By John Vandermosten, CFA
NYSE:AZTR
READ THE FULL AZTR RESEARCH REPORT
Azitra, Inc. (NYSE:AZTR) reported full year 2025 financial and operational results in a February 27thpress release and provided a business update. Since our previous quarterly dispatch in November, Azitra has announced a new, world-class cancer center site for the ATR-04 program, attended several investor and partnering conferences, and raised additional capital. The company also raised the possibility of entering into the cosmeceutical space to address fine lines and wrinkles in its latest presentation. Other highlights for 2025 are the presentation of ATR-04 data at ASCO, the announcement of preclinical data from the ATR-01 program and a report of safety data from the ATR-12 Phase Ib trial.
Operational and Financial Results
Azitra reported 2025 results in a press release and Form 10-K filing with the SEC on February 27th. For the year ending December 31st, 2025 and versus the prior year’s comparable period, no revenues were reported. Net loss for the twelve-month period totaled ($11.0) million or ($2.25) per share.[1] Operating expenses were essentially flat year over year as slightly lower general and administrative (G&A) expenses were offset by slightly higher research and development (R&D) expenses. Below, we detail 2025 financial results compared to the prior year period:
- No revenues were recognized compared with $8,000 related to the Bayer Joint Development Agreement;
- Research and development expenses increased by 2% to $4.8 million from $4.7 million stemming from greater clinical trial costs related to the ATR-04 and ATR-01 programs. Higher laboratory supplies were partially offset by lower payroll and benefits, reduced consultant expenditures and a fall in other costs;
- General & Administrative expenses totaled $6.1 million, down 2% from $6.3 million due to a reduction in legal fees, salaries and benefits, listing costs, insurance expense and other overhead. These declines were partially offset by an increase in the use of business consultants, public relations expenditures, software and equipment and accounting costs;
- Net interest income was $63,000 compared to $11,000 with the prior year dominated by interest income related to a higher cash balance;
- Other expense was $43,000 compared with income of $2.0 million;
- Net loss was ($11.0) million or ($2.25) per share vs. ($9.0) million or ($15.78) per share;[2]
As of December 31st, 2025, cash recognized on the balance sheet totaled $2.1 million. This compares with the $4.6 million balance in cash at the end of 2024. There is no debt. Cash burn for 2025 was $11.4 million, slightly higher than the $10.6 million consumed in the prior year period. Cash from financing was $8.9 million representing proceeds from public offerings, a private placement and the equity line of credit with Alumni Capital. This was partially offset by principal payments on finance leases. We expect that Azitra will regularly access the equity line to continue funding its pipeline activities. Post year end, Azitra raised $0.2 million with the equity line of credit.
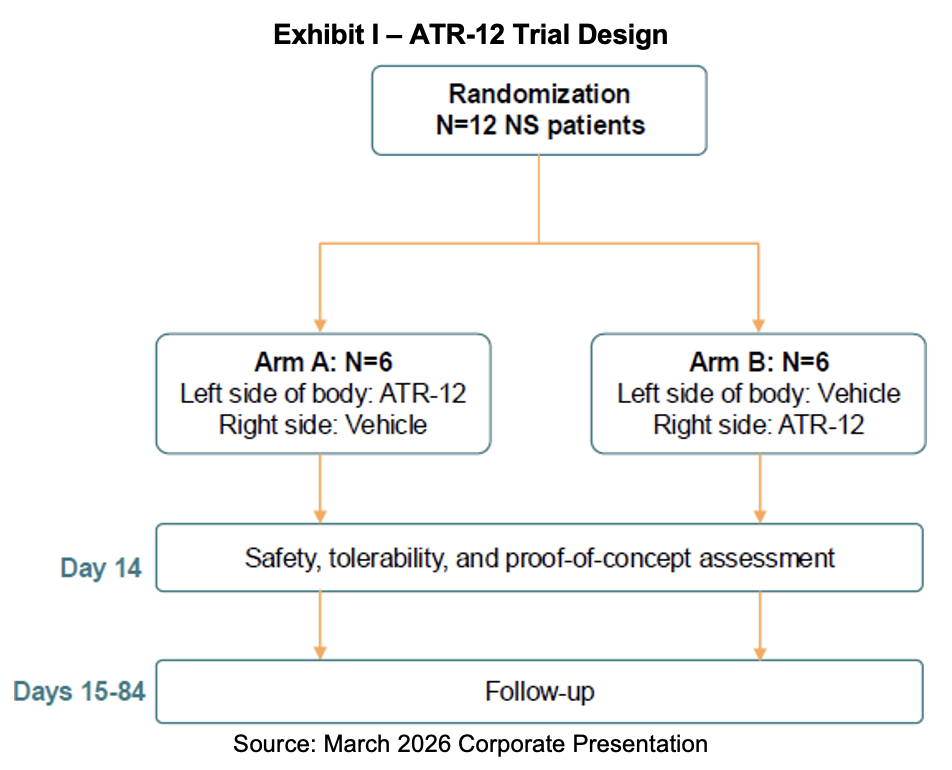
ATR-12 Clinical Trials
