By David Bautz, PhD
NASDAQ:CVKD
READ THE FULL CVKD RESEARCH REPORT
Business Update
Acquires 12-LOX Inhibitor VLX-1005 for the Treatment of HIT
In December 2025, Cadrenal Therapeutics, Inc. (NASDAQ:CVKD) announced the acquisition of VLX-1005 and related 12-lipoxygenase (12-LOX) assets from Veralox Therapeutics. VLX-1005 is a first-in-class, highly selective small molecule inhibitor of 12-LOX for the treatment of heparin-induced thrombocytopenia (HIT). Two Phase 1 clinical trials in healthy volunteers have shown safety and tolerability across dosing, with no deaths or serious adverse events. A Phase 2 study in suspected HIT patients reported encouraging reductions in thromboembolic events, which is a clinically meaningful endpoint for regulators and clinicians. VLX-1005 holds Orphan Drug Designation and Fast Track status in the U.S. and Orphan Drug Designation in the E.U. This acquisition both strengthens Cadrenal’s pipeline with a late-stage asset and provides an opportunity to address another underserved therapeutic opportunity in the $40 billion global anticoagulation market.
Background on HIT
HIT is a severe, immune-mediated prothrombotic disorder triggered by exposure to heparin, an anticoagulant used widely in clinical practice (e.g., surgery, dialysis). It is estimated to occur in approximately 1 in 1500 hospital admissions (Dhakal et al., 2018). Data shows that certain variables can increase the risk of developing HIT, including cardiac surgery (Pishko et al., 2017) and exposure to unfractionated heparin vs. low molecular weight heparin (Warkentin et al., 1995), while a shorter exposure to heparin appears to decrease the risk of developing HIT (Smythe et al., 2007).
Diagnosis of HIT uses the “4Ts Score”, which is a pre-test scoring system that assesses the probability of HIT (Lo et al., 2006). It is calculated as a sum of points from four components: Thrombocytopenia, Timing of platelet count fall, Thrombosis or other sequelae, and oTher causes of thrombocytopenia. Laboratory diagnosis of HIT is divided into two steps: an immunoassay and a functional assay. The immunoassay examines for the presence of anti-platelet factor 4 (PF4)/heparin antibodies. If anti-PF4/heparin antibodies are identified, a functional assay is performed to determine if those antibodies are pathogenic. The 14C-serotonin release assay (SRA) is the “gold standard” functional assay and has both high sensitivity (~0.95) and specificity (~0.95) (Warkentin et al., 2015).
Management for patients suspected of suffering from HIT includes 1) the avoidance of heparin, indefinitely if possible, and 2) use of non-heparin anticoagulation. The duration of non-heparin anticoagulation will be dictated by the presence or absence of thrombosis. Preferred agents for non-heparin anticoagulation therapy include argatroban and bivalirudin, which can be administered IV, or danaparoid, which can be administered subcutaneously.
For patients who develop HIT, there are a number of potential negative outcomes, including thrombosis, bleeding, amputation, increased risk of hospital stay, and even death. Despite decades of research, no approved therapy directly targets the core immune and platelet activation mechanisms in HIT. Current anticoagulants decrease the risk of coagulation but do not modulate immune-mediated platelet activation, which leaves patients at persistent risk of thrombosis.
