By John Vandermosten, CFA
NASDAQ: HURA
READ THE FULL HURA RESEARCH REPORT
Operational and Financial Results
On April 1st, 2026, TuHURA Biosciences, Inc. (NASDAQ: HURA) reported 2025 financial and operational results and filed its Form 10-K with the SEC. The company is conducting Phase III and Phase Ib/IIa trials in Merkel cell carcinoma (MCC) and is expected to soon start a Phase Ib/II trial for TBS-2025 in acute myeloid leukemia (AML). The IFx-2.0 Phase III is expected to read out in 2027. The TBS-2025 anti-VISTA asset is now the subject of FDA meetings centering on the development plan, and clinical trials could begin in 2H:26. Other program work includes the identification of a lead antibody drug conjugate (ADC) in AML, along with initiation of proof-of-concept studies. Outside of operational efforts, TuHURA added new team members and is presenting at scientific and investor conferences. Below, we summarize the company’s 2025 financial results.
TuHURA generated no revenues in 2025 and expended $31.8 million on operational activities related to advancing IFx-2.0, TBS-2025, and other programs, producing a net loss of $30.1 million or $0.63 per share. For the year ending December 31st, 2025, and versus the same prior year period:
- Research & development expense totaled $20.5 million, increasing 54% from $13.3 million on higher facilities, salary and personnel related costs, greater clinical spending on IFx-2.0 and TBS-2025, and increased allocations to the preclinical IFx-3.0, myeloid derived suppressor cell (MDSC), and REM-001 programs;
- General & administrative expense totaled $11.2 million, which includes acquisition related costs of $3.7 million. The total was up markedly from $4.3 million, due to increases in non-cash stock compensation, merger transaction costs, and expenses related to being a public company;
- Other items included $2.2 million that consisted of grant income related to Kintara’s REM-001 asset and reimbursements from Health and Human Services, and amounts related to share settlement to former Kineta employees, and employee retention tax credit. The most significant item was $1.6 million in income related to the fair value of Kineta merger holdback shares, partially offset by a $185,000 loss on Kineta employee separation payments;
- Net interest expense was $489,000;
- Net loss was $30.1 million or $0.63 per share. Removing the nonoperating items from the “Other” line item produces a net loss of $32.3 million or $0.67 per share.
As of December 31st, 2025, TuHURA held $3.6 million in cash on its balance sheet. Cash burn for 2025 was $27.7 million while net cash generated from financing sources was $19.9 million, which consisted of proceeds from a bridge note, warrants, and common stock issuance, partially offset by capital raising costs and transaction and liability payments related to Kintara. In October, TuHURA entered into a $3.0 million loan agreement, of which $1.5 million of the loan was advanced upon execution. In November, TuHURA entered into an at-the-market (ATM) facility with HC Wainwright as its sales agent, along with the filing of a Form S-3 registration statement, making available $50 million in capacity for the ATM. Following the end of the reporting period, the second and third tranches of December’s $15.6 million raise were completed. This added gross proceeds of $7 million.
Anti-VISTA (TBS-2025) Program
TuHURA closed the Kineta acquisition in June 2025, bringing the latter’s anti-VISTA asset into the fold. Now designated TBS-2025, the candidate is a VISTA-blocking immunotherapy developed to reverse immunosuppression in the tumor microenvironment (TME). It is a fully-human engineered IgG1 monoclonal antibody that was designed to bind to VISTA through a unique epitope at physiologic and acidic pH levels. The product is being developed as an intravenous infusion. Under TuHURA’s aegis, TBS-2025 is expected to be the subject of a Phase Ib/II trial in patients with relapsed/refractory (r/r) mutated nucleophosmin 1 (mutNPM1) AML.
Based on research that has demonstrated that mutated NPM1 and DNMT3A result in high expression of VISTA on the surface of leukemic blasts.[1] The presence of VISTA on these cells is believed to be the primary mechanism by which leukemic cells escape immune recognition and attack, resulting in a low treatment response rate and a short duration of response in AML.