Azitra received investigational new drug (IND) clearance for its Netherton syndrome candidate, ATR-12, in 2023. The company has since launched a Phase Ib clinical trial under the identifier NCT06137157 and expects to enroll 12 adult patients. Primary endpoints will examine safety and tolerability while secondary and exploratory endpoints will assess efficacy signals and biomarkers. ATR-12 will be topically administered twice daily. Clinical sites at Yale University and Stanford University have been established. As of September 30th, 2025, six patients were enrolled in the ATR-12 study; no update on the number has been provided as of March 2026.
ATR-12 Trial Details
The study will apply ATR-12 to lesions on one side of a subject’s body and apply the vehicle to the other. Application of ATR-12 and the vehicle will be performed twice daily for two weeks. Patients will be randomized to receive ATR-12 on either the right or left side. Initially, the FDA required that patients be dosed one at a time and sequentially to provide an initial safety profile before allowing parallel enrollment; however, this restriction was lifted last June. The study requires that the young patients stay at the location for two weeks for treatment. The trial’s primary endpoint is adverse events at 84 days as well as quantifying and qualifying incidence, severity, seriousness and relatedness of adverse events. Secondary endpoints include investigators’ and patients’ global assessment of severity, concentration of recombinant human lymphoepithelial Kazal-type related inhibitor (rhLEKTI) in the plasma and on the skin following topical application. Biomarkers will be evaluated including KLK5, KLK7, IL-36, TARC/CCL17, trypsin-like activity and chymotrypsin-like activity.
June 2025 Interim Update
An interim Phase Ib update reported safety data. A June 17thpress release reported no severe or serious adverse events. Application site reactions have been transient and self-resolving and included mild to moderate localized itch, redness, and a burning sensation with application. Such reactions have been observed bilaterally, suggesting this is not a drug effect. The trial remains blinded, and data regarding the effect of the drug is not yet available.
ATR-04
In late February 2026, Azitra announced that it had added MD Anderson Cancer Center in Houston, Texas as a clinical site for its Phase I/II trial evaluating ATR-04 in EGFR inhibitor-associated skin rash. Six sites are now listed on the clinicaltrials.gov website for the trial listed under NCT06830863 and include cancer centers and hospitals in Arkansas, Connecticut, New York, Ohio and Virginia. Enrollment for the ATR-04 trial has been slow due to oncodermatologists working with oncologists to prophylactically administer antibiotics and steroids. The administration of antibiotics and steroids are exclusion criteria for enrollment in the trial. There is a strong desire by physicians to reduce the use of antibiotics to avoid broad resistance to these antibiotics. Physicians also recognize the risk of topical steroid use which can cause thinning of the skin, precipitate other types of rash and increase the risk of infection. Azitra will communicate the benefits of ATR-04 more broadly throughout the oncology care team at the sites where it is running the trial. Management has also opened the MD Anderson site which is more attuned to employing new modalities. We expect that these changes will improve the enrollment rate.
