By David Bautz, PhD
NASDAQ: COCP
READ THE FULL COCP RESEARCH REPORT
Business Update
Fast Track Designation for CDI-988
On April 2, 2026, Cocrystal Pharma, Inc. (NASDAQ: COCP) announced that the U.S. Food and Drug Administration (FDA) has granted Fast Track designation to CDI-988, the company’s pan-viral protease inhibitor targeting 3CL viral proteases and being developed as a treatment for norovirus and coronavirus infections. Fast Track designation is designed to facilitate and accelerate the development of novel therapeutics for serious conditions that address unmet medical needs. It allows companies to have early and frequent communication with the FDA during the entire development process as well as a rolling review of a New Drug Application (NDA). In addition, it may qualify a product for Priority Review when the NDA is submitted.
In March 2026, the company announced that the first subject was dosed in the Phase 1b trial of CDI-988 to evaluate the drug as both a preventive and treatment for norovirus infection. It is a randomized, double blind, placebo controlled study that will enroll up to 40 healthy subjects ages 18-49. All participants in the study will be infected with the norovirus GII.2 (Snow Mountain Virus) strain (NCT07198139).
The initial cohort of subjects is to assess the infectivity rate of the challenge inoculum, GII.2 (Snow Mountain Virus). Subsequent cohorts will be orally administered CDI-988 or placebo. The primary endpoint of the trial is the efficacy of CDI-988 versus placebo in reducing the incidence of clinical symptoms. Secondary endpoints being evaluated include reduction in viral shedding and disease severity, along with safety and pharmacokinetic profile. The challenge study is designed to serve as a surrogate for clinical efficacy data.
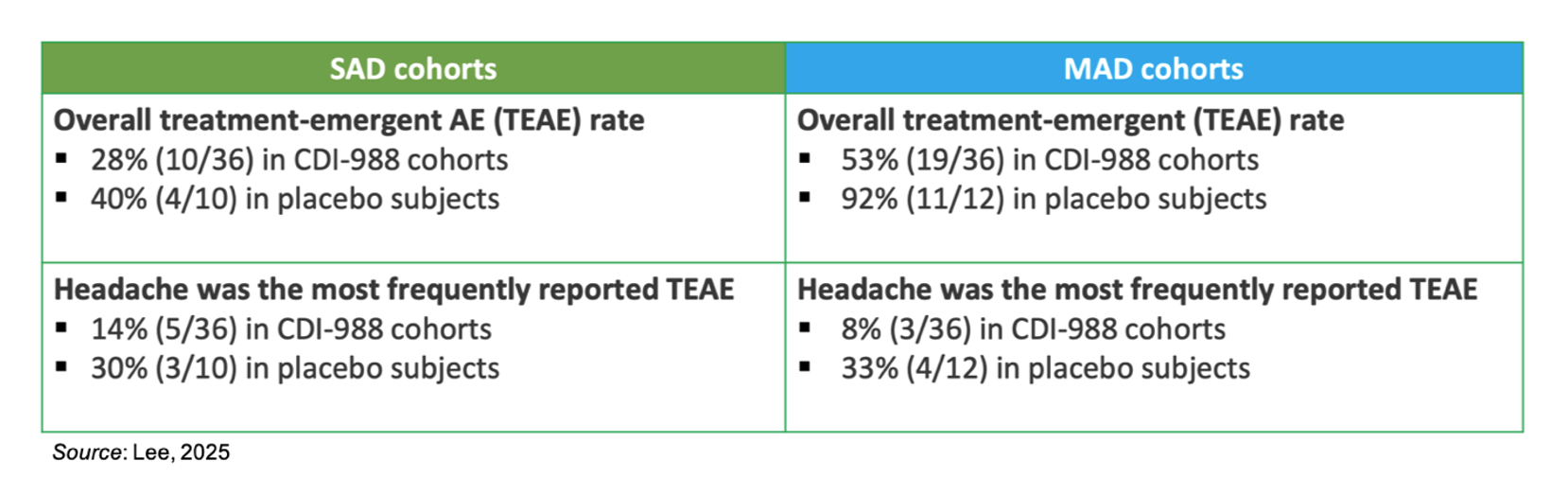
CDI-988 was previously tested in a Phase 1a study that enrolled 46 (N=36 drug; N=10 placebo) individuals into the single ascending dose (SAD) cohort and 48 (N=36 drug; N=12 placebo) individuals into the multiple ascending dose (MAD) cohort. The SAD results showed that all doses (100 mg to 1200 mg) were well tolerated, there were no reports of serious adverse events, no clinically relevant ECG changes, no clinically significant pathology results, and no discontinuations from the study or use of the study drug. Similar results were seen in the MAD cohort, as all doses (200 mg to 1200 mg) were well tolerated, there were no reports of serious adverse events, no clinically relevant ECG changes, and no clinically significant pathology results. There was one discontinuation from the study and study drug due to Grade 2 diarrhea for an individual in the 1200 mg BID Fed group. This discontinuation was deemed probably related to study drug. CDI-988 shows a strong food effect (5-fold higher plasma exposure when administered after a high-fat meal), thus that may have contributed to the Grade 2 diarrhea for that individual.
The topline safety data are summarized in the table below. Headache was the most frequently reported treatment emergent adverse event (TEAE) and overall the placebo groups had a higher frequency of TEAEs than the CDI-988 groups.