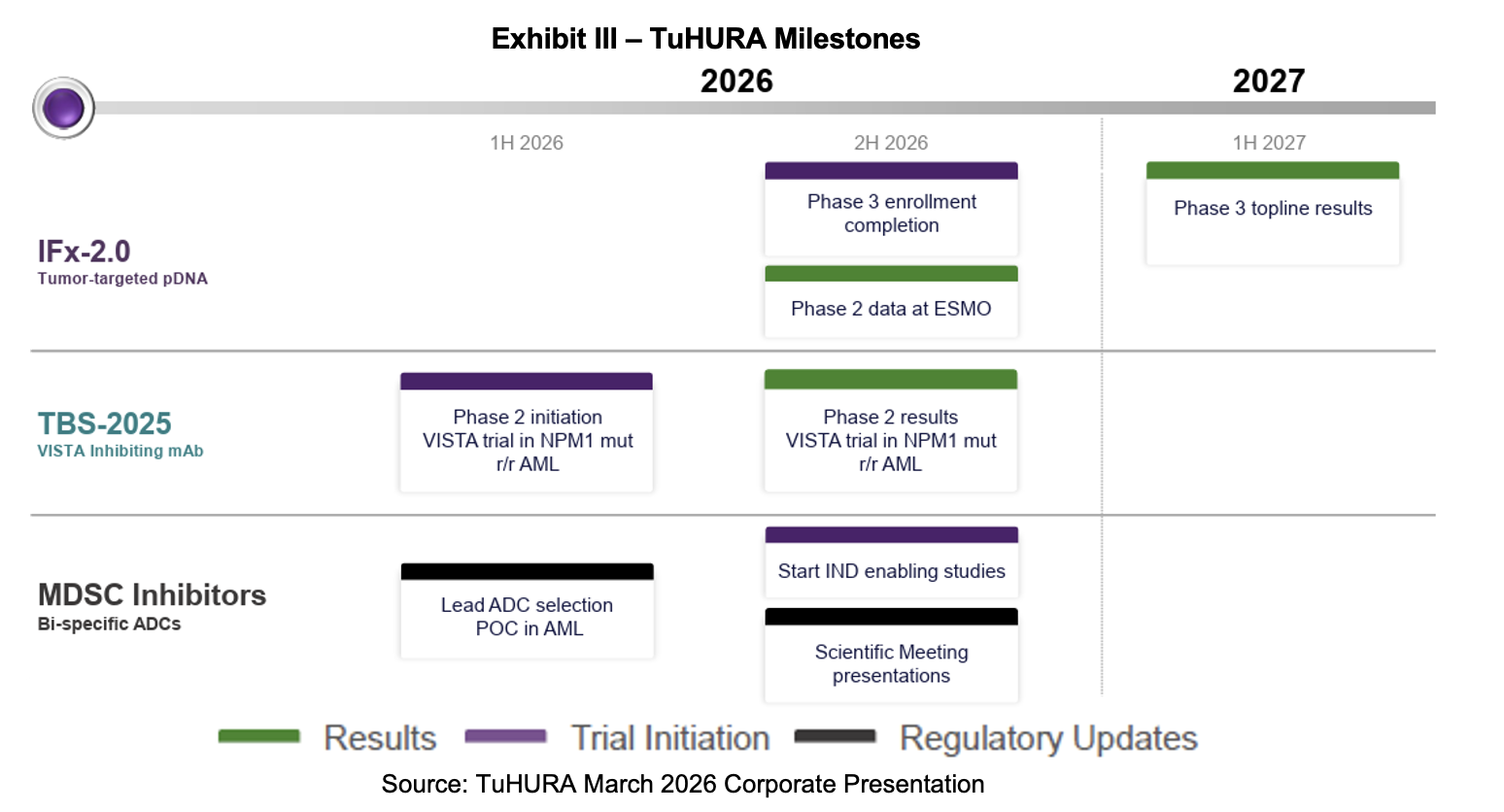
This February, TuHURA filed an Investigational New Drug (IND) Application with the FDA for TBS-2025. It submitted the document to the Division of Hematologic Malignancies for the treatment of mutNPM1 r/r AML in combination with a menin inhibitor. If cleared, the Phase Ia portion plans to enroll 14 patients and the Phase II, 26 subjects. TuHURA’s next steps with the TBS-2025 program are to determine a starting dose in an abbreviated Phase Ib trial. The subsequent Phase II trial will investigate TBS-2025 in combination with a menin inhibitor for patients with mutNPM1 r/r AML who were previously untreated with a menin inhibitor. These studies are slated to begin in 2H:26.
