By John Vandermosten, CFA
NASDAQ: CRDF
Key Opinion Leader Event Places Onvansertib in Context
Cardiff Oncology, Inc. (NASDAQ: CRDF) held a key opinion leader (KOL) event on March 25th, 2026, coinciding with National Colorectal Cancer Awareness Month. The event featured two luminaries in the oncology space who discussed the emerging treatment landscape in first-line Rat Sarcoma Virus (RAS)-mutated metastatic colorectal cancer (mCRC). The participants included KOLs Scott Kopetz, M.D., Ph.D., FACP, and Heinz-Josef Lenz, M.D., who joined Mani Mohindru, PhD, Cardiff’s interim Chief Executive Officer, for the hour-long webinar. The event began with an introduction for each of the guests.
Scott Kopetz, M.D., Ph.D., FACP, is a Professor in the Department of Gastrointestinal Medical Oncology at The University of Texas MD Anderson Cancer Center and an internationally recognized leader in colorectal cancer research and translational oncology. Dr. Kopetz’s work has helped establish new treatment approaches for molecularly defined colorectal cancers, including therapies targeting BRAF-mutated metastatic disease. He serves in multiple national leadership roles supporting gastrointestinal cancer research and clinical trial development and has led numerous Phase I–III clinical studies focused on improving outcomes for patients with gastrointestinal (GI) malignancies. His research integrates molecular profiling and translational science to advance precision medicine strategies and overcome treatment resistance in colorectal cancer.
Heinz-Josef Lenz, M.D., is a University Professor of Medicine, Population and Public Health Sciences and Cancer Biology; Professor of Medicine and Preventive Medicine of the University of Southern California (USC). He serves as Co-Leader of the Gastrointestinal Cancers Program and Co-Director of the USC Center for Cancer Drug Development. Dr. Lenz’s research focuses on molecular mechanisms of cancer development, drug resistance and biomarker-driven treatment approaches in gastrointestinal cancers, including colorectal cancer. He has authored numerous peer-reviewed publications and holds leadership roles across national oncology research initiatives, including service on National Cancer Institute committees and cooperative clinical trial groups guiding translational and clinical research in GI oncology.
The event began by highlighting the slow progress in the development of new treatments for RAS-mutated[1] CRC in recent years and reviewing Cardiff’s interim data shared in January. Our report entitled New CEO Presents Phase II Readout summarizes these results. Dr. Mohindru highlighted the deeper objective responses in patients on the 30 mg dose of onvansertib compared with the standard of care (SoC) arms. A feature of the safety profile is the similar toxicity profile between onvansertib in combination with chemotherapy and bevacizumab compared to chemotherapy and bevacizumab. The interim CEO reiterated the company’s plans to advance its drug into a Phase III pivotal study after consulting with the FDA on the trial design.
Following the presentation of the recently published data, the event turned towards questions for the KOLs. The first topic elaborated on an oncologist’s menu of options for frontline patients who present with metastatic colorectal disease. Dr. Lenz responded by noting the importance of understanding the molecular characteristics of the tumor with respect to KRAS, NRAS and BRAF status.[2] If the tumor is microsatellite instability-high (MSI-high), the tumor is responsive to checkpoint inhibitors because it presents many mutations that the immune system recognizes. If the patient harbors the KRAS mutation, there are no targeted therapies specifically for RAS mutations. Treatment for BRAF-V600E-mutated mCRC patients has recently changed and includes encorafenib and cetuximab either with or without chemotherapy.
