By David Bautz, PhD
NASDAQ: ASBP
Aspire Biopharma Holdings, Inc. (NASDAQ: ASBP) is attempting something relatively uncommon in the biotech space: building a pharmaceutical platform while simultaneously testing commercialization through a consumer-facing product.
The company’s strategy is centered on a patent-pending sublingual drug delivery system designed to improve how quickly and efficiently certain medications are absorbed. By reformulating already approved drugs, Aspire is attempting to reduce development risk while targeting meaningful improvements in performance, particularly in situations where speed of action is critical.
At the same time, the company has launched “Buzz Bomb,” a single serving 50-mg caffeine product sprinkled under the tongue, targeting the large and growing wellness, energy, and sports performance market. This commercial supplement opportunity does not require FDA approval and provides Aspire with an early opportunity to generate revenue while it pursues development of a variety of existing approved drug formulations that could utilize its sublingual technology to improve efficacy or reduce side effects.
Aspire represents a differentiated approach within the broader biotech landscape by combining platform innovation with early-stage commercialization. If successful, Aspire’s approach could offer a unique path to value creation that combines lower-risk drug reformulation with a scalable delivery platform.
A Platform-Driven Approach to Drug Delivery
Aspire Biopharma is not a single-asset company, but rather is developing a drug delivery platform that can be utilized to enhance multiple classes of already approved pharmaceuticals. Its core technology is designed to enable rapid sublingual absorption that delivers drugs directly into the bloodstream through oral tissues rather than through the digestive system, thus bypassing first-pass metabolism in the liver.
This distinction is important. Traditional oral medications must pass through the gastrointestinal tract and liver before entering systemic circulation, which can delay onset and reduce the amount of active drug available. Aspire’s approach is designed to bypass this process, potentially improving both speed of onset and bioavailability.
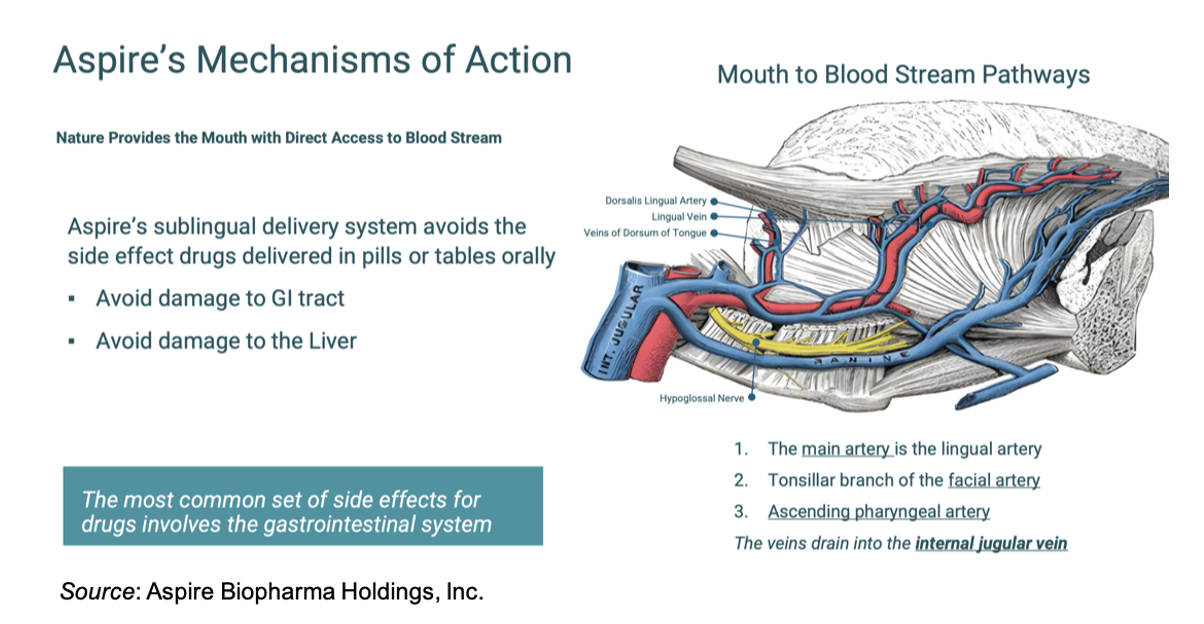
Aspire’s delivery system is engineered to support multiple formats, including powders, tablets, and oral suspensions, which suggests that the platform could be applied broadly across a range of therapeutic areas and compounds. In addition, it offers a number of advantages for drug delivery, including:
- Fast Acting: Medications enter the bloodstream much more rapidly compared to oral tablets and capsules
- Convenience and Ease of Use: Dissolves easily under the tongue; perfect for those who cannot swallow pills
- Dose Management: Drugs avoid first-pass metabolism in the liver, thus potentially increasing the amount of active compound in the bloodstream
- Bypasses Digestive Tract: Could potentially reduce or eliminate adverse reaction in the gastrointestinal tract
- Reduces Pills and Tablets: Decreases burden on patient and caregivers
Rather than developing entirely new drugs, Aspire is focused on reformulating existing approved generic drugs that address large markets. This strategy allows the company to potentially leverage known safety profiles while concentrating on improving how drugs are delivered and how quickly they act.
