By David Bautz, PhD
NASDAQ: DWTX
READ THE FULL DWTX RESEARCH REPORT
Business Update
143 Patients Enrolled in Phase 2b Trial of Halneuron® in CINP; Topline Results in 3Q26
Dogwood Therapeutics, Inc. (NASDAQ: DWTX) is currently conducting the Phase 2b HALT-CINP (Halneuron® Treatment of Chemotherapy-Induced Neuropathic Pain) trial. The Phase 2b trial is a four-week study that is examining the safety and efficacy of Halneuron in patients with moderate-to-severe CINP. The primary efficacy endpoint is the change from baseline at Week 4 in the weekly average of daily 24-hour recall pain intensity scores, which will be recorded in e-diaries on participants’ smartphones. Secondary efficacy endpoints include Patient Global Impression of Change (PIGC), PROMIS Fatigue, PROMIS Sleep, PROMIS-29, Pain Interference, Hospital Anxiety and Depression Scale (HADS), and Neuropathic Pain Symptom Inventory (NPSI).
In December 2025, Dogwood announced positive interim results for the trial. Unblinded data were reviewed by the independent statistical review committee, and they concluded that Halneuron-treated patients are demonstrating separation from placebo-treated patients in pain improvement over the four-week study. The interim analysis included data from 97 patients that have completed treatment.
The company recently announced that it has completed over 50% of the planned enrollment of the study and that 143 patients have been enrolled. Dogwood is planning to finish enrollment at the end of July 2026 with a projected total patient population of 210 to 240 patients, which would provide 80-85% power to detect a statistically significant Halneuron treatment difference. We anticipate topline results from the trial in the third quarter of 2026.
Previously, the company announced that data from the first 116 patients to complete the study showed an overall dropout rate of approximately 4.3%, which is much lower than the dropout rates for both duloxetine (20%+) and pregabalin (30-40%). The patient population had a mean 5-year duration of moderate-to-severe neuropathic pain, and it includes a majority of refractory CINP patients, as judged by the fact that approximately 2/3rd of the population was on concomitant pain medications.
Dogwood also recently completed a financing of up to $26.9 million, of which $12.5 million has been received, which will finance operations through the release of topline data. In addition, the recent financing will also allow Dogwood to initiate an open-label extension that will test different dosing regimens and frequencies to help complete the data package that the company intends to present to the FDA following release of the Phase 2b topline data, if positive. The data obtained from these studies will be used to guide the design of a Phase 3 program. In addition, Dogwood now has sufficient capital to conduct a bridging study with synthetic Halneuron, which the company is planning to use in the Phase 3 program.
SP16 Trial to Initiate in Mid-2026
In September 2025, Dogwood announced a royalty-free, global license deal with Serpin Pharma for SP16 for the management of cancer related pain (CRP), including a broad range of chemotherapy induced neuropathy symptoms. SP16 is an intravenously administered peptide that functions as an LRP1 agonist and has shown anti-inflammatory and neural repair activity. For a full overview of SP16, please see our previous report here.
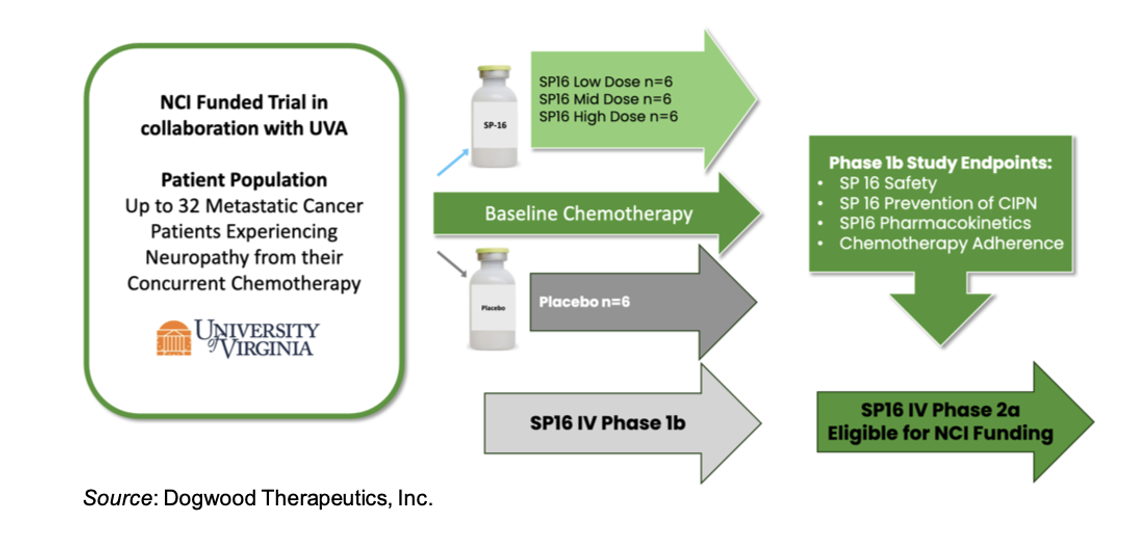
A Phase 1b trial of SP16 in Chemotherapy Induced Peripheral Neuropathy (CIPN) is scheduled to initiate in mid-2026 and is fully funded by the National Cancer Institute (NCI). It will take place at the University of Virginia. Up to 32 patients with metastatic cancer who are experiencing neuropathy from their concurrent chemotherapy will be enrolled into one of four dosing cohorts, with each cohort having six patients on SP16 and two on placebo. Endpoints for the study include safety, pharmacokinetics of SP16, the prevention of CIPN, and adherence to chemotherapy. Positive results from the trial may lead to a Phase 2a trial that could be eligible for funding from NCI.
Financial Update
On March 18, 2026, Dogwood announced financial results for 2025. As expected, the company did not report any revenues in 2025. R&D expenses for the full year ending December 31, 2025, were $21.8 million compared to $3.5 million for the full year ending December 31, 2024. The increase was primarily due to acquired in-process research and development expenses of $12.0 million associated with the licensing agreement of SP16 and the impact of the business combination with Pharmagesic Inc, along with increased clinical trial, drug development, and manufacturing expenses. G&A expenses for 2025 were $6.1 million compared to $8.7 million for 2024. The decrease was primarily due to a decrease in nonrecurring transaction costs of $3.9 million related to the combination of Pharmagesic in October 2024 and decreased costs associated with being a public company, partially offset by increases in salaries, legal and professional fees, franchise tax fees, and other general and administrative costs.
As of December 31, 2025, Dogwood had approximately $6.5 million in cash and cash equivalents. As mentioned above, subsequent to the end of the year, the company executed a financing for up to $26.8 million, with gross proceeds of $12.5 million already received and the potential for up to an additional $14.4 million in gross proceeds from the exercise of warrants. We estimate this is sufficient to fund operations past the announcement of topline results for the Phase 2b trial of Halneuron. As of March 10, 2026, Dogwood had approximately 33.4 million common shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 38.9 million.
Conclusion
Now that Dogwood has secured sufficient financing to fund operations past the anticipated data release from the Phase 2b HALT-CINP trial, the company can turn its attention to finishing enrollment ahead of the anticipated data readout in the third quarter of 2026. The financing will also allow the company to collect additional data on dosing regimens through an open-label extension that will help guide the design of the Phase 3 program. We look forward to additional enrollment updates from the company as the year progresses and topline results in the third quarter of 2026. With no changes to our model, our valuation remains at $15 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.