VLX-1005 for HIT
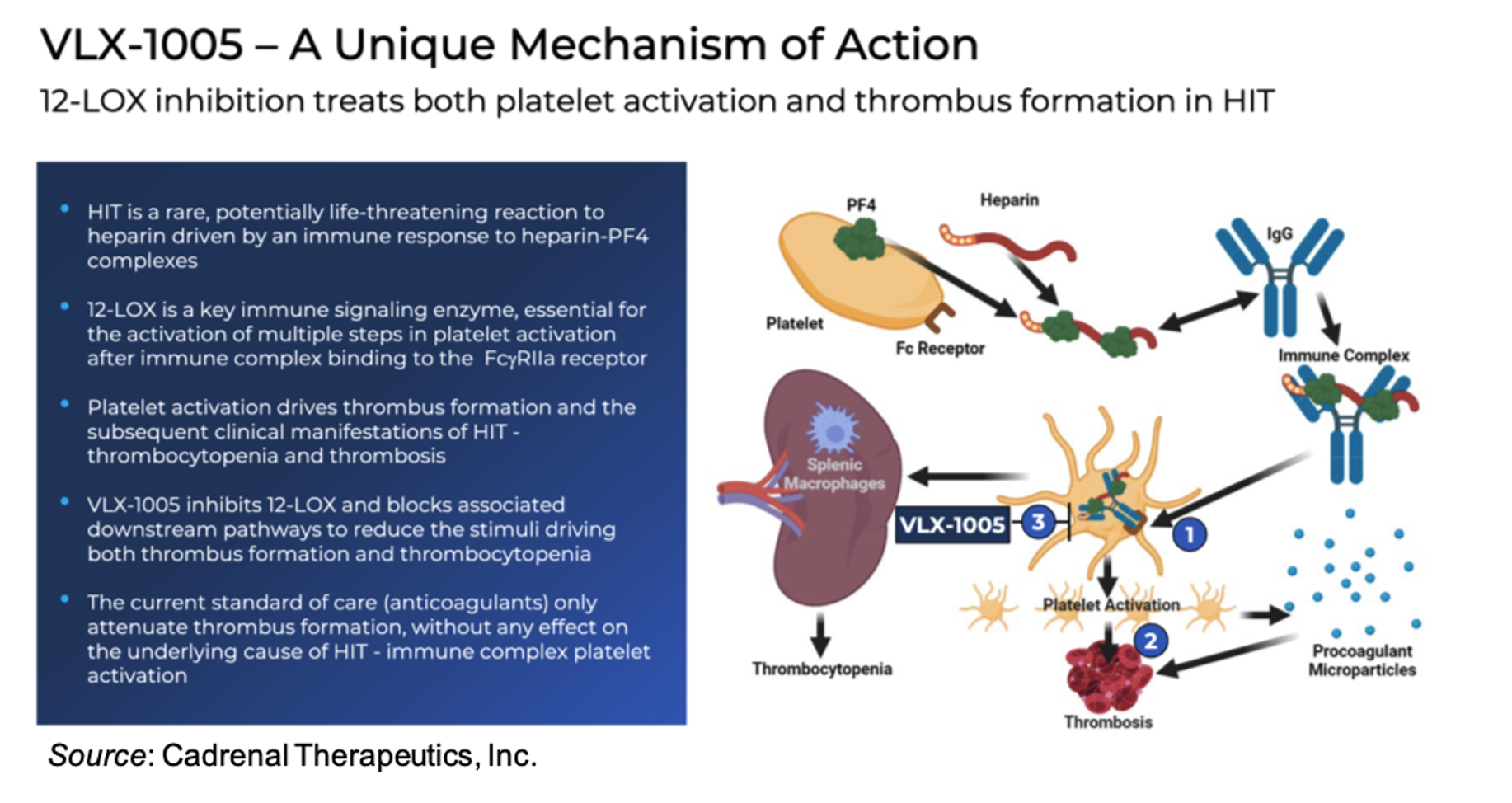
VLX-1005 is a highly selective inhibitor of human 12-LOX and is designed to reduce or prevent platelet activation and the downstream pro-thrombotic cascade in HIT. By inhibiting 12-LOX activity, VLX-1005 reduces the production of proinflammatory and procoagulant 12-LOX metabolites that feed into platelet activation loops. The drug also addresses the immune-driven aspect of HIT pathogenesis, which is the pathway that links immune complexes and platelet hyperactivity, a mechanism wholly distinct from direct anticoagulation. Lastly, preclinical and Phase 1 clinical trial results indicate there is no increased bleeding signal, which distinguishes VLX-1005 from traditional anticoagulants that reduce the risk of clotting at the cost of an increased risk of bleeding.
12-LOX Biology
12-LOX (encoded by the ALOX12 gene) catalyzes the oxidation of arachidonic acid to proinflammatory lipid mediators such as 12-hydroxyeicosatetraenoic acid (12-HETE) (Dobrian et al., 2011). 12-LOX is expressed in platelets (Contursi et al., 2022); however, unlike cyclooxygenase-1 (COX-1), which drives thromboxane A2 (TXA2) production and classical platelet aggregation, 12-LOX does not primarily initiate aggregation but instead acts as a potentiator and amplifier of platelet activation. The main bioactive product of 12-LOX, 12-HETE, is a lipid molecule that easily transits cell membranes and can induce its effects both intracellularly, where it promotes oxidative stress, and extracellularly, where it impacts a variety of signaling pathways to modulate inflammatory activity. Importantly, 12-HETE does not cause strong platelet activation alone; it just lowers the activation threshold, thus making platelets hyper-responsive (Tamang et al., 2024).