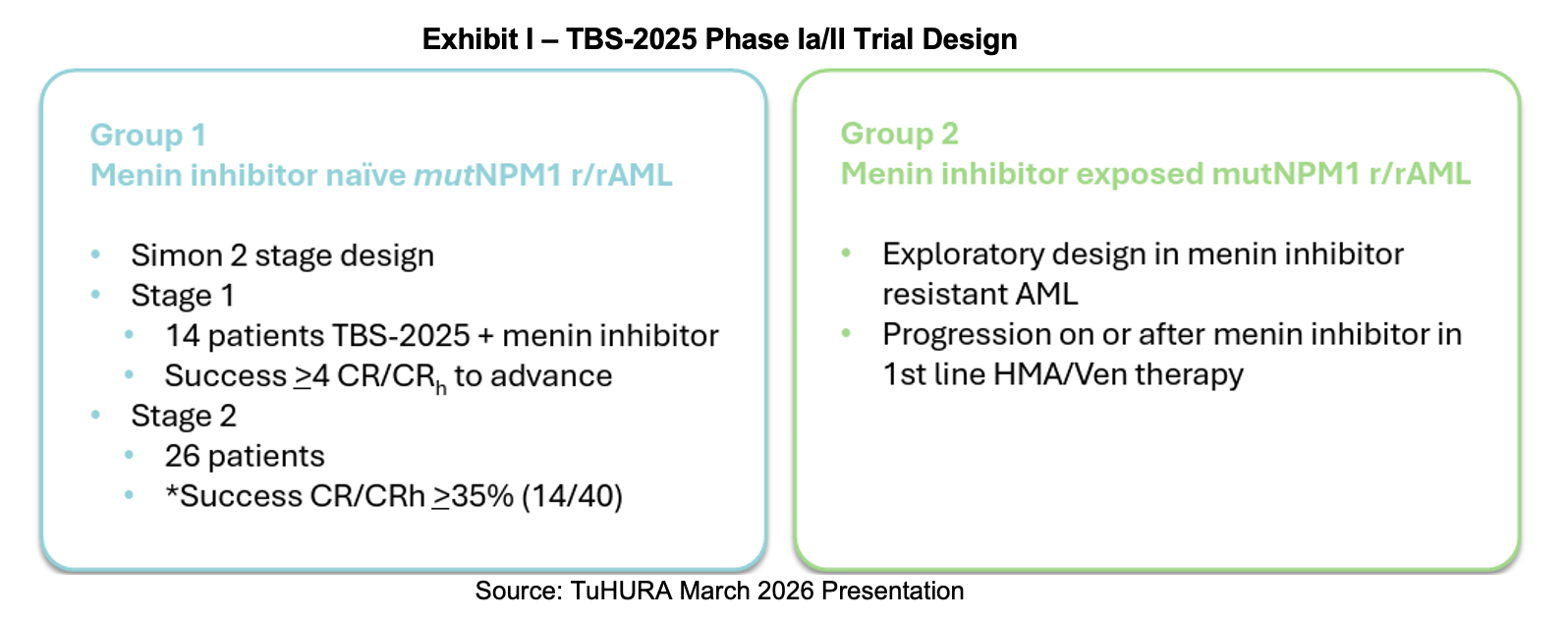
In the press release announcing the IND, Dr. Bianco pointed out that leukemogenic mutations common in AML may drive the expression of VISTA on the surface of leukemic cells, which in turn shut down the immune response. The anti-VISTA antibody’s mechanism raises the shield so the immune system can kill these cells. He continued, noting that complete response rates using menin inhibitors as monotherapy are below 25% and of short duration. Adding TBS-2025 to the treatment paradigm may markedly increase both the magnitude of response and its duration. Success in this endeavor would provide TuHURA the data it needs to seek an accelerated approval route with the FDA.
