By David Bautz, PhD
NASDAQ: FBLG
READ THE FULL FBLG RESEARCH REPORT
Business Update
Advancing Fibroblast Therapy on Multiple Fronts
In the past month, FibroBiologics, Inc. (NASDAQ: FBLG) has disclosed the advancement of its fibroblast therapy platform into additional indications, including:
- On March 2, 2026, the company announced that the U.S. Patent and Trademark Office (USPTO) issued U.S. Patent No. 12,544,407 B2 that covers methods of treating bone diseases or disorders, including osteoporosis, through the administration of fibroblast cells. The claims encompass both systemic and local administration of fibroblasts to modulate bone remodeling through inhibition of osteoclast activity and/or promotion of osteoblast activity. Osteoporosis affects millions of individuals worldwide, and using fibroblasts to modulate bone resorption and formation pathways would represent a paradigm shift in the way that disease is treated.
- On March 12, 2026, the company announced the expansion of its wound care platform into the treatment of patients with acute and chronic burn injuries. In preclinical models, treatment with CYWC628 was associated with a nearly four-fold increase in tissue-level IL-10, which supports wound healing and tissue repair. In addition, treatment with CYWC628 resulted in a significant reduction in IL-1β, which promotes inflammation in burn wounds. The combination of tissue repair and inflammation reduction could accelerate wound closure and improve the quality of wound healing.
Site Onboarding Complete for Phase 1/2 Trial of CYWC628
FibroBiologics recently announced that onboarding of the clinical sites for the upcoming Phase 1/2 clinical trial of CYWC628 in diabetic foot ulcers (DFUs) has been completed. The planned study will enroll 120 patients across up to 10 study sites in Australia. The company has already received both public and private Human Research Ethics Committee (HREC) approvals and has fulfilled all other regulatory requirements. The company will be completing the manufacturing of CYWC628 drug product in the 1Q26, with plans to dose the first patient in the 1H26. Interim data could be available by mid-2026, with the full topline results expected by the end of 2026.
The trial is a multicenter, randomized study designed to evaluate the safety, tolerability, and efficacy of FibroBiologics topically administered allogenic fibroblast therapy, CYWC628. Study subjects will receive up to 12 weeks of treatment using either standard of care (SoC) plus a high or low dose of CYWC628, or SoC alone. Study outcomes include wound healing, efficacy of response, and safety parameters. An interim analysis will be conducted after a predefined number of participants complete six weeks of treatment to assess primary safety and efficacy endpoints.
DFUs cause significant morbidity for the 6.3% of diabetic adults (~33 million) that develop them. Of those, 20% will require lower extremity amputation, and 10% will die within the first year of their first DFU. In addition, once a DFU forms, there is a high rate of recurrence, both at one year (40%) and three years (70%).
Fibroblasts have excellent therapeutic potential in the treatment of DFUs due to the critical role they play in every stage of wound healing, including hemostasis, inflammation, proliferation, and remodeling. Importantly, fibroblasts are the key cells that secrete extracellular matrix proteins that maintain all the tissues and organs in the body.
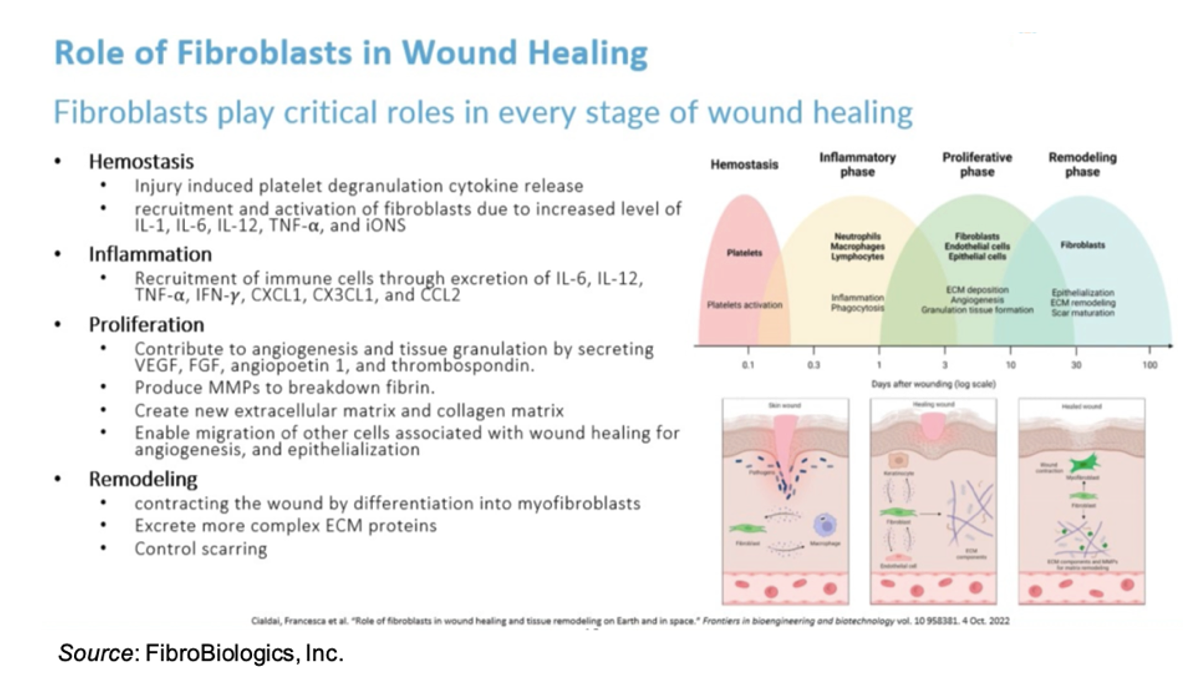
The company does not utilize single cell fibroblasts for treatment but instead a fibroblast spheroid, which is composed of approximately 3,000 fibroblasts and is administered to the top of the wound, at which time the cells migrate from the surface of the wound and release various cytokines and growth factors to initiate the wound healing process. The use of spheroids is more practical from a therapeutic perspective as they have higher viability than single cells, they don’t require pre-culturing before administration, they can be easily frozen and thawed, and they have a significantly higher potency and efficacy compared to single cells.
