By John Vandermosten, CFA
NYSE:PLX
READ THE FULL PLX RESEARCH REPORT
Third Quarter 2025 Financial and Operational Review
Protalix BioTherapeutics, Inc. (NYSE:PLX) announced 3Q:25 financial and operational results in a November 13th, 2025, press release and in its Form 10-Q filing. The reports were followed by a conference call, which discussed recent achievements, regulatory updates, trial timelines and financial performance. During the third quarter, Protalix revenues were flat overall as growth in Elelyso sales were offset by a decline in Elfabrio sales, which was attributable to Chiesi’s initial inventory build in 3Q:24. Protalix and Chiesi’s application for a longer dosing regimen for Elfabrio was denied, leading them to seek re-examination of the opinion from the European Medicines Agency (EMA).
Management refined its timeline to launch the PRX-115 Phase II trial in the next few weeks, enroll the first patient before year-end end and generate topline results in 2027. Gilad Mamlok takes over as CFO to review financial performance and joins the conference call for the first time. Protalix has also participated in investor conferences, including HC Wainwright and the Investor Summit, since our last update.
Financial results for the quarter ending September 30th, 2025, compared to the same quarter in the prior year:
- Revenues were $17.9 million, down 1% from $18.0 million, attributable to an increase in sales of Elelyso offset by a decline in Elfabrio to Chiesi. Sales to Pfizer were $2.8 million, down by $625,000, and sales to Brazil were $6.1 million, up $4.1 million due to timing of orders. Protalix also recognized $178,000 in license and R&D services revenue;
- Cost of revenue was $8.3 million vs. $8.4 million, declining at a similar rate to revenues;
- Research and development expenses increased to $4.5 million from $3.0 million. Higher salary, materials, and other expenses were partially offset by lower subcontractor-related expenses, which supported preparation activities for the anticipated Phase II PRX-115 study;
- Selling, general, and administrative expenses rose 13% to $2.9 million versus $2.6 million on higher salary and selling expenses;
- Net financial income was $108,000 compared to ($148,000). The change in financial expense was due to the absence of notes and their associated interest, which were present for most of the quarter in 3Q:24;
- Income tax benefit of $116,000 compared to an income tax expense of $607,000 due to faster deductibility of research and development expenses resulting from new tax legislation in the United States;
- Net income was $2.3 million versus $3.2 million, or $0.03 per share versus $0.04 per share;
The cash and equivalents balance on September 30th, 2025, totaled $29.4 million versus $34.8 million at the end of 2024. During the first nine months of 2025, Protalix generated $9.2 million from the sale of common stock and exercise of warrants and options. Cash burn was $15.3 million for the first nine months. Following the end of the quarter, stock options were exercised, generating $94,000 in proceeds.
Four Week Dosing of Elfabrio
In December 2024, Protalix’s partner Chiesi submitted a Variation Application to the EMA that requested a change in the dosing regimen for Elfabrio. Based in part on the findings in the BRIGHT study and on new pharmacokinetic data, the sponsors sought a less frequent dosing regimen at a dose of 2 mg/kg body weight administered every four weeks in adult patients with Fabry disease in the European Union. Analysis of the BRIGHT study concluded that treatment with Elfabrio every four weeks could offer a new treatment option for patients with Fabry disease.[1]
On October 17th, 2025, Chiesi and Protalix announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) had issued a negative opinion on the request to approve the dosing regimen of 2 mg/kg body weight infused every 4 weeks for Elfabrio.
Two and a half weeks after the negative opinion, Chiesi and Protalix issued a press release stating that they will seek re-examination of the EMA’s negative opinion for Elfabrio and the four-week alternative dosing regimen. The sponsor must submit a written notice to the EMA within 15 days of the CHMP opinion and 60 days later submit the grounds for examination. A different rapporteur and co-rapporteur will be appointed to conduct the re-examination. Chiesi and Protalix have consultants and/or internal personnel with EMA and CHMP experience who will help develop the argument for four-week dosing. In the meantime, two-week dosing remains approved and the standard for administering Elfabrio.
