By David Bautz, PhD
NASDAQ:ESLA
Business Update
On February 9, 2026, Estrella Immunopharma, Inc. (NASDAQ:ESLA) announced that results from the Phase 1 Trial of CD19-ARTEMIS® T cells (EB103) in patients with aggressive B Cell Non-Hodgkin Lymphoma (NHL) were presented at the 2026 ASTCT & CIBMTR Tandem Meetings (American Society for Transplantation and Cellular Therapy and Center for International Blood & Marrow Transplant Research). The results showed a 100% overall response rate (ORR) and complete response (CR) rate at dose level 2 through the data cutoff, including a patient with Primary Central Nervous System Lymphoma (PCNSL), a highly aggressive NHL subtype with an approximately 30% 5-year survival rate. Importantly, there were no Grade ≥ 3 cytokine release syndrome (CRS) events and only one Grade 3 immune effector cell–associated neurotoxicity syndrome (ICANS) event observed among the nine evaluable patients.
EB103
EB103 is based on Estrella’s ARTEMIS (Antibody-T-Cell Receptor Engagement System) platform, a proprietary antibody-T-cell receptor (AbTCR) architecture licensed that is designed to combine specific antibody targeting with physiologic T-cell receptor–like signaling in order to improve safety, persistence, and overall anti-tumor activity. The ARTEMIS approach represents a departure from conventional