By David Bautz, PhD
NASDAQ:MTVA
READ THE FULL MTVA RESEARCH REPORT
Business Update
DA-1726 48 mg Cohort Study Extended to Eight Weeks
On August 6, 2025, MetaVia, Inc. (NASDAQ:MTVA) announced that the company has extended the 48 mg multiple ascending dose (MAD) cohort of the Phase 1 clinical trial of DA-1726, the company’s oxyntomodulin (OXM) analog that is a dual agonist of the glucagon-like peptide-1 receptor (GLP1R) and glucagon receptor (GCGR), for the treatment of obesity. The purpose of the extension is to explore the safety and longer-term early efficacy along with other secondary endpoints. We anticipate topline results in the fourth quarter of 2025.
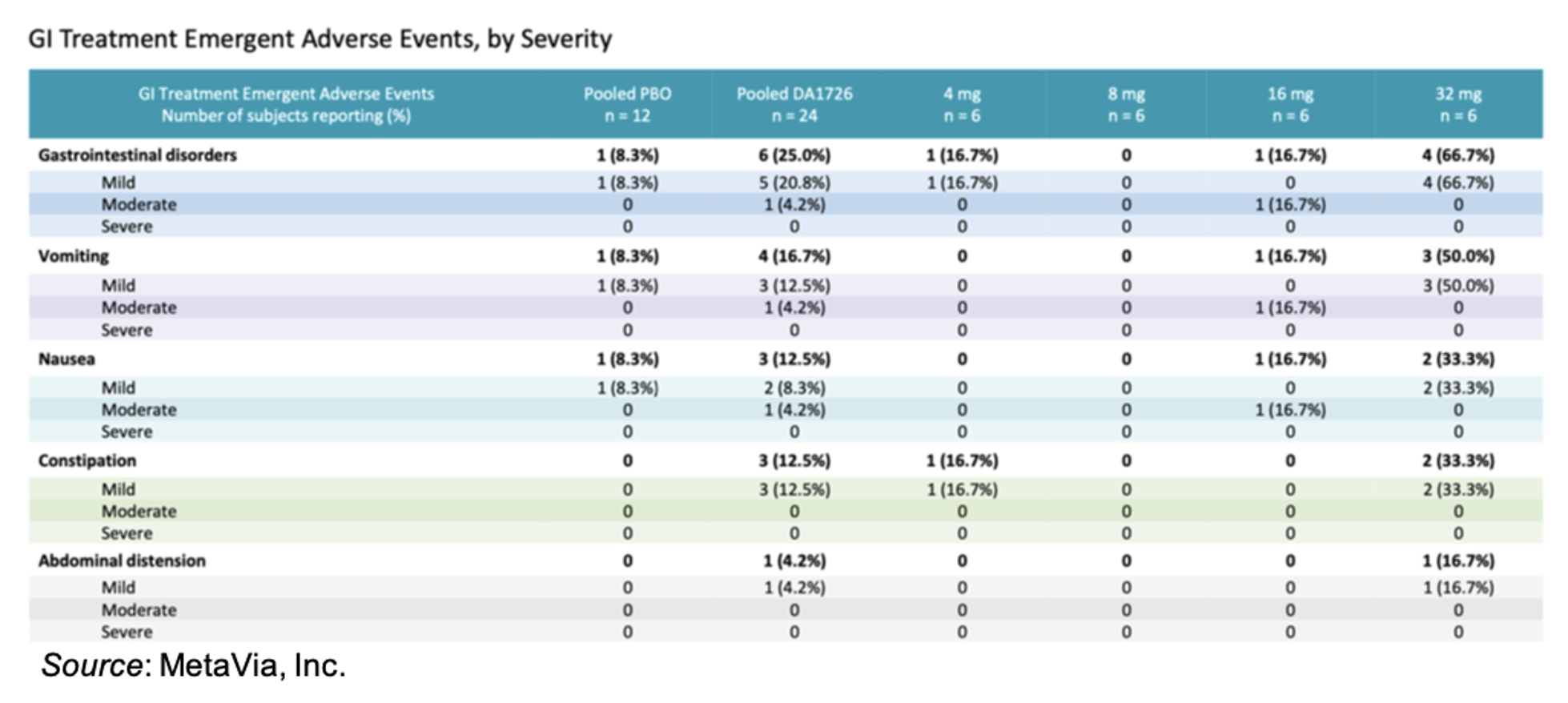
MetaVia previously announced positive results from the 32 mg dose that demonstrated a maximum reduction in body weight of -6.3%, a mean body weight reduction of -4.3% (-3.1% placebo adjusted; P=0.0005), early satiety in 83% of patients, and waist reductions of up to 3.9 inches by Day 33. In addition, DA-1726 showed an overall favorable safety and tolerability profile, as shown in the following chart. All of the adverse events (AEs) in the 32 mg cohort were considered mild to moderate and there were no treatment-related discontinuations. There was no diarrhea reported as well as no significant changes in heart rate in this cohort. The three subjects who experienced vomiting in the 32 mg cohort all reported it after the first dose but with no reoccurrence. Similarly, the two subjects who experienced mild nausea after the first dose reported that it resolved within 12 hours and there were no additional reports of nausea after the 2nd and subsequent doses. Overall, we view the AE profile observed thus far as a clear differentiator for DA-1726.