Sublingual Aspirin as a Proof of Concept
Aspire’s most advanced pharmaceutical program is a high-dose sublingual aspirin formulation, which the company is positioning for use in acute settings such as suspected heart attack or stroke.
In a completed clinical study, Aspire reported that its sublingual aspirin achieved significantly faster absorption and therapeutic effect compared to standard chewable aspirin tablets. The following graph shows that meaningful drug impact levels were reached within two minutes following dosing, five times faster than a chewable aspirin tablet.
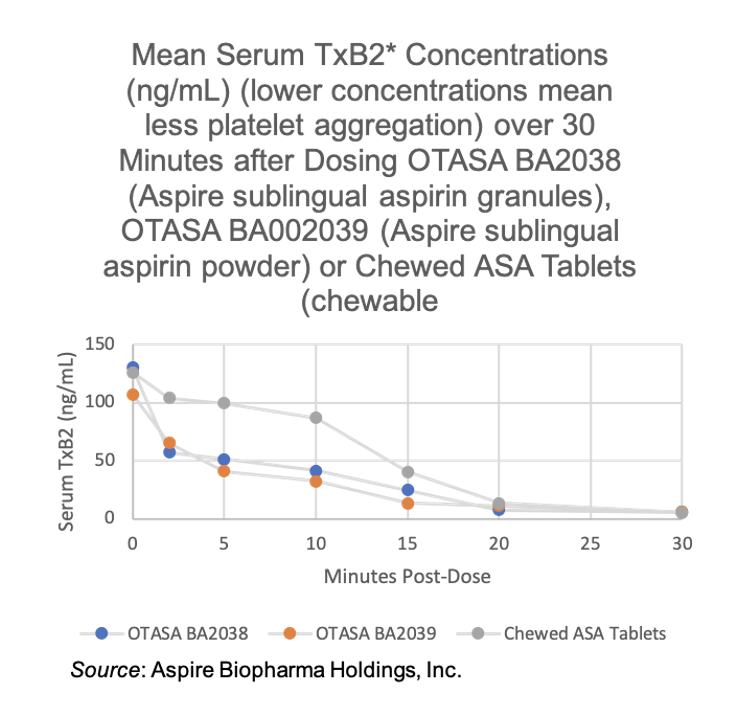
This speed advantage could be particularly important in emergency situations, where faster platelet inhibition may help reduce damage during a cardiac event. However, it could also offer an advantage for other pain management indications as well (headache, post-surgery, general pain relief, etc.).
The company’s go-to-market strategy will likely involve a product launch initially in the prescription market with a licensing or partnership agreement. This could also open the door for additional licensing opportunities with partners experienced in end-to-end marketing and distribution. The market opportunity for sublingual aspirin is significant, with the combined opioid and non-opioid markets currently estimated at approximately $80 billion, which is expected to grow to $100 billion over the next five years.
Aspire expects to conduct the final clinical trial for high-dose aspirin in mid-2026, followed by a potential regulatory submission later in the year. Notably, the company has received feedback from the FDA supporting a potential regulatory pathway under the 505(b)(2) framework, which is commonly used for reformulated versions of existing drugs. If successful, sublingual aspirin could serve as an initial validation of the platform’s ability to deliver clinically meaningful improvements.