CAR-T constructs, which typically fuse a single-chain variable fragment (scFv) to an intracellular signaling domain composed of CD3ζ in combination with costimulatory domains (e.g., 4-1BB or CD28). The following figure gives an overview of the ARTEMIS construct in comparison to traditional CAR-T constructs.
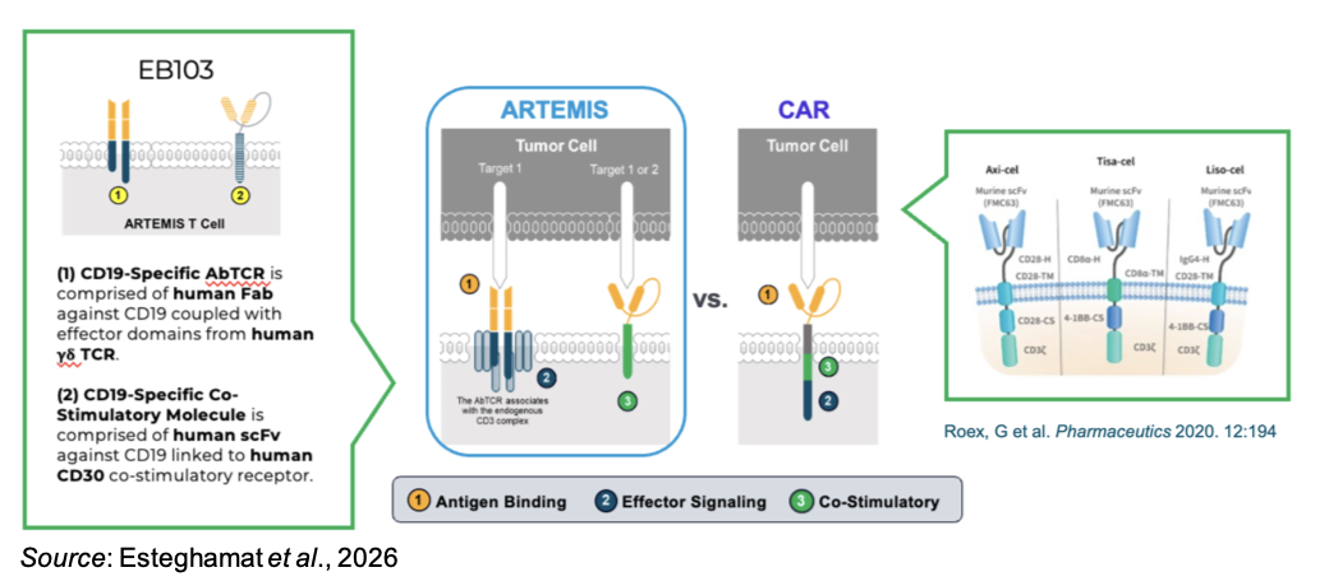
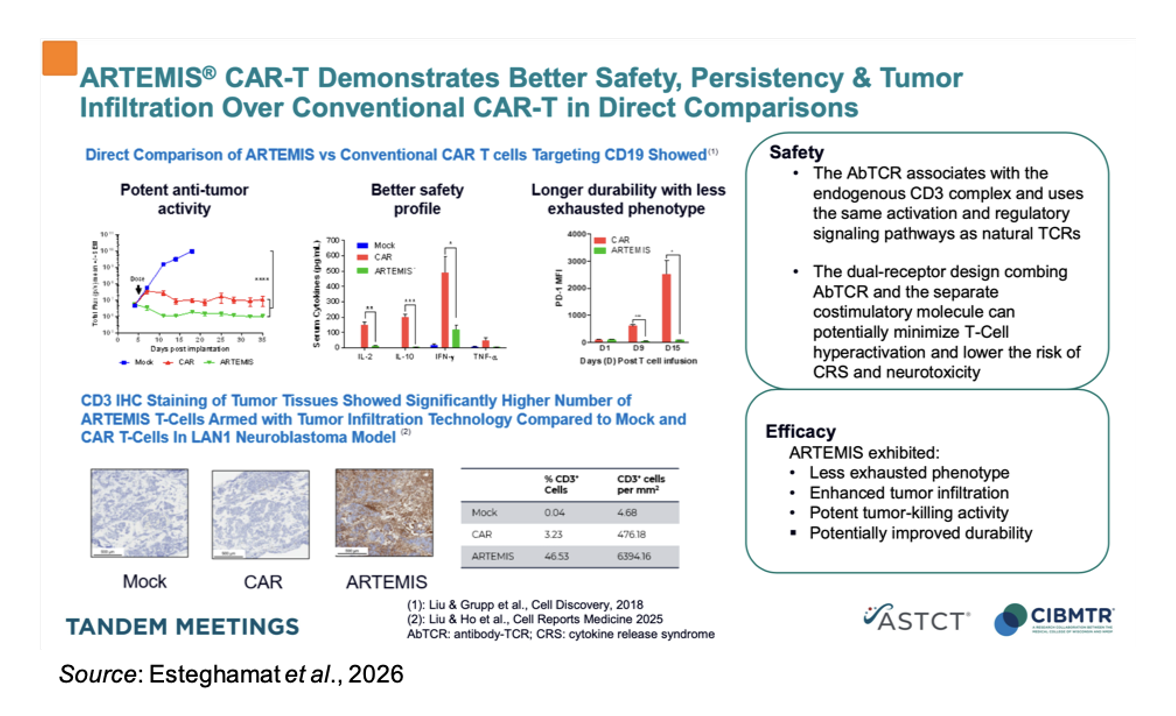
While CAR-T modalities have demonstrated potent antitumor responses in select hematologic malignancies, their synthetic signaling architecture has been implicated in supraphysiologic T-cell activation, excessive cytokine release, and premature T-cell exhaustion, all factors that collectively limit durability and widen the therapeutic window. ARTEMIS seeks to address these limitations by integrating antigen recognition through antibody fragments with native T-cell receptor signaling machinery, thereby aligning target engagement with more regulated and nuanced T-cell activation pathways. The following figure shows a direct comparison of ARTEMIS vs conventional CAR-T cells in a preclinical B-cell lymphoma model. The ARTEMIS cells showed less exhausted phenotype, enhanced tumor infiltration, potent tumor-killing activity, and potentially improved durability.
STARTLIGHT-1 Trial
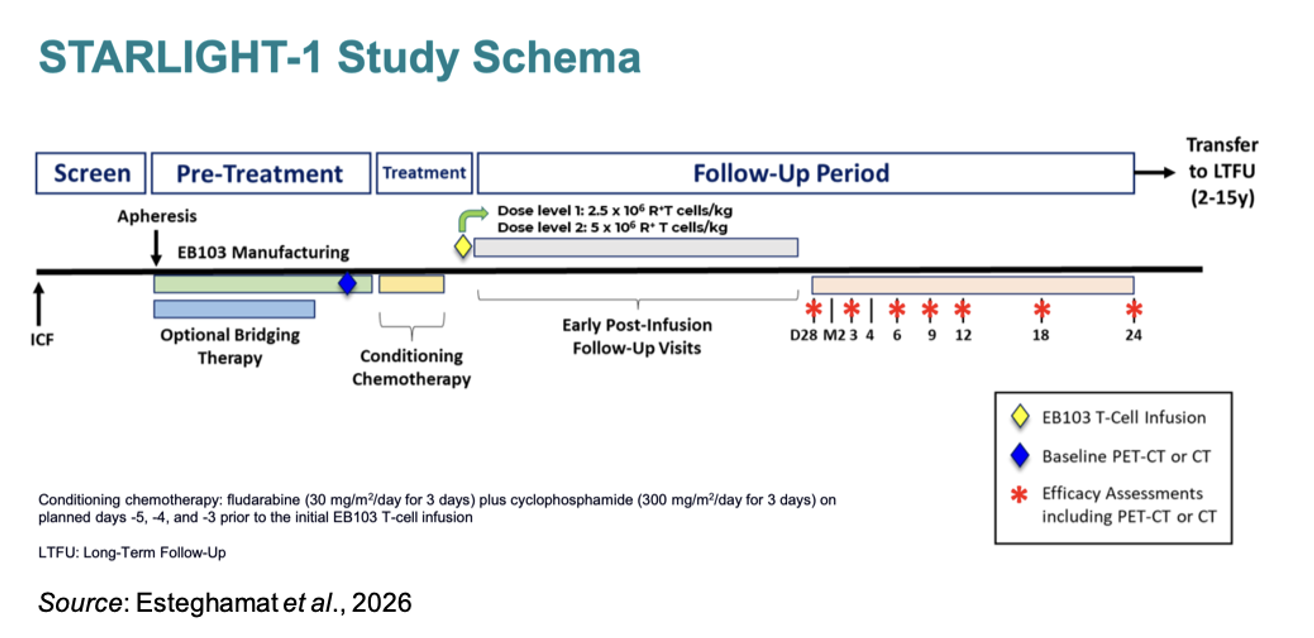
