By John Vandermosten, CFA
NASDAQ: BLRX
READ THE FULL BLRX RESEARCH REPORT
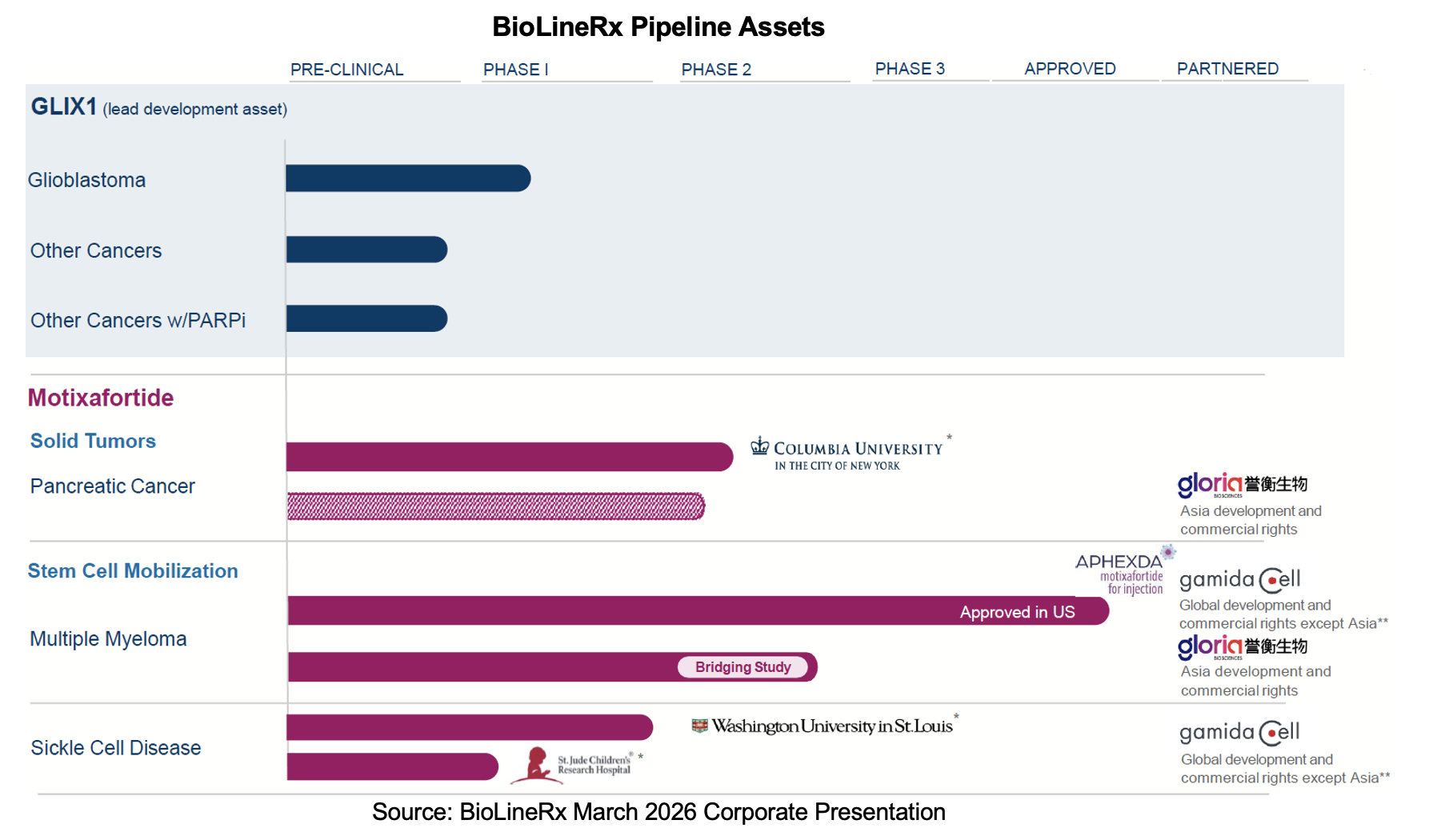
BioLineRx Ltd. (NASDAQ: BLRX) reported 2025 financial and operational results in a March 23rd, 2026, press release. For the year, it produced license revenues of $1.2 million and a net operating loss of $10.3 million. The joint venture (JV) with Hemispherian, called Tetragon Biosciences, expects to begin screening patients by the end of this month for the glioblastoma trial evaluating GLIX1. The Phase I trial seeks to establish the safety, recommended dose, and proof-of-concept for the drug. In support of the GLIX1 program, BioLineRx secured a Notice of Allowance for a patent covering cancers that do not express cytidine deaminase beyond a certain threshold. Enrollment for Columbia’s CheMo4METPANC Phase 2b study continues, and management anticipates that an interim readout will occur later in 2026. Efforts to advance motixafortide in sickle cell disease (SCD) are in high gear with both a poster presentation last December and continued enrollment in a second Phase I study by St. Jude. In the Asian sphere of influence, Gloria Biosciences launched its stem cell mobilization (SCM) trial in November 2025 and is expected to report data from the study approximately 18 months later.