ATR-01 Preclinical Data
On October 20th, Azitra announced its upcoming presentation of ATR-01 data at the BIO-Europe conference. ATR-01 is a filaggrin-secreting strain of S. epidermidis for ichthyosis vulgaris. Ichthyosis[3] vulgaris is an inherited skin disorder that is characterized by dry, scaly skin that is similar in appearance to fish scales. The presentation emphasized that ATR-01 offers a positive pharmacology profile across multiple preclinical models. In in vitro models, ATR-01 secreted functional filaggrin, as measured by keratin binding assays. Furthermore, in ex vivo human skin, it was found to deliver filaggrin through the stratum corneum, as was measured with fluorescence immunohistochemistry. In this model, ATR-01 delivered filaggrin below the skin barrier (p < 0.05). Finally, in an ex vivo damaged pig skin model, ATR-01 was shown to significantly reduce transepidermal water loss compared to vehicle control (p < 0.002). Together, these data demonstrate positive pharmacological activity and biodistribution. Azitra plans to conduct additional investigational new drug (IND)-enabling studies and submit an IND in 2026.
ATR-01 Cosmeceutical Derivatives
At the March BIO conference, COO Travis Whitfill introduced a new opportunity in cosmeceutical indications. The product is produced by inserting a gene fragment that encodes filaggrin into S. epidermidis to be later used in skin applications. The filaggrin is a byproduct of ATR-01 and may free amino acids and natural moisturizing factors that could improve the appearance of fine lines and wrinkles. One of the advantages of a cosmeceutical application is that the regulatory pathway is shorter and less costly. Additionally, there is substantial demand from cosmetics companies, especially in Asia, for new products that show benefits. This is an early-stage initiative, but merits further review as it evolves.
Milestones
- Share purchase agreement with Alumni Capital – April 2025
- Presentation of ATR-04 data and clinical plan at ASCO – May 30 to June 3, 2025
- Initial safety data from Phase Ib ATR-12 – June 2025
- First patient dosed with ATR-04 for EGFRi rash (Phase I/II) – August 2025
- NYSE American issues deficiency letter for equity below $4 million – October 2025
- Report of ATR-01 preclinical data – October 2025
- Pricing of $1.5 million private placement – November 2025
- Notice of Acceptance of Listing Standards Compliance Plan from NYSE American – December 2025
- Presentation at Biotech Showcase – January 2026
- Presentation at BIO Investment & Growth Summit – March 2026
- Special Meeting of Shareholders – March 6th, 2026
- ATR-04 topline announcement – Mid-year 2026
- Topline report for Phase Ib ATR-12 trial – 2H:26
- ATR-01 IND submission – 4Q:26

Summary
Azitra reported 2025 financial and operational results generating an operating loss of $11.0 million and cash burn of $11.4 million. The use of cash was funded by net cash from financing of $8.9 million from equity line of credit, private placement and public offering proceeds. Enrollment for the company’s trials has slowed; however, action has been taken through the addition of a new clinical trial site at MD Anderson and increased education for the oncology teams that guide patients through treatment. Beyond the work Azitra has done with its three candidates, members of its management team have met with investors and other stakeholders at multiple conferences. As we look ahead, we see several milestones including topline results from the two clinical programs and, potentially, an IND submission before year end. We update our price target to reflect existing and anticipated shares outstanding over the next year. The result of this calculation generates a target price of $1.70 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] We update per share amounts in this report to reflect the 15:100 reverse stock split that took place on August 21st, 2025.
[2] We use financial statement data as originally reported and apply a 1:6.66 reverse stock split ratio for periods prior to August 21st, 2025. Prior year numbers in our reporting may not match Azitra’s current period comparisons.
[3] The root of the word Ichthys is Greek for fish (ἰχθύς).