In HIT, platelets are activated via FcγRIIa binding of anti-PF4/heparin antibody complexes (Yeung et al., 2024). 12-LOX acts downstream of FcγRIIa signaling: FcγRIIa activation leads to phospholipase A2 activation that leads to arachidonic acid release. The arachidonic acid is then converted to 12-HETE, and it is the 12-HETE that feeds back to sustain and amplify the activation signal. By inhibiting 12-LOX, pathologic amplification is selectively inhibited by reducing 12-HETE generation. This targets the disease itself and, as opposed to current HIT therapies, does not result in a residual thrombotic risk. The high selectivity of VLX-1005 helps to preserve baseline platelet function and allows it to be used with standard anticoagulants while being short-lived and controllable in an in-patient setting.
Phase 1 Results
Veralox previously completed a Phase 1a study in healthy volunteers that consisted of a single ascending dose (SAD) and multiple ascending dose (MAD) portion to evaluate the safety, tolerability, pharmacokinetics (PK and pharmacodynamics (PD) of VLX-1005. In that study, VLX-1005 was found to be well tolerated with no reports of serious adverse events (SAEs), dose-limiting toxicities (DLTs) or discontinuations. The data showed a dose linear increase in key PK metrics with no upper limits on tolerability to the maximum dose tested.
Veralox also completed a Phase 1b drug-drug interaction (DDI) study of VLX-1005 in combination with argatroban, an anticoagulant drug approved for the treatment of HIT. The results showed that co-administration of VLX-1005 with argatroban was well tolerated with no SAEs. Analysis of the PK and PD data showed no evidence for DDI.