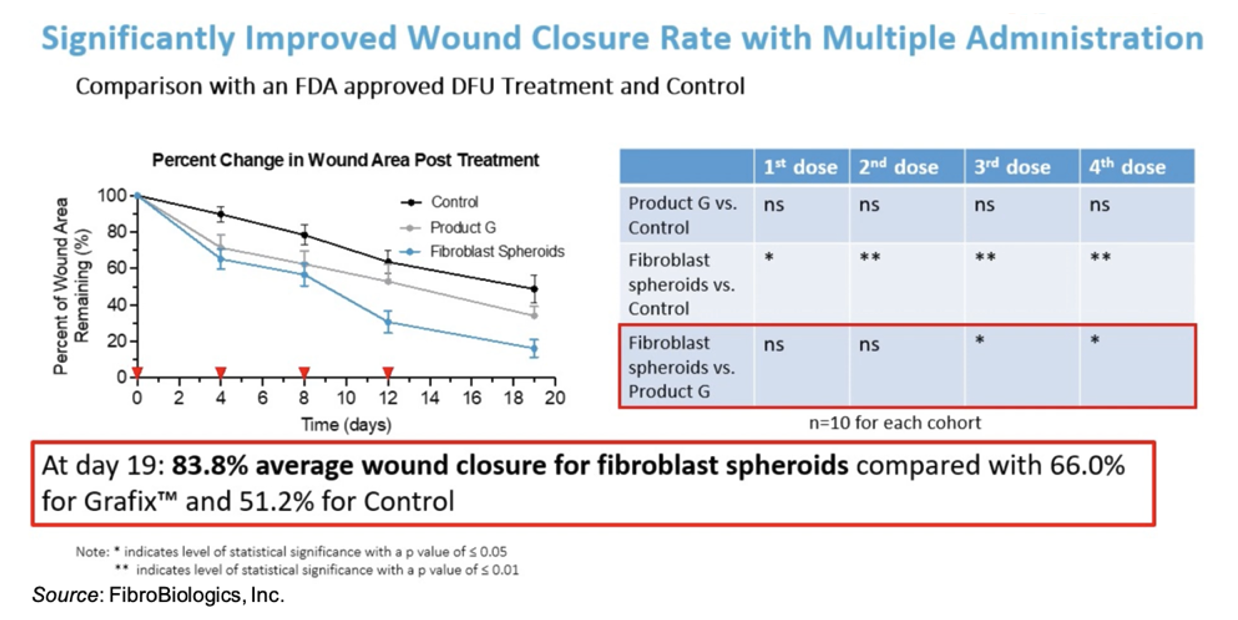
FibroBiologics has compiled a robust pre-clinical data set showing the efficacy of fibroblast spheroids in the treatment of wounds. For example, the following figure shows results using a diabetic mouse model in which administration of fibroblasts led to a statistically significant average 83.8% wound closure by Day 19 compared to 66.0% for Grafix™ and only 51.2% for control.
While wound healing is important, the quality of the wound healing is just as important. FibroBiologics has data on seven key biomarkers that are key to demonstrating the quality of the wound healing. Fibroblast treatment shows much better re-epithelialization, granulation, cell proliferation, neo-vascularization, recruitment and proliferation of fibroblasts, keratinocyte migration, and epithelial-mesenchymal transition. Intriguingly, even though the fibroblast spheroids are administered topically, there appears to be a systemic effect on cytokine levels, including IL-6, TNF-α, IL-1β, and IL-10.
The use of fibroblast spheroids for wound healing can be thought of as a platform technology. In addition to the treatment of DFUs, fibroblast spheroids could also be used for the treatment of burns and surgical wounds.
In regard to safety, the company has performed a number of experiments to examine any potential adverse events associated with fibroblast-based therapy. The cells do not graft into tissue. Following application, the cells stay on the surface of the wound and initiate the healing process before gradually dying off within four days of treatment. In addition, there is no impact on CBC, WBC, liver function, or kidney function, thus showing that administration of fibroblasts appears to be quite safe.
Financial Update
On March 25, 2026, FibroBiologics announced a 1-for-20 reverse stock split to regain compliance with Nasdaq’s minimum bid price requirement. The reverse stock split became effective on March 30, 2026, and reduced the number of outstanding shares from approximately 70.3 million to approximately 3.5 million.
On April 1, 2026, the company announced the pricing of a $3 million public offering of a total of 2,272,728 shares of common stock and warrants to purchase up to 2,272,728 shares of common stock at a combined offering price of $1.32 per share and accompanying warrant. The warrants have an exercise price of $1.32 per share and will expire five years following stockholder approval.
Conclusion
Everything is in place for the Phase 1/2 clinical trial of CYWC628 and we look forward to the initiation of the trial, with the potential for interim results in mid-2026 and topline results before the end of this year. In addition to initiating the DFU trial, the company is continuing to push its fibroblast therapy into additional indications, as shown by the patent issuance for treating osteoporosis and the preclinical data supporting its use in burn treatment. We have adjusted our model based on the reverse split and the recent financing, and our valuation is now $19 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.