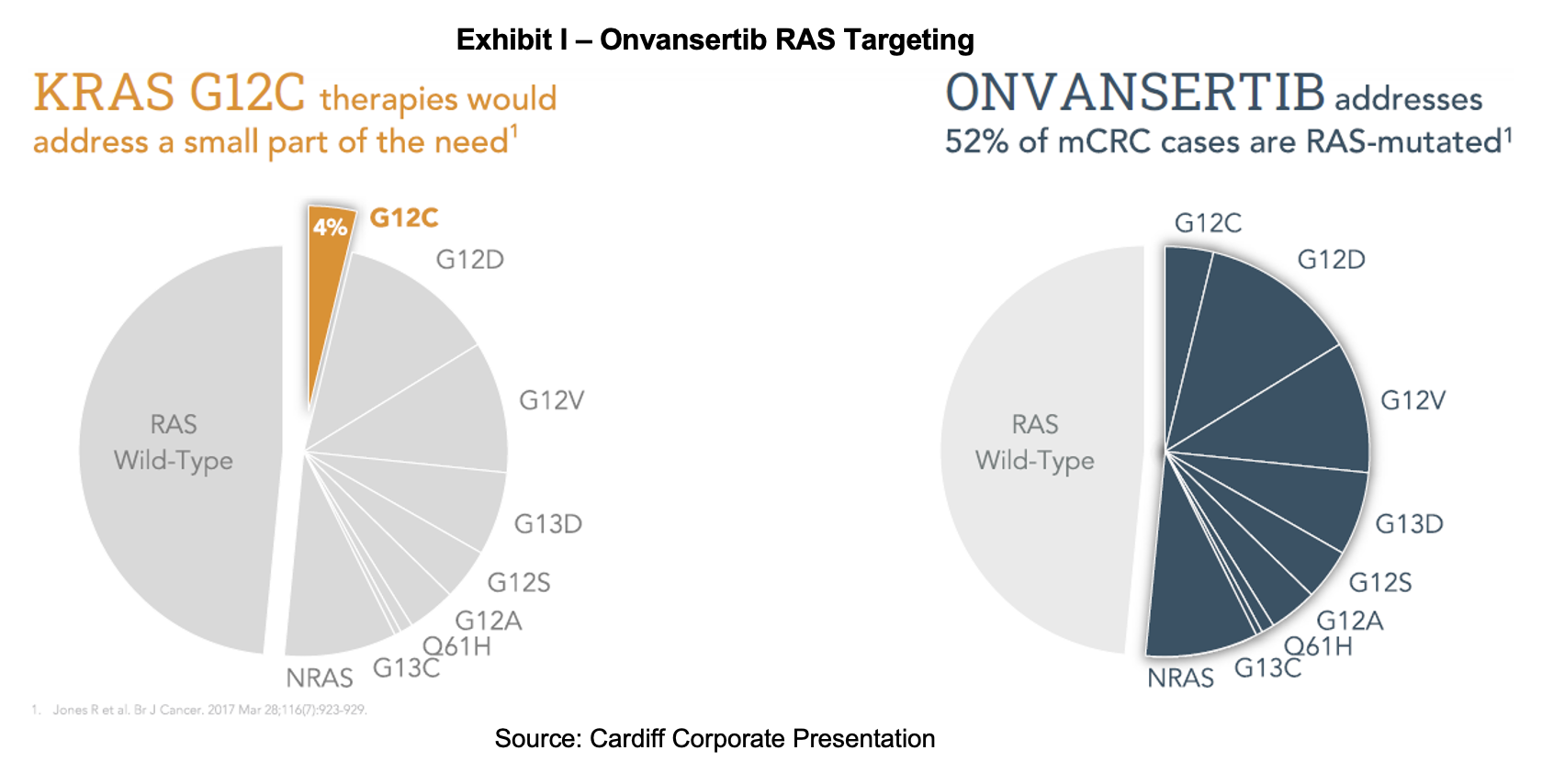
The KOLs also discussed treatment options for patients with the G12C genetic alteration. In the last few years, combination therapies using sotorasib/panitumumab or adagrasib/cetuximab have been approved for this small group in later-line settings. The KOLs also noted the failure of cytotoxic chemotherapy to work in later lines of therapy. This is attributable to the tumor’s evolution to become resistant to treatment. Bevacizumab may develop a similar resistance, highlighting the importance of using it in first-line therapy with onvansertib.
Both of the KOLs have experience with onvansertib, recognize its favorable toxicity profile and emphasize the importance of a regimen that is tolerable for patients. Tolerability is especially important in earlier lines of therapy and for extended periods of treatment. The experts also identified several of the agent’s attractive features, including its synergistic role in combinations and the role of the PLK1 inhibitor class in DNA damage repair.[3]
The conversation shifted to the role of angiogenesis in mCRC (See our initiation for a discussion). Angiogenesis supplies tumors with new blood vessels, which helps the malignancies grow, survive and spread. In mCRC, this process is especially important because it supports both primary tumor expansion and metastatic lesions by improving oxygen and nutrient delivery and by enabling tumor cells to enter the bloodstream. Vascular Endothelial Growth Factor (VEGF)-driven signaling is one of the central angiogenic pathways in CRC. Importantly, onvansertib can disrupt the hypoxia-HIF signaling axis by helping regulate hypoxic responses and angiogenic signaling. This creates a hypoxia-PLK1-angiogenesis feedback loop in which HIF activation upregulates PLK1. When PLK1 is inhibited, anti-angiogenic therapies such as bevacizumab can be effective by further disrupting this survival pathway. Onvansertib may directly reduce HIF1 levels creating a synergy with bevacizumab which neutralizes VEGF.