Phase 2 Results
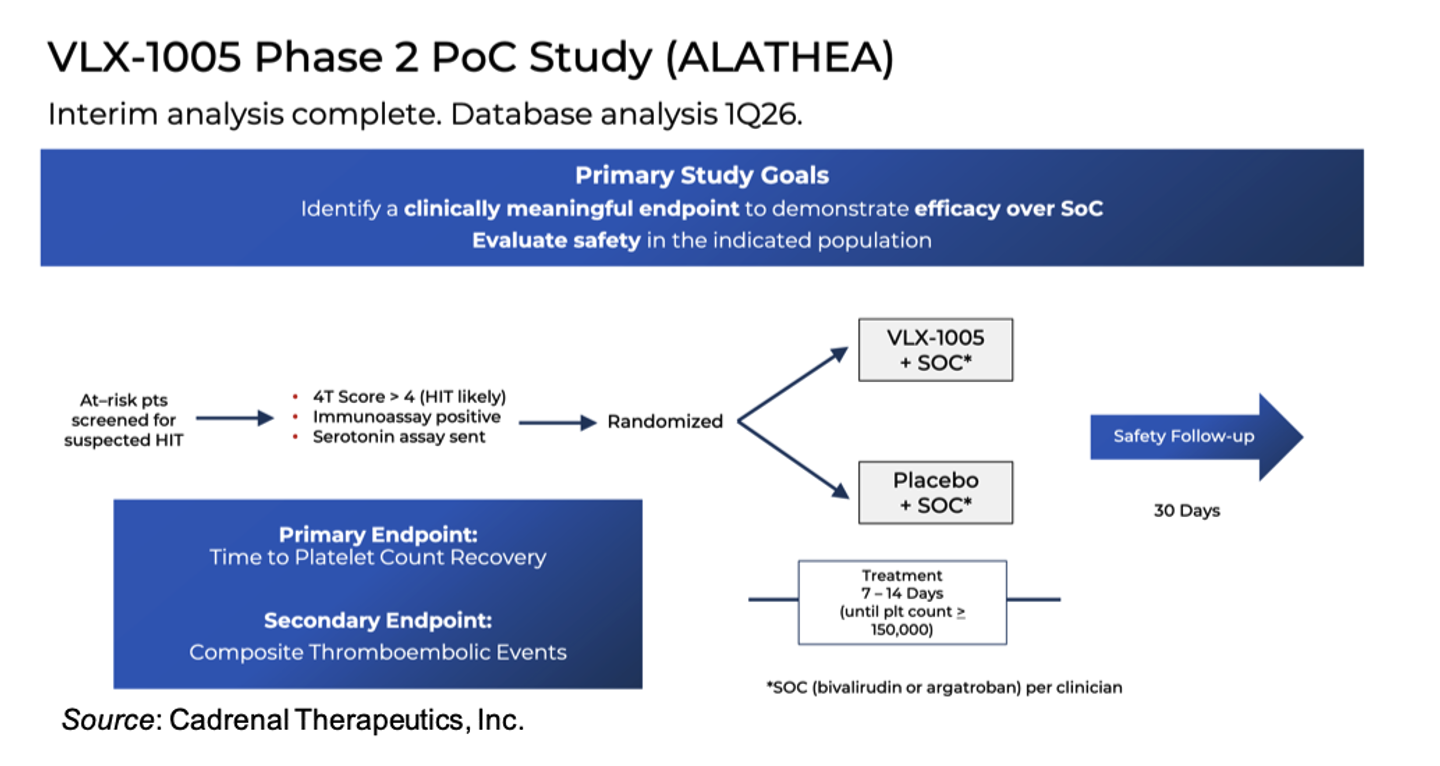
A randomized, double blind, placebo controlled Phase 2 proof-of-concept study evaluated VLX-1005 against placebo in confirmed/suspected HIT patients on standard-of-care anticoagulation therapy (e.g., argatroban or bivalirudin) (NCT05785819). An overview of the study is given below. Emerging data from that trial suggests that VLX-1005 may reduce thrombotic complications, which are a major driver of morbidity and mortality in HIT, thus making reductions in those events a highly relevant efficacy signal.
Regulatory Next Steps
VLX-1005 has Orphan Drug Designation (ODD) and Fast Track status from the FDA, as well as ODD in the E.U. Cadrenal is planning to meet with the FDA for an ‘End-of-Phase 2’ meeting as it prepares for a Phase 3 program. We expect details of a Phase 3 trial to be announced after the company receives formal feedback from the FDA on suitable endpoints, total number of patients, etc.
Market Opportunity
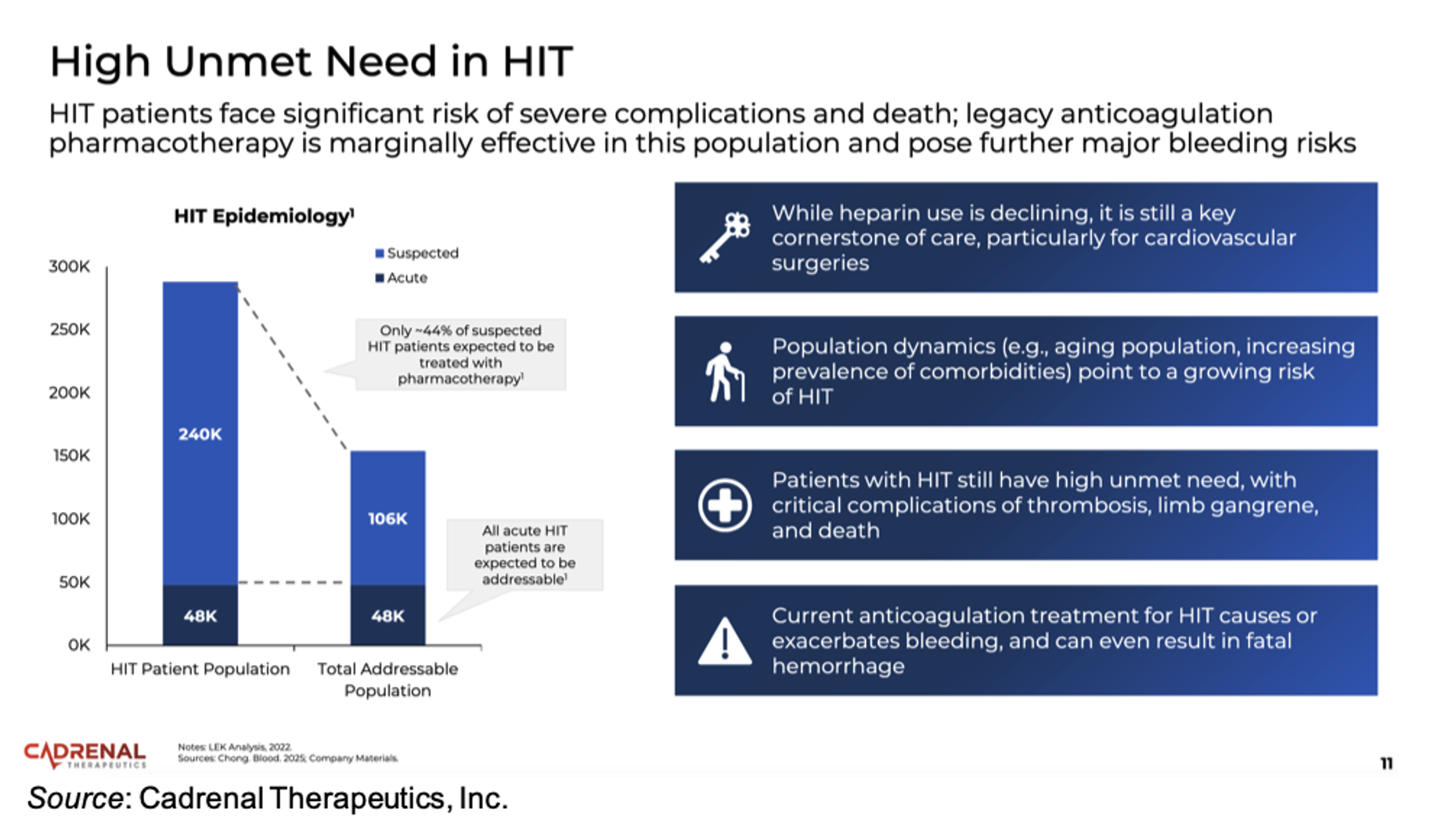
Currently approved HIT treatments focus on non-heparin anticoagulation, which mitigate clot propagation but do not directly modulate immune platelet activation, which is the core driver of HIT pathology. Thus, there is a high residual risk of thrombosis that persists in HIT patients despite anticoagulant therapy. In addition, there are no approved agents that target the immune-mediated platelet activation