The following figure summarizes the STARLIGHT-1 Phase 1/2 clinical trial, which enrolled patients with aggressive B cell lymphomas and included a dose escalation and dose expansion phase. Dose level 1 (DL1) was 2.5 x 106 receptor-positive T cells/kg, while dose level 2 (DL2) was 5.0 x 106 receptor-positive T cells/kg. The primary endpoints of the study were the type, frequency, and severity of adverse events (AEs) and determining the recommended Phase 2 dose (RP2D) based on the maximum tolerated dose and manufacturing capability. Secondary endpoints included clinical outcomes along with pharmacokinetics.

The following figure shows the study schematic. Prior to treatment, patients received conditioning chemotherapy: fludarabine (30 mg/m2/day for 3 days) plus cyclophosphamide (300 mg/m2/day for 3 days) on planned days -5, -4, and -3 prior to the initial EB103 T cell infusion. Efficacy assessments are scheduled for Day 28, Month 3, Month 6, Month 9, Month 12, Month 18, and Month 24.
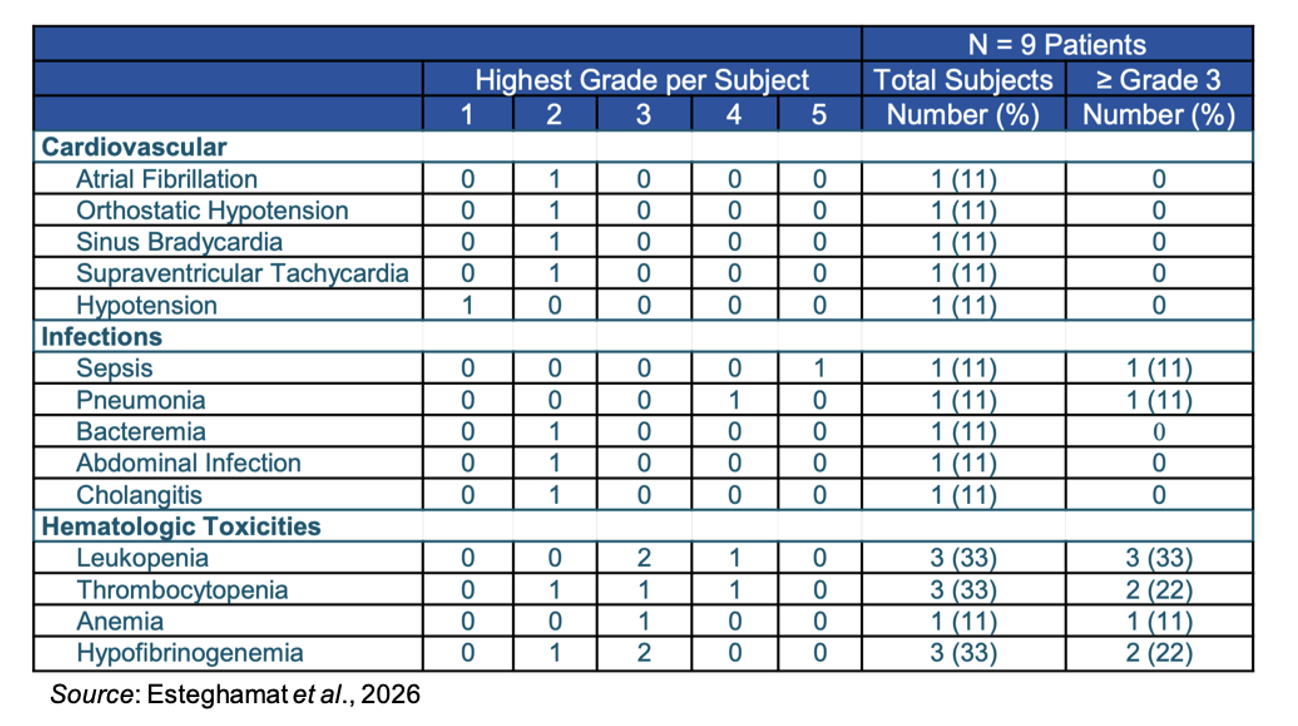
The following table gives the adverse events not due to CRS or ICANS for the nine evaluable patients. Two subjects experienced serious adverse events; however, neither of them was due to EB103: one patient had grade 3 blunt trauma, and one patient in DL2 died from pneumonia and respiratory failure.
