By David Bautz, PhD
NYSE:NNVC
NanoViricides, Inc. (NYSE:NNVC) is a clinical-stage biopharmaceutical company developing a new class of broad-spectrum antiviral drugs known as “nanoviricides”. These agents mimic cell surface structures to trap and dismantle viruses before they infect host cells. The company’s lead development candidate, NV-387, has shown promising preclinical activity against multiple viruses, including SARS-CoV-2, Respiratory Syncytial Virus (RSV), Influenza, and Mpox (monkeypox). In May 2025, NanoViricides received ethics approval in the Democratic Republic of Congo (DRC) to proceed with a Phase II clinical trial of NV-387 for Mpox clade I, which is the deadlier of the two known clades. This positions NanoViricides at the forefront of developing urgently needed antiviral treatments for one of the world’s most concerning re-emerging infectious diseases.
Background on Nanoviricides
The company’s core nanoviricide® technology utilizes molecular mimicry to bind virus particles and prevent their entry into the cell. Rather than targeting the virus directly through specific antibodies or depending on the host immune response, nanoviricides use a bio-mimetic approach. These compounds are engineered to resemble the human cell receptors that viruses latch onto, particularly sulfated proteoglycans (SPGs) like heparan sulfate in the case of NV-387, the lead candidate. A nanoviricide consists of a small molecule ligand that mimics the receptor utilized by the virus to gain cellular entry. The ligand is covalently attached to a flexible polymer backbone comprised of polyethylene glycol (PEG) and alkyl chains. The PEG forms a hydrophilic shell while also conferring non-immunogenicity. The alkyl chains make up the flexible core. Multiple chemically reactive sites allow for “packaging” of one or more active pharmaceutical ingredients (APIs) within the core of the nanoviricide. This structure then forms a flexible nanomicelle via self-assembly. A cartoon representation of a nanoviricide is shown below.
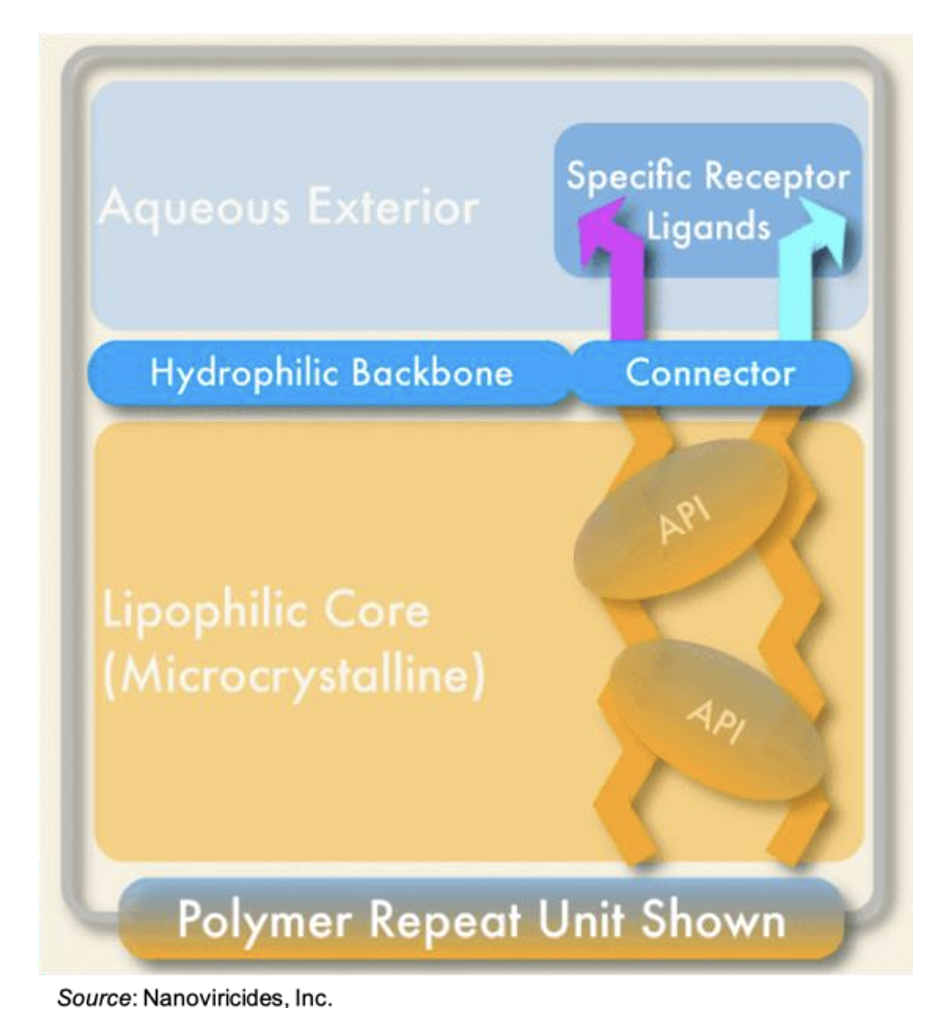
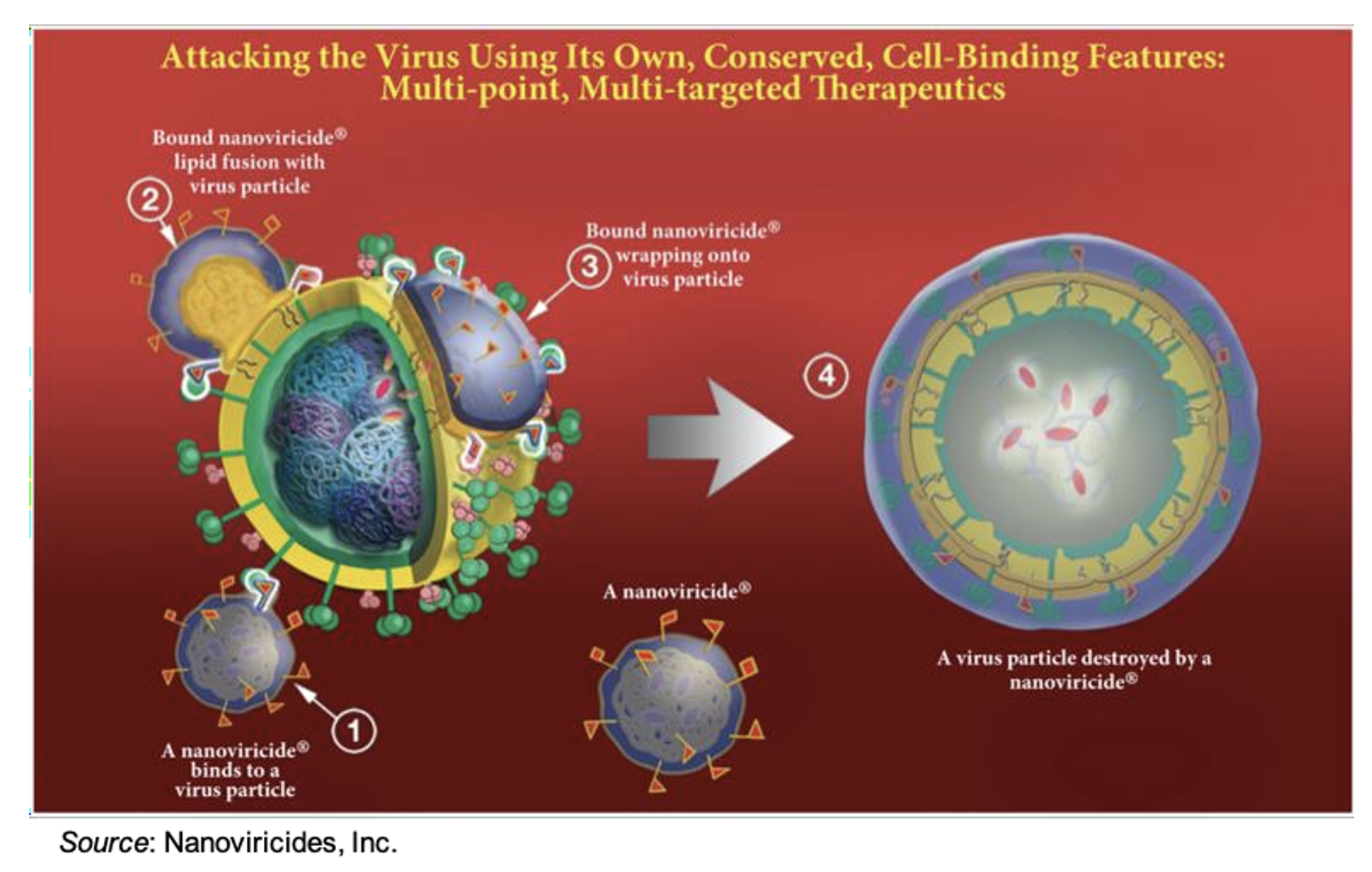
Upon encounter with a target virus, binding occurs between the ligand displayed on the nanoviricide micelle and the viral receptor protein. The micelle then fuses with the lipid-coated surface of the virus through phase-inversion and “lipid-lipid mixing”, a well-studied physicochemical effect. This is shown in the following figure. Since the binding site on the human cellular receptor for a particular virus does not change, despite mutations occurring to the receptor-binding domain and other areas of the virus, it is thought that nanoviricides will not be susceptible to viral mutations that are known to render other treatments ineffective.