pathway, thus positioning VLX-1005 as a first-in-class drug. There are approximately 240,000 suspected cases and approximately 48,000 confirmed cases of HIT in the U.S. each year, thus offering a large patient population that could potentially benefit from a safe and effective HIT therapy.
Acquires Portfolio of Factor XIa Inhibitors
In September 2025, Cadrenal announced it had acquired a portfolio of Factor XIa inhibitors from eXIthera Pharmaceuticals. Included in the acquired assets is frunexian, a Phase 2-ready asset that is being developed as an acute anticoagulant therapy for prevention of coagulation, where medical devices play a significant role, such as in cardiopulmonary bypass and catheter thrombosis. The portfolio also includes EP-7327, an oral Factor XIa inhibitor for the prevention and treatment of major thrombotic conditions.
Anticoagulation therapy is utilized for the prevention and treatment of thrombosis without disrupting hemostasis. Current anticoagulants target FXa or thrombin, which are enzymes in the extrinsic pathway of coagulation. However, inhibition of Factor XIa is a new area of focus since epidemiological and animal data support the concept that Factor XIa is essential for thrombosis but not required for hemostasis. Individuals with congenital FXI deficiency do not have an increased risk for spontaneous bleeding, while at the same time these patients are at a lower risk for venous thromboembolism (VTE) and ischemic stroke. Conversely, patients with elevated FXI levels are at a higher risk for VTE and stroke.