A Broad Pipeline of Reformulated Drugs
Beyond aspirin, Aspire is already positioning its platform across multiple therapeutic categories. The company has outlined additional programs involving reformulated versions of widely used drugs, including:
The company is currently undergoing formulation work on alprazolam and expects to commence its first clinical trial in 3Q 2026. The other therapeutic targets in the pipeline are in the planning stage, but they highlight the potential scalability of the platform. If Aspire can demonstrate consistent improvements in absorption and onset across different compounds, it could open the door to a broader portfolio of reformulated products targeting large, established markets.
This platform-driven model introduces a different type of upside profile compared to traditional biotech companies. Rather than relying on the success of a single novel drug, Aspire is attempting to build a system that can be applied across multiple existing therapies.
Early Commercialization and Real-World Validation with Buzz Bomb
In parallel with its pharmaceutical development efforts, Aspire has entered the consumer market with Buzz Bomb, a single-serving caffeine supplement designed for quick energy and convenience. The product delivers 50mg of caffeine in a compact powder format that is used without water, offering a convenient alternative to traditional energy drinks, soda, coffee, or pre-workout supplements.

Aspire is positioning Buzz Bomb around several potential advantages, including faster impact, precise servings, zero sugar, and convenience, which could appeal to athletes and consumers seeking more control over their caffeine intake. The company has already taken steps to scale production and distribution, including a broader product relaunch and expanded marketing efforts.
The pre-workout supplement market is currently estimated at approximately $20 billion and is expected to reach approximately $28 billion by 2030 (Grand View Research). This growth is being driven by a number of factors, including rising fitness culture, expanding demographics, and digital fitness and influencer-led marketing. The market is highly competitive with a wide range of options for energy boosting and hydration products. However, most products are based on a “mix + water” combination that takes upwards of 20-30 minutes to provide benefits. Consumers are most interested in products that show immediate results and are easy to use, two areas where Buzz Bomb could differentiate itself, drive market penetration, and demonstrate the commercial opportunity for Aspire’s delivery approach in a real-world setting.
Intellectual Property and Financial Position
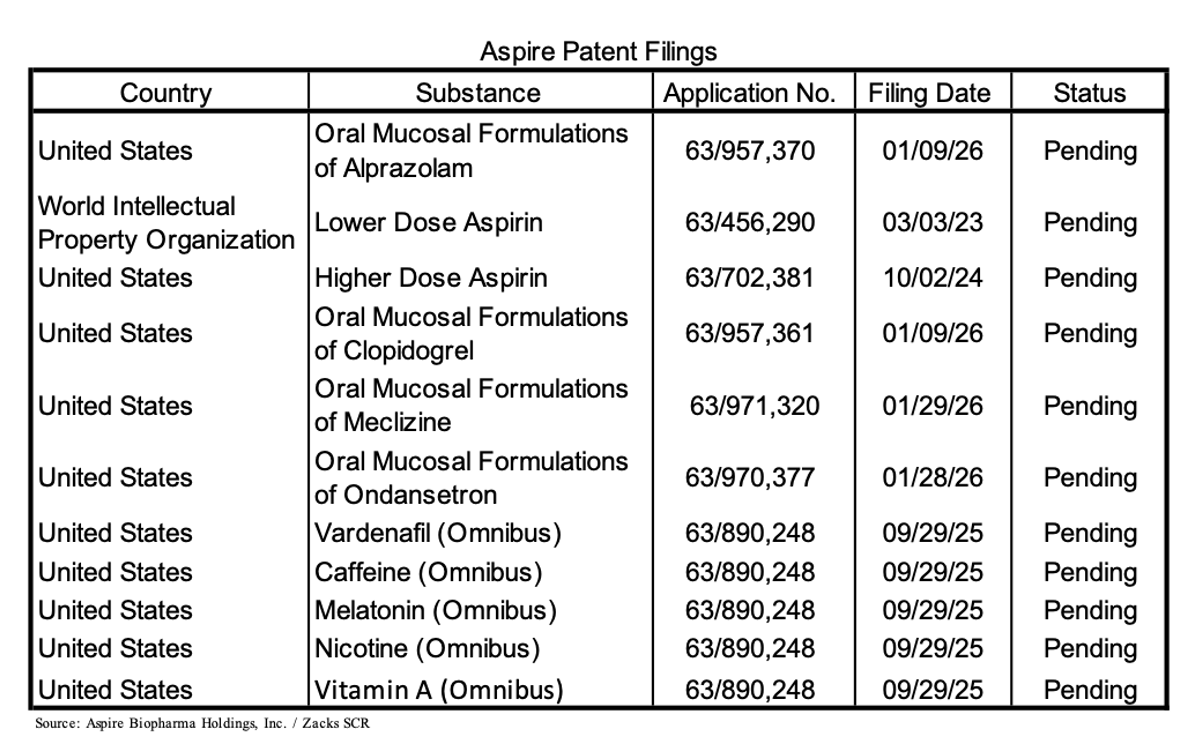
Aspire’s goal is to build a foundation of intellectual property protection around its sublingual delivery platform. The current strategy is centered on a series of recently filed and pending applications covering its formulation technology, delivery methods, and potential use across eleven drugs and supplements. These filings include both specific product formulations, such as the sublingual aspirin program, as well as broader claims designed to protect the underlying delivery platform across different therapeutic areas.