The conversation shifted to available therapies for specific mCRC patient groups. As a whole, patients with a RAS mutation do not have approved therapies to address their molecularly distinct cancer and they must receive chemotherapy with bevacizumab. Patients may also enter into clinical trials for experimental immunotherapy treatment.
In the last portion of the call, the host and KOL participants answered attendee questions. The first question related to the durability of onvansertib compared to both standard of care and other assets in development for frontline colorectal cancer. Dr. Lenz responded identifying results achieved in previous onvansertib trials as relevant benchmarks to exceed. He cited onvansertib’s 12-month duration of response (DoR) and 15-month progression-free survival (PFS). These were reported for second-line treatment patients in the Phase II trial bevacizumab-naïve subgroup. In the CRDF-004 study, DoR and PFS have not yet been reached. The KOLs also acknowledged the attractive hazard ratio below 0.5. However, there are few studies available that provide comparative data.
Another question asked about preferences for FOLFOX vs. FOLFIRI. The consensus appeared to be that FOLFIRI is more favorable because it has fewer restrictions and toxicities and can serve a larger population.
A final query was the proportion of mCRC, RAS-mutant tumors that are observed in the KOL’s practices. Dr. Kopetz believes it is about 50%, which was confirmed by Dr. Lenz. Of this population, about 15% is targetable with allele-specific treatments either in development or approved such as the G12C inhibitors sotorasib (Lumakras) and adagrasib (Krazati). This is in contrast to onvansertib, which is mutation agnostic.
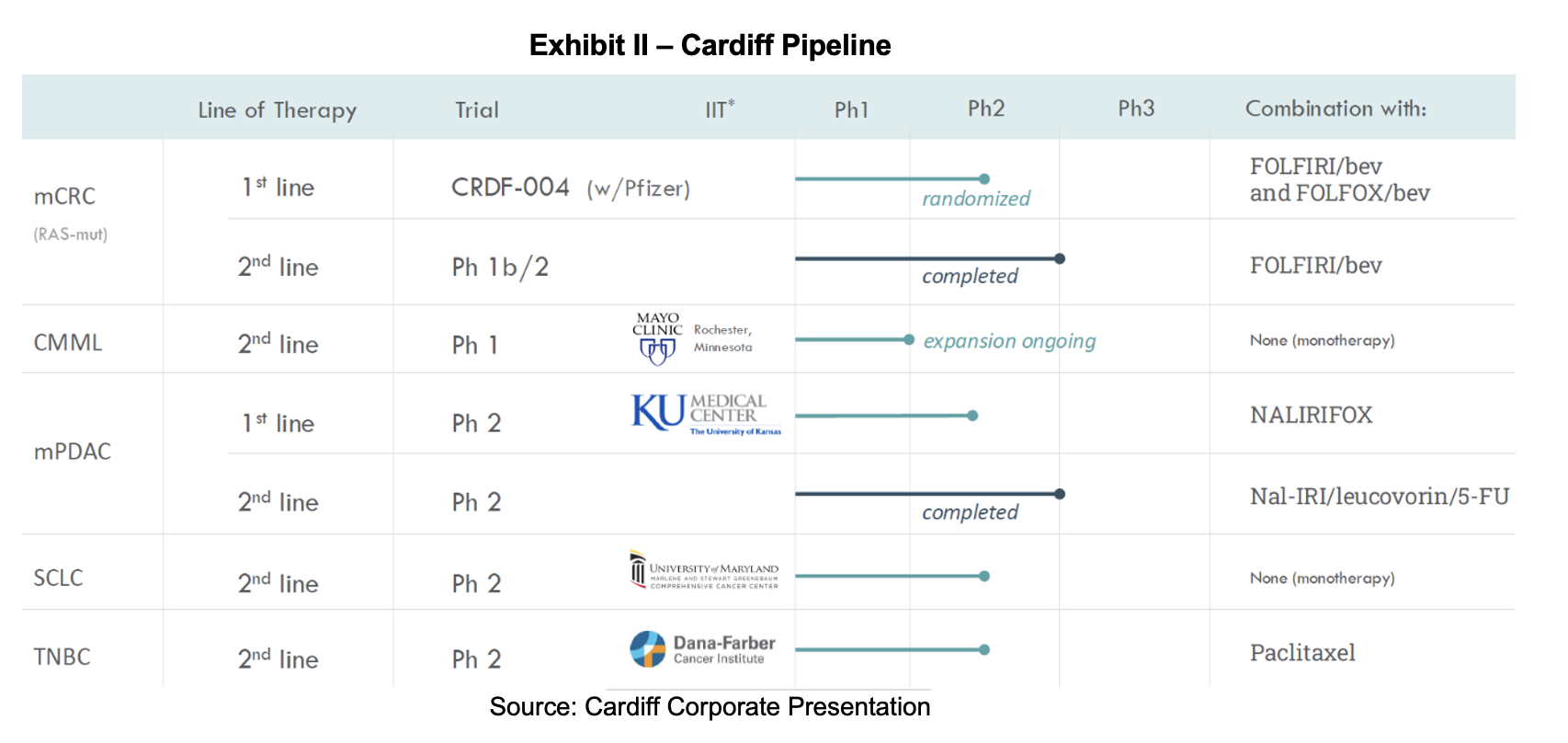
Company Pipeline
Summary
Cardiff and its interim CEO held a KOL event featuring two physicians who served as principal investigators for the onvansertib clinical trials. Their intimate knowledge of both the indication and the drug provided valuable experience that informed their presentations and responses. Outside of treatments serving small populations with genetic alterations, such as G12C, there has been little advancement in mCRC care. A pan-RAS therapy such as onvansertib could provide relief to patients with the RAS mutation and improve related survival metrics. Recent data shows that the 30 mg dose of onvansertib produced superior overall response rate and the PFS hazard ratio. While the median