When looking specifically at CRS and ICANS, all subjects experienced CRS, however none had ≥ Grade 3 and 7/9 patients had ICANS, with six patients experiencing Grade 1-2 and only one patient having Grade 3. The duration of the ICANS event for the Grade 3 patient was two days.
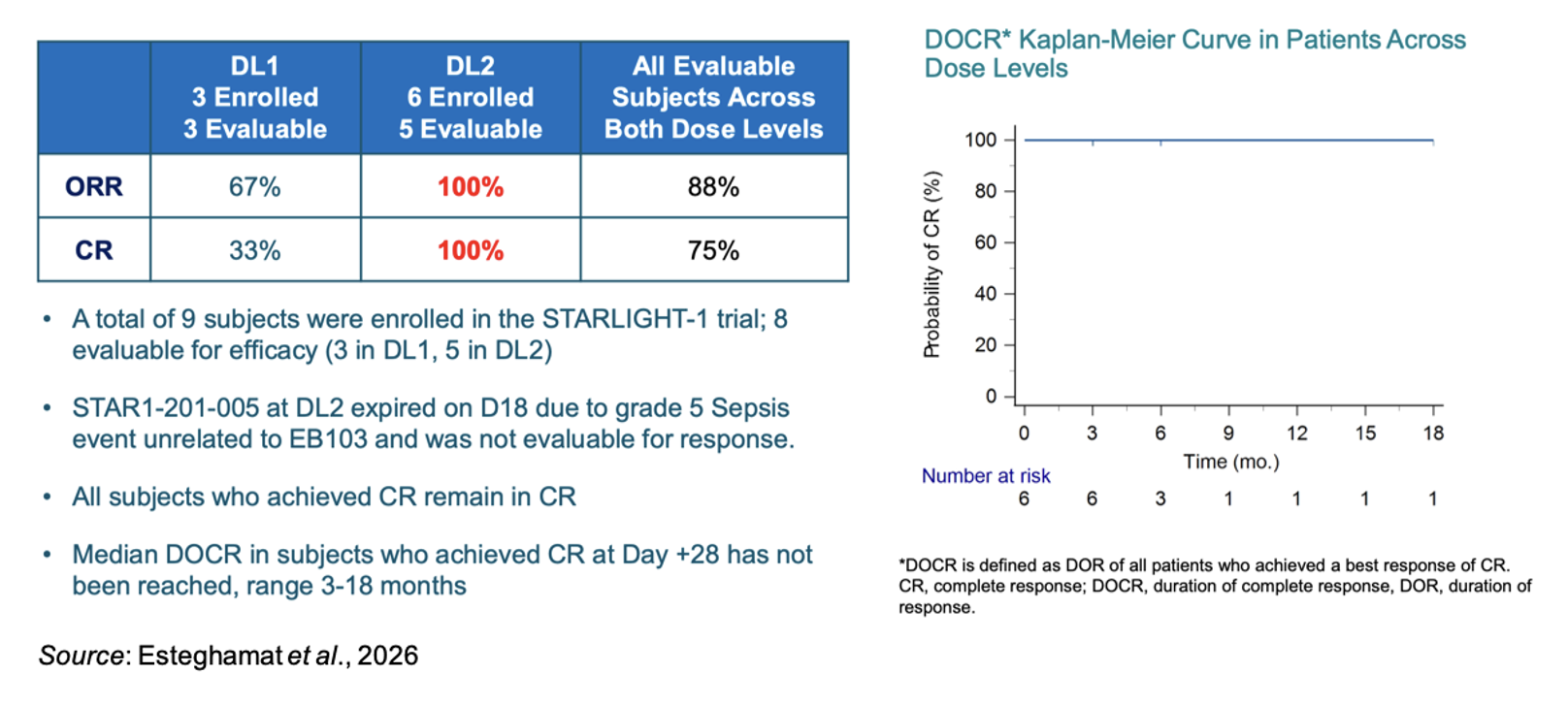
The following figure shows the efficacy results for the eight evaluable patients (3 in DL1 and 5 in DL2). All subjects who achieved a CR remained in CR at the time of cutoff. The median duration of complete response (DOCR) has not been reached with the CR’s ranging from 3-18 months.