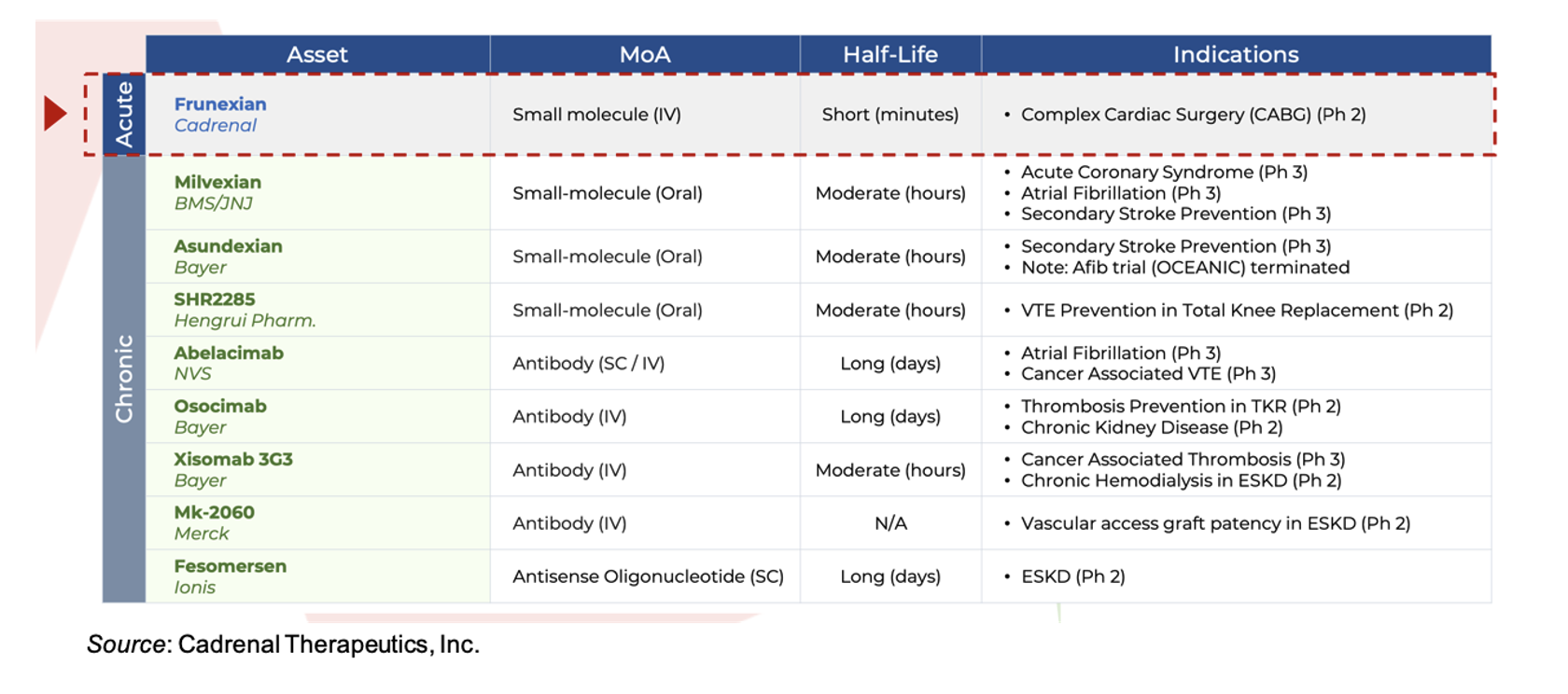
Frunexian is a small-molecule inhibitor of Factor XIa and is designed for intravenous administration. While there are a number of other FXIa inhibitors in development (as shown in the following table), frunexian is the only one we are aware of that is being developed in the acute care setting.
Frunexian has been successfully tested in two Phase 1 clinical trials in healthy volunteers. The results showed that the drug was well tolerated at doses up to 2.25 mg/kg/h, and there were no reports of serious adverse events. Steady-state plasma levels of the drug were rapidly achieved, and importantly, the drug was quickly cleared following cessation of dosing, an important factor for use in the acute care setting. The company has additional work to perform regarding the manufacturing of frunexian before initiating the clinical trial plan, which may initially focus on complex cardiac surgery.
Financial Update
In November 2025, Cadrenal announced financial results for the third quarter of 2025. As expected, the company did not record any revenues for the three months ending September 30, 2025. R&D expenses in the third quarter of 2025 were $0.7 million compared to $0.8 million in the third quarter of 2024. The decrease was primarily due to decreased consulting expenses, partially offset by an increase in transaction-related expenses. G&A expenses were $2.0 million in the third quarter of 2025 compared to $1.7 million in the third quarter of 2024. The increase was primarily due to increased public company expenses and non-cash stock-based compensation.
As of September 30, 2025, Cadrenal had approximately $3.9 million in cash and cash equivalents. While the company has sufficient capital to fund operations for the next 12 months, it will need to raise additional capital to conduct the planned Phase 3 program for VLX-1005. The company currently has approximately 2.1 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 3.1 million.
Conclusion
Cadrenal has now enhanced its pipeline with the acquisitions of VLX-1005 and frunexian, and we look forward to additional information regarding their development. We anticipate VLX-1005 being the main focus for the company going forward, as it is ready for Phase 3 testing. HIT is a logical disease area to focus on as VLX-1005 targets the pathological drivers of the disease, while there are few effective treatment options currently available. We have incorporated VLX-1005 into our model along with estimated costs for the Phase 3 program, and our valuation is now $25 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.