By David Bautz, PhD
NASDAQ:SNGX
READ THE FULL SNGX RESEARCH REPORT
Business Update
Dusquetide (SGX945) Granted Orphan Drug Designation for the Treatment of Behcet’s Disease
On August 18, 2025, Soligenix, Inc., (NASDAQ:SNGX) announced that the U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation (ODD) to dusquetide, the active ingredient in SGX945, for “treatment of Behcet’s Disease”. This designation comes following the release of highly encouraging Phase 2a data for SGX945 in Behcet’s Disease (BD) in which SGX945 exhibited biological efficacy.
The Phase 2a study was modeled after the Phase 3 clinical trial of apremilast (Otezla®), which served as the basis for marketing approval of that drug for oral ulcers in BD (Hatemi et al., 2019). A total of eight patients were enrolled in the study and administered SGX945 IV twice a week for four weeks. A four-week follow-up was also conducted following cessation of SGX945 treatment. The primary endpoint was the area under the curve (AUC) of the mean number of ulcers versus time.
The results of the trial showed that after 4 weeks of treatment, the SGX945-treated group had a 40% improvement relative to the placebo group from the Phase 3 apremilast study. This compares to a 37% improvement for the apremilast group relative to placebo. The improvement in the SGX945-treated group was sustained through the 4-week follow-up period. A 32% improvement relative to placebo was noted at Week 8, even though treatment had stopped at Week 4. Apremilast-treated patients, who were administered drug continuously through Week 12, showed a 41% improvement at Week 8.
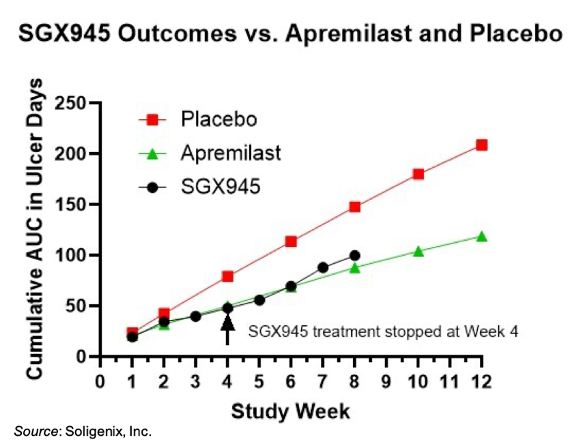
Notable reports from the SGX945 study include 7/8 patients reporting a perceived benefit from treatment, including reduced duration of oral ulcers, reduced number of oral ulcers, and reduced oral pain. One of the patients had a skin ulcer that resolved during the 4-week treatment period with SGX945. Importantly, SGX945 was well tolerated with no treatment-related adverse events. Commonly reported adverse events in the apremilast study included diarrhea (41%) and nausea (19%), neither of which were reported for SGX945-treated patients.
Since SGX945 is currently administered intravenously, Soligenix is planning to reformulate SGX945 as a subcutaneous injection that can administered by patients at home, similar to GLP-1 therapies. Following the reformulation, we anticipate the company performing a placebo-controlled Phase 2b trial of SGX945 in BD patients.
Background on BD
BD is a chronic recurrent multisystemic disease that causes oral aphthous ulcers, genital ulcers, skin lesions, and other pathologies (Mendes et al., 2009). Interestingly, the epidemiology of BD is distributed along the ancient Silk Road from Mediterranean countries (Turkey has 370 cases per 100,000 population), to Middle Eastern and East Asian countries. In contrast, there are very few cases found in Northern Europe (0.64 cases per 100,000 population), North America (0.12-0.33 cases per 100,000 population), Australia, and Africa (Deuter et al., 2007). Thus, BD is an orphan disease in the U.S., however there may be as many as 500,000 people worldwide with the disease.
There are no standardized regimens for treating BD. Systemic corticosteroids, interferon-alpha (INF-a) therapy, and anti-tumor necrosis factor alpha (TNF-a) therapy are all used as first-line agents and have shown good efficacy. As mentioned above, apremilast was approved by the FDA for the treatment of oral ulcers in BD patients.
Results from Phase 2a Psoriasis Trial Expected Before End of 2025
Soligenix is currently evaluating SGX302 (synthetic hypericin) as a treatment for mild to moderate psoriasis. The Phase 2a trial is a randomized, double blind, placebo controlled study this is enrolling patients with mild-to-moderate, stable psoriasis covering 2% to 30% of their body. Placebo or SGX302 is being administered twice weekly for up to 18 weeks, with each treatment consisting of application followed approximately 24 hours later with visible light activation. The efficacy endpoints include lesion clearance along with patient quality of life indices.
Following evaluation of the first five subjects enrolled in the trial, a clear biological signal was seen as evidenced by an improvement in PASI (psoriasis area and severity index) score. In Cohort 2, five additional patients were treated with a more rapid escalation and higher final dose level of light to more closely match how the drug will be utilized in the “real world” clinical setting. The results from Cohort 2 showed that there were no drug-related adverse events reported. Four of the patients were evaluable (one patient withdrew early in the study for personal reasons unrelated to the study), with two of them achieving an Investigator Global Assessment (IGA) score of 1 (“Almost Clear”), a standard clinical measure for treatment success in psoriasis. In addition, the patients in Cohort 2 had a mean drop of approximately 50% in the PASI score.
The company is continuing to test different light levels and light intensity/duration to optimize the conditions for psoriasis patients and we anticipate results from the next cohort of patients before the end of 2025. Once the optimized conditions are established, the ultimate goal is to transition to “at home” use prior to initiating a Phase 3 trial. The ability for patients to treat themselves at home would be a clear differentiator from other psoriasis treatments on the market.
Financial Update
On August 14, 2025, Soligenix announced financial results for the second quarter of 2025. The company reported no revenue for the second quarters of 2025 and 2024. R&D expenses for the second quarter of 2025 were $1.7 million, compared to $0.5 million for the second quarter of 2024. The increase was primarily due to costs associated with the Phase 2 study in Behcet’s Disease and the confirmatory Phase 3 CTCL trial along with third-party manufacturing costs. G&A expenses for the second quarter of 2025 were $1.1 million, compared to $1.2 million for the second quarter of 2024. The decrease was primarily due to decreases in professional expenses.
Soligenix exited the second quarter of 2025 with approximately $5.1 million in cash and cash equivalents. We estimate this is sufficient to fund operations through the first quarter of 2026. As of August 7, 2025, Soligenix had approximately 4.3 million shares outstanding, and when factoring in stock options and warrants, a fully diluted share count of approximately 6.0 million.
Conclusion
The FDA granting ODD to dusquetide is further confirmation of the encouraging results seen in the Phase 2a trial in Behcet’s Disease. We look forward to additional updates on the reformulation of SGX945 and potential timelines for when a Phase 2b trial could get underway. In addition, we look forward to data from the Phase 2a psoriasis trial, which we anticipate before the end of 2025. With no changes to our model our valuation remains at $35 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.