Efficacy Comparisons
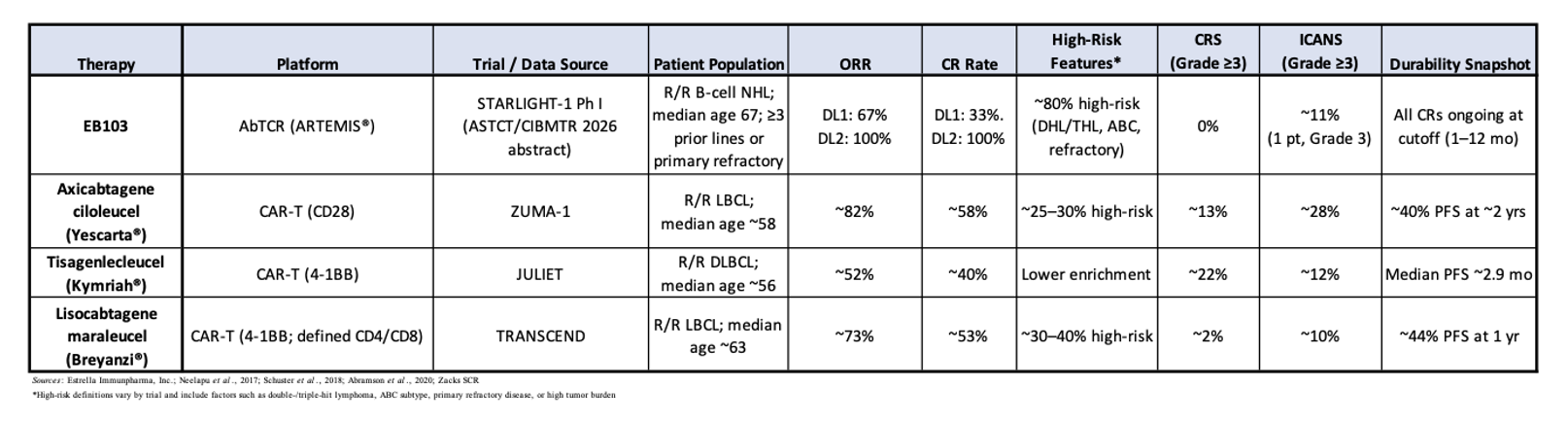
When benchmarked against approved CD19 CAR-T therapies, EB103’s early efficacy compares favorably, particularly when adjusted for patient risk profile. In pivotal trials of first-generation CAR-T therapies in aggressive B-cell lymphoma:
- ZUMA-1 (axicabtagene ciloleucel): reported an ORR of ~82% and CR rate of ~58% in a population with fewer high-risk features and a median age of ~58 years (Neelapu et al., 2017).
- JULIET (tisagenlecleucel): reported an ORR of ~52% and CR rate of ~40%, with slower expansion kinetics and broader eligibility (Schuster et al., 2019).
- TRANSCEND (lisocabtagene maraleucel): reported ORR ~73% and CR ~53%, with a more controlled toxicity profile (Abramson et al., 2020).
The following table shows how EB103’s DL2 response rate compares favorably with the approved CAR-T therapies, particularly when adjusted for older age, heavier pretreatment, and unusually high enrichment for high-risk disease biology. While difficult to make cross-trial comparisons and knowing that all results must be interpreted cautiously, especially in small Phase I cohorts, the magnitude of response at DL2 is notable given the high-risk patient population.
Conclusion
The early results from the Phase 1/2 STARLIGHT-1 trial are very encouraging and show how EB103 could offer equal or greater efficacy than the currently approved CD19 CAR-T therapies with a more benign AE profile. While a direct comparison to results from other clinical trials is not possible, the results certainly support moving to the dose expansion phase of the study and further evaluation in a larger population is warranted to confirm the Phase 1 results.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.