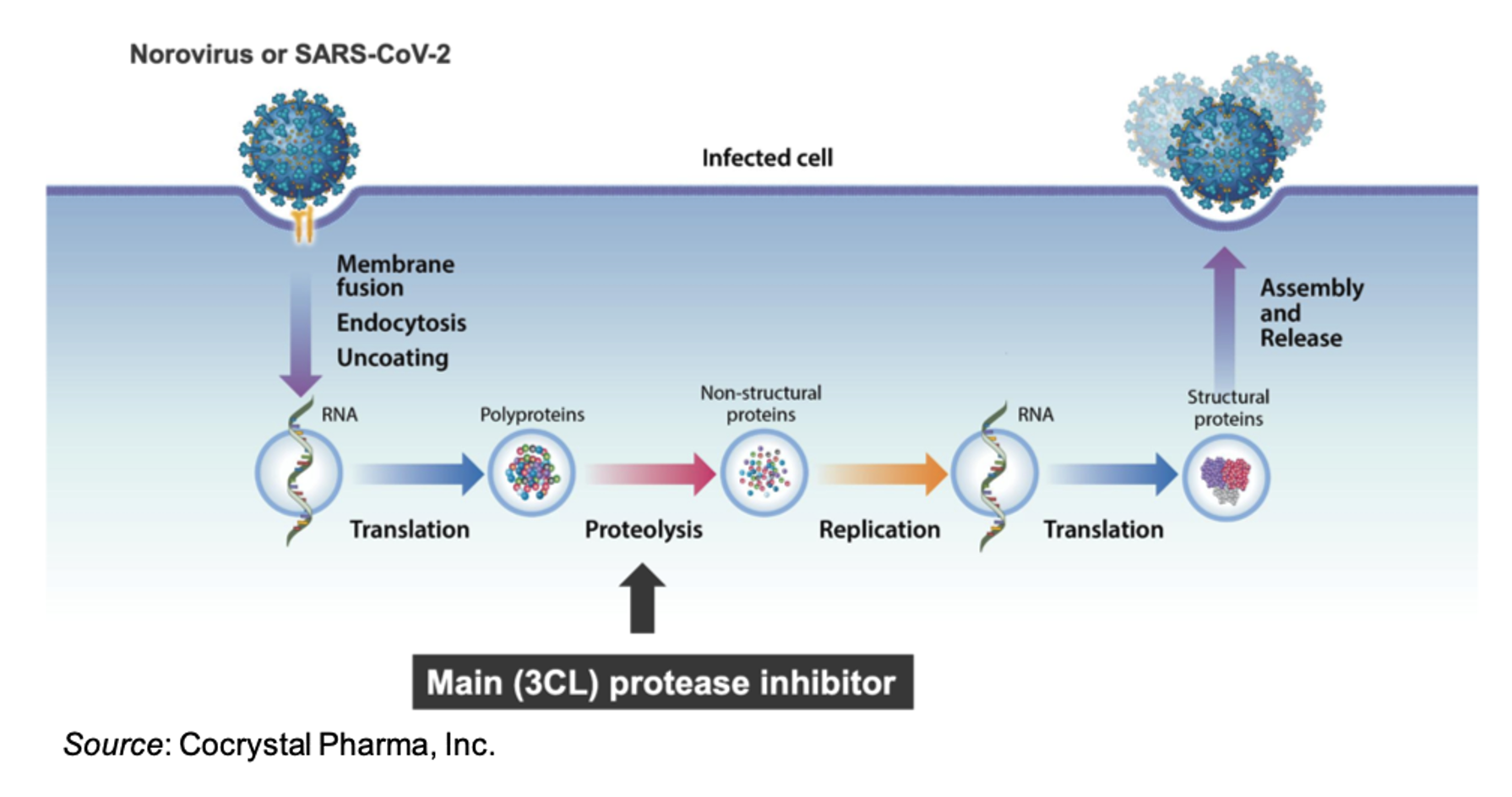
There are no approved therapies for norovirus infection, which is the most common cause of acute gastroenteritis. According to the Centers for Disease Control (CDC), there are an estimated 685 million cases and 200,000 deaths caused by norovirus infection each year worldwide. In the U.S., norovirus infection causes over 2 million outpatient clinical visits annually and approximately 100,000 hospitalizations. Norovirus prevention and/or therapy is particularly relevant for the military, where a pathogen of that nature can result in significant operational risks. There are multiple military installations, for instance, on navy vessels, where norovirus could spread rapidly and possibly hinder combat readiness. Due to this, work on CDI-988 may qualify for various military-sponsored grants or other funding mechanisms, however we are unaware of any specifics at this point.
CDI-988 was developed using Cocrystal’s proprietary drug discovery platform technology. It binds to a highly conserved region in the active site of noroviruses and coronaviruses 3CL viral proteases and exhibits pan-viral activity against pandemic norovirus and coronavirus strains.
Financial Update
On March 31, 2026, Cocrystal announced financial results for the year ending December 31, 2025. As expected, the company did not report any revenues in 2025. R&D expenses for 2025 were $5.1 million compared to $12.5 million in 2024. The decrease was primarily due to the winddown of the Phase 2a influenza study and reduction in employee-related expenses. G&A expense in 2025 were $4.0 million compared to $5.3 million in 2024. The decrease was primarily due to a reduction in compensation, insurance, and corporate expenses.
Cocrystal exited 2025 with approximately $7.7 million in cash and cash equivalents. As of March 24, 2026, the company had approximately 13.8 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of 21.6 million.
Conclusion
Fast Track designation for CDI-988 is an important milestone for Cocrystal as it allows the company to have more frequent interactions with the FDA as the compound advances through clinical trials. We look forward to updates regarding the Phase 1b study of CDI-988 as the year progresses, along with updates regarding the company’s influenza program, for which another Phase 2a trial is currently being planned. With no changes to our model, our valuation remains at $8 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.