2025 Operational and Financial Results
BioLineRx reported 2025 sales of $1.2 million, producing a net loss from operations of $2.0 million or $0.00 per share. Non-operating income offset $8.1 million of the operational loss. Absent this non-cash item, net loss would have been $10.1 million, still $0.00 per share. The results were announced in a press release on March 23rd, 2026, followed by a conference call with management and the filing of Form 20-F providing additional information.
Below we summarize financial results for the twelve-month period ended December 31st, 2025, compared to the prior year period:
- Total and license revenues were $1.2 million from the sale of Aphexda compared to $28.9 million, with the latter related to the out-licensing transaction with Gloria and direct commercial sales. Ayrmid’s Aphexda product sales for 2025 were $6.7 million, which implies a royalty rate of about 18%;
- Cost of revenues was $230,000, which represents license fees and royalty pass-throughs to Biokine as a royalty on motixafortide revenues, vs. $9.3 million. Prior year cost of revenues included amounts for license fees, direct costs related to license revenues, amortization of an intangible asset, and cost of product sales;
- Research and development expenses totaled $8.1 million, down 12% from $9.1 million, with the decline attributable to lower expenses related to motixafortide due to the out-licensing of rights to Ayrmid and a reduction in compensation arising from lower headcount. These amounts were partially offset by an increase in expenses related to the GLIX1 program;
- Sales and marketing expenses were $0.0 vs. $23.6 million due to the shutdown of U.S. commercial operations in the fourth quarter of 2024 following the Ayrmid out-licensing transaction;
- General and administrative (G&A) expenses were $3.1 million, down 50% from $6.3 million primarily due to the reversal of a provision for doubtful accounts following receipt of an overdue milestone payment from Gloria, as well as a decrease in payroll and share-based compensation related to lower headcount and an overall reduction in a number of general and administrative expenses;
- Non-operating income was $8.1 million vs. $18.4 million, predominantly reflecting changes in fair-value adjustments of warrant liabilities on the balance sheet;
- Net financial income amounted to $184,000, reflecting interest income exceeding interest expense;
- Net loss was $2.0 million compared to $9.2 million, or $0.00 and $0.01 per share, respectively.
Cash, equivalents, and short-term bank deposits as of December 31st, 2025, totaled $20.9 million, up from the year end 2024 balance of $19.6 million. Cash burn for 2025 was $8.1 million, and net cash from financing was $8.9 million. $2.4 million in cash from the Gloria milestone was received in June. Financing cash contributions came from issuance of share capital and warrants, as well as net proceeds from the ATM agreement with H.C. Wainwright. The ATM raised $5.0 million in 2025. Following the end of the reporting period, an additional 825,010 ADSs were sold under the program, generating net proceeds of $9.2 million. As of year-end 2025, loans were carried at $8.9 million on the balance sheet. This was partially offset by loan repayment over the 12-month period of $4.5 million. The term loan is expected to be fully repaid by the end of 2027. Management forecasts sufficient cash to support operations until 1H:27.
BioLineRx also reported a favorable outcome from the binding arbitration related to Biokine’s complaint related to compensatory damages related to the transfer of motixafortide licenses. In February 2026, the arbitrator issued a final award in favor of BioLineRx, denying the claim and awarding BioLineRx expenses, including legal fees. Given the jurisdiction of the claim in Israel, we estimate this will amount to a few hundred thousand dollars.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.