The use of nanoviricides provides several key advantages over currently available antiviral therapies, including:
- Less susceptible to viral resistance: Since nanoviricides contain host-like binding domains rather than a synthetic molecule or antibody, it will likely be much more difficult for the virus to evolve resistance.
- Broad-spectrum activity: Since over 90% of human pathogenic viruses use a similar first attachment point to gain entry into human cells (Heparan SPGs), a single drug like NV-387 can potentially target many
- Host-independent action: Unlike vaccines or antibody therapies, nanoviricides do not require a functioning immune system to exert their antiviral activity.
- Complementary use: As opposed to other antiviral agents under development that only target the intracellular life cycle of viruses, nanoviricides attack the virus outside the cell and can be loaded with antiviral agents to work synergistically inside the cell as well.
- Multiple modes of administration: Nanoviricides can be administered intravenously, orally, or through inhalation. For the SARS-CoV-2 program the company utilized oral gummies, however for other programs, such as the treatment of severe acute respiratory infection of viral origin the drug could be delivered intravenously to hospitalized patients.
In April 2024, NanoViricides reported the completion of a Phase 1 clinical trial of NV-387 in India. A single ascending dose part of the study (Phase 1a) and a multiple ascending dose part of the study (Phase 1b) were both completed with healthy subjects. There were no reported adverse events for either drug product studied, an oral syrup and an oral gummy form of NV-387. The trial was scheduled to enroll COVID-19 patients, however no infected patients could be identified despite the addition of a second study site. The company anticipates receiving the final clinical trial report in the near future. Importantly, the study was able to show that NV-387 was safe and well tolerated and provided the basis for the proposed Phase 2 trial in Mpox.
Background on MPox
MPox (monkeypox) is a viral illness caused by the monkeypox virus, a species in the genus Orthopoxvirus, which includes variola, cowpox, and vaccinia (the cause of smallpox) (Alakunle et al.,2020). There are two distinct clades: clade I (with subclades Ia and Ib) and clade II (with subclades IIa and IIb) (Happi et al., 2022). Prior to 1970 there were no documented reports of human MPox infections. The first human MPox case was a 9-month-old boy in the Democratic Republic of the Congo (DRC) in August 1970 (Ladnyj et al., 1972). Since that time, MPox infections have become endemic in multiple African countries, including Benin, Cameroon, DRC, Gabon, Ivory Coast, Liberia, Nigeria, the Republic of the Congo, Sierra Leone, and South Sudan (WHO). Between 1970 and 2021, MPox cases were sporadic and limited to the endemic countries. In 2003 there was an MPox outbreak in the U.S. linked to imported rodents from Ghana (Anderson et al., 2003). A global MPox outbreak of the clade IIb virus started in May 2022 and was declared a public health emergency of international concern in July 2022 (Thornhill et al., 2022). By October 2023 there were almost 100,000 confirmed cases reported from 116 countries (WHO).
The MPox virus can enter the host via the respiratory tract and damaged skin. During the most recent outbreak of MPox infections the most commonly reported symptoms were skin lesions, fever, lymphadenopathy, fatigue, myalgia, and headache (Liu et al., 2023). Symptoms typically last 2-4 weeks and while most will make a full recovery, some individuals can become very sick with complications such as pneumonia or infections of the skin, blood, and other organs. Prior to the 2022 outbreak, the case fatality rate (CFR) was 8.7% and varied based on the clade of the virus (10.6% for clade I and 3.6% for clade II) (Bunge et al., 2022). In contrast, the 2022 outbreak caused by MPox Clade IIb had an estimated CFR of 0.08% (WHO).
Treatment of MPox infection is primarily aimed at supportive care that is focused on alleviating pain, pruritis, and ensuring adequate hydration. The prevention and treatment of secondary bacterial infections is also critical for ensuring a full recovery. Two different trials tested the drug tecovirimat for the treatment of MPox. Tecovirimat is one of two antiviral drugs approved for treating orthopoxvirus infections (including MPox) with brincidofovir being the other. Both drugs are stockpiled by the U.S. for smallpox preparedness.
- In August 2024, the results of the PALM007 trial showed that tecovirimat did not reduce the duration of lesions in children and adults with clade I MPox in the DRC. The drug was safe and well tolerated with no drug-related serious adverse events.
