identified generically as Ga-68 edotreotide and acquired as part of the deal with Evergreen. It is a registrational-stage PET diagnostic imaging agent targeting Somatostatin Receptor-Positive (SSTR+) neuroendocrine tumors (NETs) and was submitted using the 505(b)(2) pathway. On October 30th, Lantheus announced that the FDA had assigned a Prescription Drug User Fee Act (PDUFA) date of March 29th, 2026. Lantheus estimates that there are over 170,000 persons with NETs, with many of them misdiagnosed leading to delayed detection and treatment.
SSTR+ NETs tumors overexpress somatostatin receptors, particularly SSTR2, which allows for highly specific and effective targeting. SSTR agents can bind with high affinity to these receptors, leading to localized delivery of radiation directly to the tumor cells. This targeted approach induces apoptosis in the tumor cells and even affects adjacent cells through a bystander effect, which is therapeutically beneficial beyond what conventional targeted agents provide.
LNTH-2501 can form a theranostic pair with Lantheus’ PNT-2003, which is waiting for the expiration of Novartis’ intellectual property protection for Lutathera before commercialization. Lutathera (Lu-177 dotatate) is the primary FDA-approved radiopharmaceutical for treating SSTR+ NETs and Lantheus expects to be able to take share in this market with PNT-2003 at the end of the Hatch-Waxman 30 month stay in late 2026.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] 340B pricing refers to the discounted drug prices that pharmaceutical manufacturers must offer to eligible health care organizations under the U.S. federal 340B Drug Pricing Program. The program’s purpose is to enable safety-net providers to provide more care to underserved populations. Manufacturers who participate in Medicaid and Medicare must agree to be subject to this program. The price is calculated by subtracting the unit rebate amount from the average manufacturer price (AMP). It is implemented on a delayed basis which can be several months after the AMP is determined.
[2] We calculate gross margin as 1-COGS/(revenues from Pylarify, Definity, TechneLite, NeuraCeq and Other Precision Diagnostics) as reported in company filings. We exclude revenues from Strategic Partnerships and Other from the calculation.
[3] The $309 million is what appears in the cash flow statement which contrasts with the $355.2 million reported in the text of the earnings release. Differences are due to cash balance adjustments and other undisclosed items.