In March 2026, TuHURA announced that Dr. Craig Tendler would lead the anti-VISTA program in AML. Dr. Tendler’s first public association with TuHURA was the company’s announcement that he would join TuHURA’s board of directors in March 2025. Last month, it was announced that he would take on the responsibilities consistent with those of Chief Medical Officer (CMO) and lead the TBS-2025 program. He will continue his role on the board. A press release provided a biography for the thirty-year industry veteran, noting his tenure at Johnson & Johnson. Joining Dr. Tendler is Amanda Garofalo, who was announced as SVP of Clinical Operations on April 7th, 2026. She will assist with the development of TBS-2025 and TuHURA’s other clinical programs.
Menin Inhibitor Background
Menin inhibitors are a targeted therapy for NPM1-mutated r/r AML that work by disrupting the menin-dependent transcriptional program that leukemic cells use to maintain HOX/MEIS1 expression, which helps drive survival and differentiation block.[2] In practice, these small molecules can induce differentiation and remissions in this disease, and they are now part of the treatment landscape for this molecular subtype, with activity seen most often in heavily pretreated patients. NPM1-mutated AML is biologically dependent on menin-mediated signaling, so blocking menin can turn off an oncogenic program rather than just broadly killing dividing cells. That makes menin inhibition especially relevant in r/r mutNPM1 AML, where options are limited and targeted therapy is needed.[3],[4]
TBS-2025 may pair well with a menin inhibitor because the combination targets two different resistance layers in AML. Anti-VISTA antibodies may reverse immune suppression in the tumor microenvironment, while menin inhibitors suppress the leukemic oncogenic transcription program in mutNPM1 AML. In murine mutNPM1 AML, loss of VSIR (the gene encoding VISTA) was associated with an immune response and improved survival. This suggests VISTA blockade could help the immune system clear leukemic cells that remain after menin inhibition.[5]
Financing and Corporate Update
TuHURA announced a registered direct offering on December 9th that raised $15.6 million through the issuance of 9,462,423 shares of common stock and warrants. The purchase price for the equity shares was set at $1.65 and the exercise price for the warrants was $1.95. Warrants can be exercised six months after issue. The closing of the transaction was scheduled to occur in three tranches, with all completed by the end of February 2026.
Phase III IFx-2.0 Trial in MCC
TuHURA launched its pivotal Phase III study for its IFx-2.0 candidate in Merkel cell carcinoma (MCC) in June 2025. In the latest earnings report, TuHURA provided its latest set of milestones for the program. They include obtaining Orphan Drug Designation for IFx-2.0 in MCC in 1H:26, reporting preliminary data from the Phase Ib/IIa study of IFx-2.0 in 2H:26, and a topline readout of the Phase III study in 2H:27.