PRX-115
Results from the Phase I PRX-115 trial were presented at the American College of Rheumatology (ACR) Convergence annual meeting last November. The poster is entitled Prolonged Plasma Urate Lowering after a Single Intravenous Administration of PRX 115, a Novel PEGylated Uricase, in Participants with Elevated Urate Levels and was presented by Protalix’s Dr. Orit Cohen Barak. Results from the Phase I support moving on to the next stage of development. Planning for the PRX-115 Phase II trial was conducted during 2025, and an investigational new drug application was filed with the FDA in October. It was cleared in November, and Protalix anticipates starting its Phase II study for PRX-115 in the next few weeks.
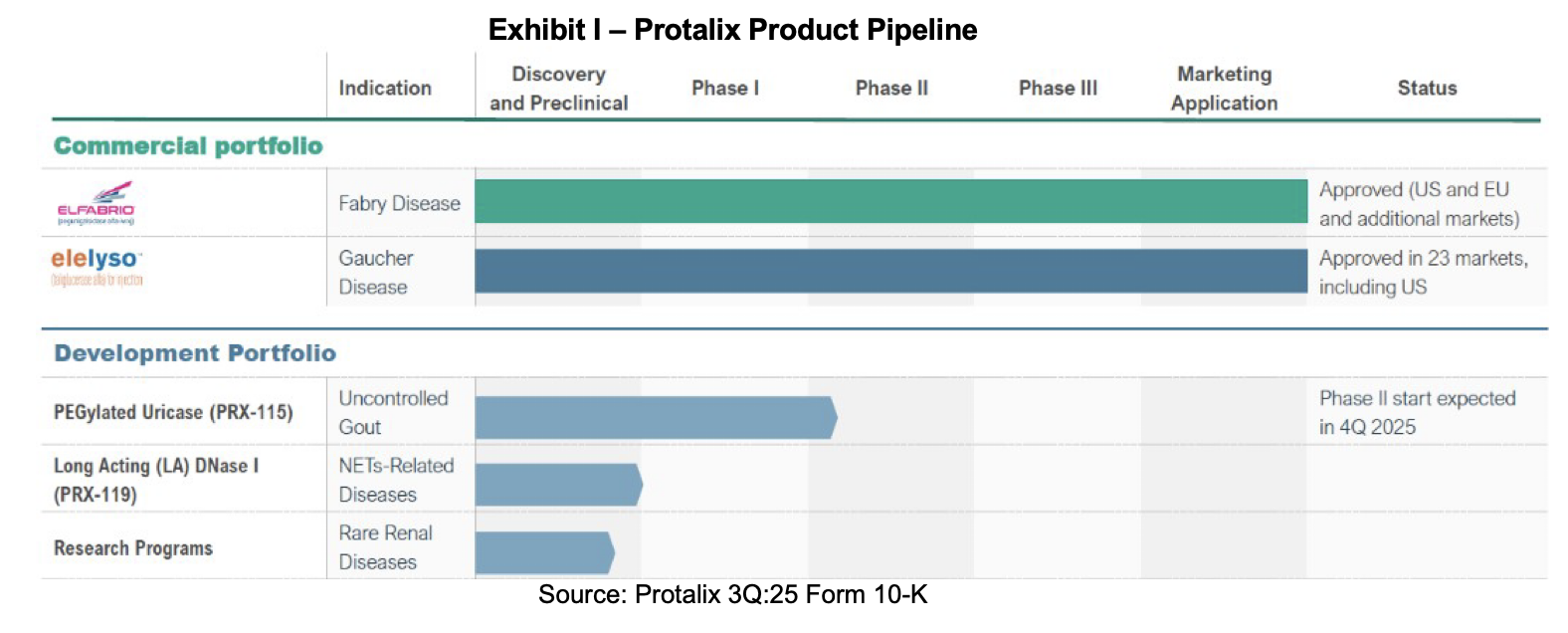
Pipeline
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] Holida, M., et al. A phase III, open-label clinical trial evaluating pegunigalsidase alfa administered every 4 weeks in adults with Fabry disease previously treated with other enzyme replacement therapies, Journal of Inherited Metabolic Disease. October 2024.