By John Vandermosten, CFA
NASDAQ:CRDF
READ THE FULL CRDF RESEARCH REPORT
2025 Financial Results
Cardiff (NASDAQ:CRDF) reported 2025 financial and operational results in a press release and Form 10-K filing with the SEC on February 24th, 2026. Later that day, CEO Dr. Mani Mohindru participated in Oppenheimer’s Healthcare Conference, providing the company’s latest status. For the twelve-month period ending December 31st, 2025, revenues of $593,000 were reported, and operational expense of $49.0 million was recognized. Loss per share was $0.69. Operational expenses rose less than 1% as higher General and Administrative (G&A) expenses were offset by lower research and development (R&D) expenses. For the year ending December 31st, 2025, and versus the same prior year period:
- Revenues of $593,000 compared to $683,000 and represent Cardiff’s sales based and usage-based royalties on assets unrelated to onvansertib;
- Research and development expenses totaled $35.3 million, down 4% from $36.9 million, attributable to a reduction in clinical trial expenses and a decrease in preclinical activities for the CRDF-004 clinical trial. The decrease was partially offset by higher stock compensation from new grants;
- Selling, General & Administrative expenses were $14.2 million, up 14% from $12.5 million. Increases relate to higher professional fees, specifically strategic advisory services and an increase in patent fees. Salaries and staff costs related to an employee severance agreement also contributed to the increase;
- Net interest income of $3.1 million was down compared with prior period levels due to lower cash levels and other income of $5,000 compared to other expense of $39,000;
- Net loss was $45.9 million vs. a net loss of $45.4 million or $0.69 and $0.95 per share, respectively.
As of December 31st, 2025, cash totaled $58.3 million. This amount compares to the $91.7 million balance in cash held at the end of 2024. Cash burn for 2025 was $38.0 million versus $37.8 million for 2024. Cardiff’s cash is expected to support operating activities until 1Q:27.
Starting a New Chapter
On January 27th, 2026, Cardiff Oncology, Inc. announced that its CEO, Dr. Mark Erlander, and CFO, James Levine, stepped down from their roles. The new interim chief executive officer is Dr. Mani Mohindru, who has been on Cardiff’s board since 2021; the lead finance role was assumed by Brigitte Lindsay, who was appointed as Chief Accounting Officer. Along with the appointment of new executives, the company presented select data from its Phase II trial in onvansertib in first line RAS-mutated metastatic colorectal cancer (mCRC). Results from the CRDF-004 trial supported the selection of the 30 mg onvansertib dose for the anticipated Phase III program in first-line RAS-mutated mCRC. Results also validate previously reported results from the Phase II second-line mCRC trial, where bevacizumab-naïve patients demonstrated a clinical benefit from the use of onvansertib. Along with the data readout, company management explained the data and answered analyst questions in a conference call.
Milestones
- Roger Sidhu appointed CMO – June 2025
- Interim update on CRDF-004 trial – July 2025
- Poster presentation of investigator-sponsored data in CMML at ASH – December 2025
- Presentation at Sidoti’s investor conference – December 2025
- Mani Mohindru appointed interim CEO – January 2026
- Topline release from CRDF-004 – January 27th, 2026
- Nerviano files notice alleging material breach of onvansertib licensing agreement – February 2026
- Investor Presentation at Oppenheimer Healthcare Life Sciences Conference – February 2026
- Presentations at TD Cowen, Barclays & Leerink healthcare conferences – March 2026
- Meetings with FDA for Phase III trial design – 1H:26
- Additional data release from CRDF-004 – 2Q:26
- Launch of Phase III onvansertib trial (CRDF-005) - 2026
Company Pipeline
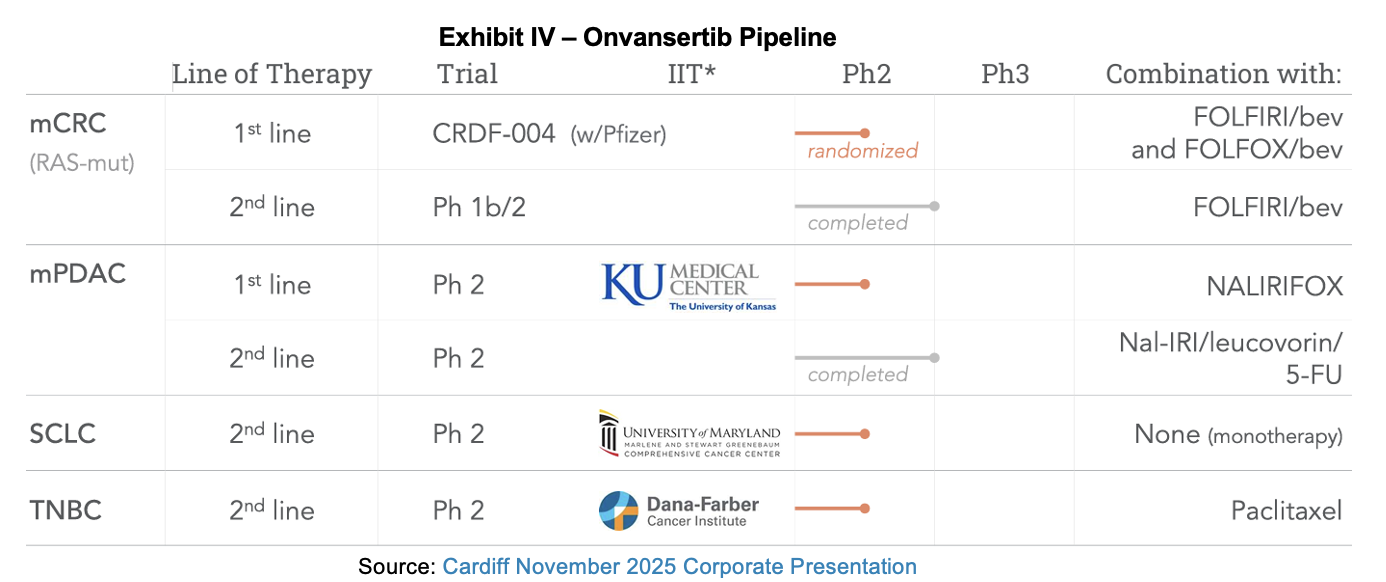
Summary
Cardiff reported full year 2025 financial results just a few weeks after the presentation of topline data from its Phase II CRDF-004 study and a change in senior management. Operating expense and cash burn were in-line with expectations. The CRDF-004 study results support the use of the 30 mg dose of onvansertib in the anticipated Phase III trial that will be designed with the FDA in the coming months. Dr. Mohindru has taken the reins of the company and will continue in an interim capacity while the board conducts a search to identify a new CEO and CFO.
Data from the CRDF-004 trial highlighted a favorable ORR and hazard ratio for the 30 mg onvansertib arm. Median PFS has not yet been reached. An important early indicator, PFS at six months, was more than five percentage points better at 94.1%. While the data were positive, the trial is still ongoing, and there is other data that will further clarify the safety and efficacy of the regimen. Management has indicated that they expect to move forward with the Phase III study using onvansertib + FOLFIRI/bev to the combined SoC arm following a meeting with the FDA.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.