- In December 2024, the Study of Tecovirimat for MPox (STOMP) trial stopped further enrollment of patients after the Data Safety and Monitoring Board (DSMB) review after results showed that the drug did not reduce the time to lesion resolution or have an effect on pain among adults with mild to moderate clade II MPox and a low risk of developing severe disease.
These results show that tecovirimat is not an effective treatment for MPox infections. Similar unsuccessful results were seen in the treatment of three Mpox patients in the U.K. with brincidofovir between 2018 and 2021; all three patients developed elevated liver enzymes and cessation of therapy (Adler et al., 2022). While another trial of brincidofovir for the treatment of MPox has been initiated under the MPox Study in Africa (MOSA) protocol (McCarty et al., 2025), there exists a critical unmet need for additional MPox treatment options.
NV-387 for the Treatment of MPox
In May 2025, NanoViricides announced that the company received approval from the National Ethics Committee for Health (CNES) of the Ministry of Public Health (MSP) of the DRC, which means a Phase 2 clinical trial of NV-387 for the treatment of patients with MPox disease is cleared for further regulatory filing of a Clinical Trial Application (CTA). The company previously announced it had engaged a Contract Research Organization (CRO) to conduct the Phase 2 trial. The CRO subsequently engaged the Medical Hospital at the University of Kinshasa as the site for the study. The full CTA is expected to be filed in August 2025, with the clinical trial initiating in the fourth quarter of 2025. NanoViricides has already initiated manufacturing of drug product for the trial in its wholly owned manufacturing facility.
The planned Phase 2 trial is expected to enroll 80 hospitalized MPox patients and will consist of two parts that will compare the standard of care (SOC) to SOC + NV-387. The Phase 2a portion of the study will evaluate a fixed dose of NV-387 administered to MPox patients for six days, at which time the patients will be evaluated to determine the safety and tolerability of the drug and whether dosing should continue for longer. Ten patients will be treated with SOC and ten will be treated with SOC + NV-387. The Phase 2b portion of the study will compare SOC to SOC + NV-387 in MPox patients randomized 1:2 to each cohort using the dosing schedule determined in the Phase 2a portion. A total of 60 patients will be evaluated in the Phase 2b portion of the trial. The efficacy outcomes of the study are complete lesion recovery, elimination of new rash formation, viral load, and 28-day outcome. If the trial were to initiate in the fourth quarter of 2025 we anticipate topline results in the first quarter of 2026.
Market Opportunity in MPox
There are multiple reasons that NanoViricides has decided to focus on MPox. First, the continuing epidemic in the African Region and the failure of tecovirimat has highlighted the need for additional treatment options for MPox patients. In addition, there should be a large pool of available patients to conduct the trial in a timely manner given the continued MPox outbreak in the DRC and conducting trials in the African Region is far less expensive than other parts of the world, thus allowing the company to conserve its financial resources. Second, proof of efficacy for NV-387 in MPox would validate the company’s animal model data and establish that those data are predictive of clinical outcomes. The company has substantial data showing NV-387 is efficacious in multiple viral models, including RSV, influenza, Covid, and MPox, thus success in MPox could serve as a positive read through to additional indications for NV-387. Third, there are only two approved therapies that are part of the U.S. Strategic National Stockpile (SNS) for the treatment of smallpox, and neither drug has shown efficacy against MPox in humans. Thus, were NV-387 shown to be effective against MPox, it could present an opportunity for that drug to be tested for use in bioterrorism readiness and potentially to be included in the SNS.
The inclusion of NV-387 in the SNS would be a tremendous opportunity for NanoViricides. Siga Technologies (SIGA), which manufactures TPOXX® (tecovirimat), signed an initial contract with the
U.S. Biomedical Advanced Research and Development Authority (BARDA) in September 2018 for up to 1,488,000 courses of oral TPOXX and 212,000 courses of IV TPOXX to be delivered to the SNS. This contract has been updated and amended but as of December 31, 2024 it contemplates up to approximately $602.5 million of payments (SIGA 2024 10-K). Thus, we estimate if NV-387 showed efficacy against MPox it could represent a multi-hundred-million-dollar opportunity.
Conclusion
The treatment of MPox represents a substantial opportunity for NanoViricides as it looks to validate its lead development candidate NV-387 as a next-generation antiviral therapy. We anticipate the company filing a CTA for a Phase 2 trial in MPox in the DRC in the third quarter of 2025 such that the trial can initiate before the end of the year. Topline results may then be available in the first quarter of 2026. Positive results would help support the inclusion of NV-387 in the SNS and would help to validate NanoViricides animal model data, which thus far has shown NV-387 is active against a multitude of viruses. We look forward to updates regarding the MPox program and the potential for efficacy data in 2026.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.