PFS had not yet been reached, the latest data suggest onvansertib generated superior results. Stakeholders who joined the call were interested in the benchmarks that will be used to measure survival performance in patients, the optimal chemotherapy to use, and the proportion of patients that each KOLs sees with a RAS mutation. We expect to see additional readouts of the CRDF-004 trial later this year, along with updates on FDA interactions and a potential start for the pivotal CRDF-005 study.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] RAS-mutated cancers are cancers driven by mutations in the RAS gene family, mainly KRAS, NRAS, and HRAS. These mutations allow the RAS protein to transmit constant growth signals and cells divide uncontrollably. RAS mutations are common in pancreatic, colorectal and lung cancers, though they can also appear in skin, ovarian, breast, and other tumors. More specifically, mutant RAS is present in nearly all pancreatic cancers, about half of colorectal cancers and about one-third to one-quarter of lung cancers, depending on the source and tumor subtype. RAS mutations are important because they often make cancers more aggressive and harder to treat. For many years, mutant RAS was considered difficult to target directly, which is why it has been such a major focus of cancer drug development.
[2] Kirsten Rat Sarcoma Virus Oncogene (KRAS) is found in 40% of colorectal cancers; these patients typically do not respond to EGFR-inhibitor therapies. Neuroblastoma Rat Sarcoma Virus Oncogene (NRAS) is a member of the same protein family as KRAS but is associated with a different set of primary malignancies. They are historically difficult to target directly. B-Raf Proto-Oncogene (BRAF) is a protein kinase that sits immediately downstream of the RAS proteins in the signaling chain and is found in about 10% of colorectal cancers. The most common BRAF mutation is V600E, which is generally associated with a poorer prognosis and requires specific aggressive treatment combinations.
[3] PLK1 has a dual role in the DNA damage response by promoting homologous recombination repair and blocking checkpoint signaling so cells can re-enter the cell cycle after damage is addressed. PLK1 inhibitors prevent repair mainly by blocking PLK1-dependent phosphorylation events that promote homologous recombination. Without PLK1 activity, cells have trouble recruiting and clearing key repair proteins so double-strand break repair is less efficient.