The company has also taken a layered approach to its intellectual property, combining patent filing with trade secrets, proprietary processes, and ongoing development work. In addition, Aspire has filed an “omnibus” patent application intended to extend protection across multiple classes of compounds, reflecting a platform-oriented strategy rather than a single-product focus.
The following table provides an overview of the company’s patent filings as of February 2026.
To strengthen its balance sheet, Aspire recently completed a $21 million private placement through the sale of 26,250 shares of Series A Convertible Preferred Stock at a purchase price of $800 per Preferred Share. The proceeds from the financing will be used to support working capital, to fund a portion of the cash component of the proposed DCS acquisition, and other general corporate purposes.
Aspire also recently secured a commitment letter from a leading financial institution for up to $22.5 million to finance the remainder of the DCS purchase.
Diversification into Automotive Supply
In a development that may come as a surprise to investors, Aspire recently announced plans to acquire DCS, a designer and manufacturer of automotive driver control systems, for $30 million. While the transition is outside the company’s core strategy, management has indicated that the target business is cash flow positive, with the potential to provide a more stable source of revenue. When viewed from that angle, the acquisition appears to be positioned as a way to support Aspire’s long-term pharmaceutical plans by helping to fund operations and reduce reliance on external capital.
DCS is a tier-one supplier specializing in vehicle electrification, safety, and human-machine interface (HMI) systems. Based on the announced LOI from the company, for the fiscal year ended December 31, 2025 (unaudited), DCS generated revenue of more than $200 million, net income of more than $17 million, and adjusted EBITDA of more than $22 million.
This type of diversification is not typical for early-stage biotech companies, which are generally more focused on drug development. While the transaction could provide meaningful near-term financial support, it does add complexity to the company’s strategic focus and capital allocation. For now, Aspire has indicated that it intends to continue pursuing its biopharma strategy alongside this potential acquisition, making for an important development for investors to monitor as the story evolves.
What Investors Should Look For
Looking ahead, there are several key developments that investors should watch out for that could play a role in shaping Aspire’s trajectory:
- Additional clinical data from its sublingual aspirin program, with another trial planned for mid-2026
- Progress along the 505(b)(2) pathway, with a potential regulatory filing anticipated in the second half of 2026
- Expansion of the pipeline into additional reformulated drugs, including treatments for anxiety, blood clot prevention, nausea, and motion sickness, in markets that total upwards of $100 billion
- Commercial traction for Buzz Bomb, including evidence of repeat demand and broader distribution
- Partnership or licensing agreements, particularly on the pharmaceutical side
- Completion of the DCS Purchase
Conclusion
Aspire Biopharma is not following the traditional biotech model. By focusing on drug delivery rather than drug discovery, and pairing that effort with early commercial initiatives, the company is attempting to build both a platform and a business simultaneously.
The early clinical data around its sublingual aspirin program provides an initial indication that the approach may have merit, particularly in applications where speed of action is critical. At the same time, the company is working to expand its technology across a broader pipeline of reformulated drugs, while using products like Buzz Bomb to begin establishing commercial traction.
The recent move to acquire DCS adds another layer to the story and introduces both potential financial support along with strategic complexity. If successful, this approach could provide Aspire with a more flexible operating model than a traditional early-stage biotech company, though it also places greater importance on execution across multiple fronts.
Ultimately, Aspire represents a different kind of early-stage opportunity that combines elements of platform innovation, regulatory strategy, and commercial execution. As the company advances its pipeline and begins to test its commercial strategy, the key question will be whether its delivery technology can demonstrate advantages that are not only scientifically valid but meaningful in real-world use.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.