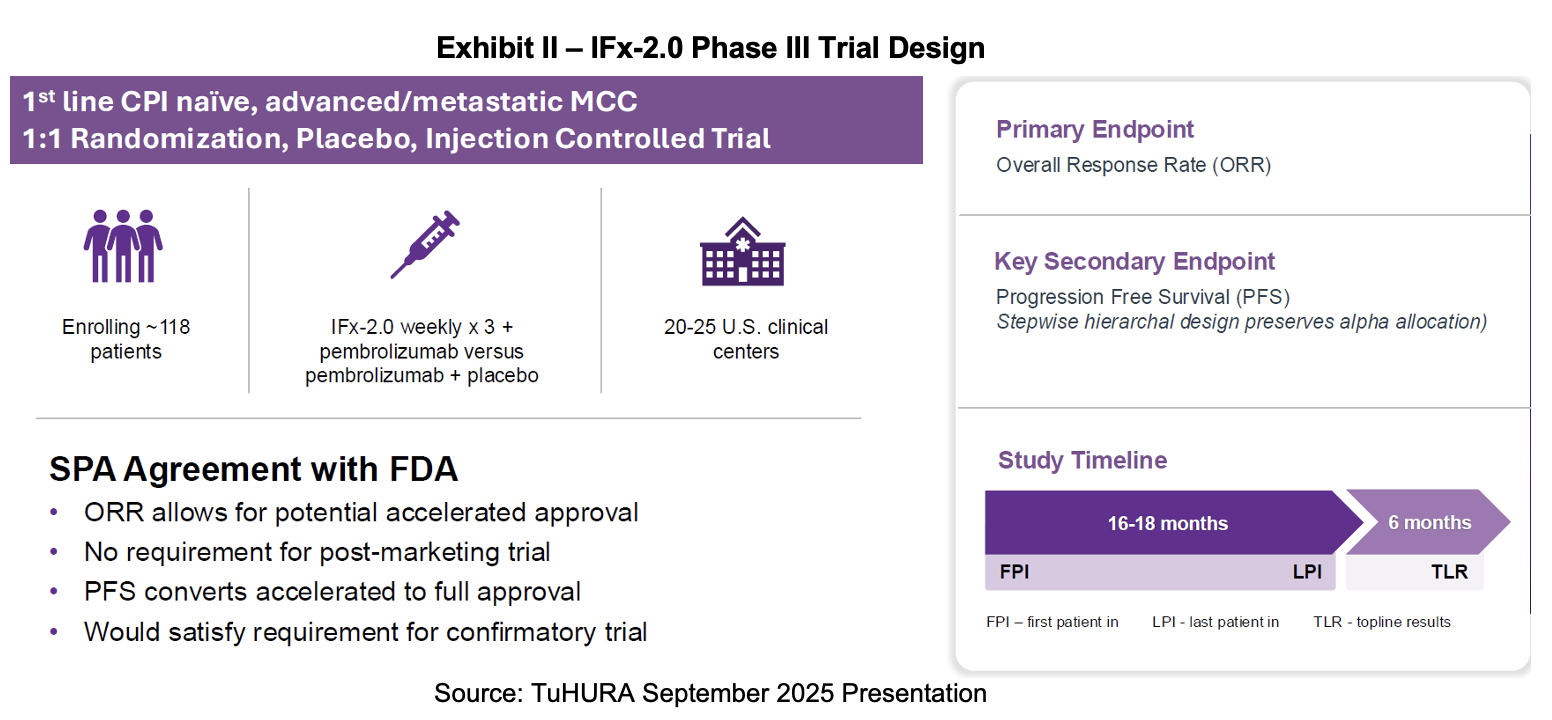
IFx-2.0 will prepare for a new drug application using the FDA’s accelerated approval program under a special protocol assessment (SPA). The trial was designed with the input of the FDA’s Oncology Center of Excellence (OCE). Accelerated approval allows the sponsor to use surrogate endpoints that predict clinical benefit. In most cases, an accelerated approval will require post-market confirmatory trials to verify the clinical benefit. However, in this case, the FDA has indicated that secondary endpoints that demonstrate clinical benefit may be used. If successfully achieved, the trial may satisfy the requirements for full approval.
Upcoming Milestones
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] NPM1 and DNA methyltransferase 3A (DNMT3A) are two of the most common mutations in AML and typically co-mutated in myelodysplasia (MDS).
[2] Differentiation block refers to a state where leukemia cells are held in an immature, rapidly dividing stage and are unable to mature into functional white blood cells.
[3] Isidori, A., Marconi, G. The role of menin inhibitors in acute myeloid leukemia. Current Opinion in Oncology. November 2025.
[4] Fiskus, W., et al. Effective Menin inhibitor-based combinations against AML with MLL rearrangement or NPM1 mutation (NPM1c). Blood Cancer Journal. January 2022.
[5]TuHURA Biosciences Pipeline. Accessed April 2026.