One of the concerns with targeting GLP-1 and glucagon receptors is the potential for increased heart rate. Novo Nordisk discontinued the development of NN1177, a dual glucagon/GLP-1 co-agonist, despite achieving weight loss up to 12.6% at week 12 after multiple Phase 1 trials showed an increase in heart rate (5-22 beats per minute), impaired glucose tolerance, and increased markers of inflammation (fibrinogen and C-reactive protein) (Friedrichsen et al., 2023). Thus, determining cardiac safety is of the utmost importance for DA-1726 given its similar mechanism of action. In the Phase 1 study, the mean heart rate for subjects on DA-1726 showed a slight decrease from baseline in all treatment groups besides the 16 mg group where the baseline was significantly lower than the others. In addition, there were no onsets of QTcF (QT interval corrected for heart rate using the methods of Fridericia) and no risk of cardiovascular events.
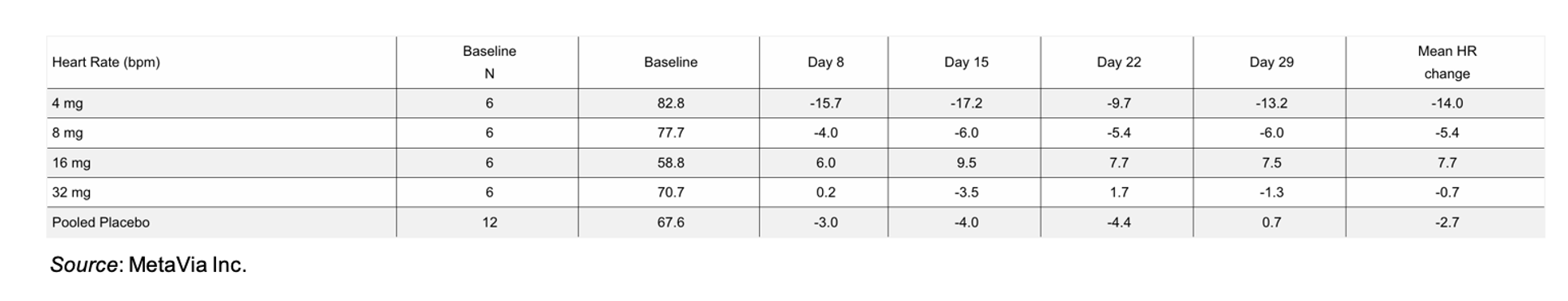
We’re very encouraged by the results from the MAD Part 2 of the Phase 1 trial of DA-1726 as the drug appears to be effective, well tolerated, and shows no signs of any potential negative cardiovascular effects. We look forward to reviewing the 8-week data from the 48 mg cohort later in 2025.
AI-Driven Collaboration with Syntekabio
On August 4, 2025, MetaVia announced a research collaboration with Syntekabio, Inc. to identify additional disease targets for DA-1241, the oral G-protein coupled receptor 119 (GPR119) agonist that is being developed as a treatment for metabolic dysfunction-associated steatohepatitis (MASH). Due to the strong safety and tolerability profile exhibited in the Phase 2a trial of DA-1241, the company will work with Syntekabio’s proprietary DeepMatcher® compound-protein interaction (CPI) AI prediction platform in an effort to expand the potential uses of the drug. Specifically, a large-scale virtual screening of more than 1,700 validated protein targets will be conducted to discover new, high-potential indications while ensuring specificity and minimizing the risk of off-target effects. Initial insights from the collaboration could be released by the company before the end of 2025.
While the company will be pursuing potential additional indications for DA-1241, MASH will remain the primary development focus. In support of this, MetaVia is working to schedule an ‘End-of-Phase 2’ meeting with the U.S. FDA to determine next steps for the MASH program.
Financial Results
On August 7, 2025, MetaVia announced financial results for the second quarter of 2025. As expected, the company did not report any revenues in the second quarter of 2025. R&D expenses for the second quarter of 2025 were approximately $2.3 million compared to approximately $8.1 million for the second quarter of 2024. The decrease was primarily due to decreased expenses related to DA-1241 and DA-1726. G&A expenses were approximately $2.0 million for the second quarters of both 2025 and 2024.
As of June 30, 2025, MetaVia had approximately $17.6 million in cash and cash equivalents. We estimate the company has sufficient capital to fund operations into the first quarter of 2026. As of August 7, 2025, MetaVia had approximately 24.2 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 32.2 million.
Conclusion
We are very interested to see the results from the 8-week, 48 mg cohort of the DA-1726 Phase 1 trial, which we anticipate being announced in the fourth quarter of 2025. Thus far, DA-1726 has had a very favorable safety and tolerability profile, with positive signs of early efficacy. If this trend continues in the 8-week data, we believe DA-1726 could become a very competitive asset in the obesity space. As we await the 8-week data there have been no changes to our model and our valuation remains at $21 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.