By David Bautz, PhD
NASDAQ: ICU
READ THE FULL ICU RESEARCH REPORT
Business Update
Presentations Highlight SAVE Registry Data and MOA Analysis
On March 30, 2026, SeaStar Medical Holding Corp. (NASDAQ: ICU) announced two poster presentations at the 31st International Conference on Advances in Critical Care Nephrology (AKI & CRRT) 2026. The posters included expanded real-world data from the SAVE Registry along with gene expression analysis on the immunomodulatory effects of the company’s selective cytopheretic device (SCD).
The Selective Cytopheretic Device (SCD) in Pediatric AKI Requiring CRRT – Eighteen-Month Post-Approval Clinical Experience from the SAVE Registry
The SCD for pediatrics (SCD-PED, QUELIMMUNE™) received FDA Humanitarian Exemption (HDE) approval in 2024 for use in critically ill children with acute kidney injury (AKI) due to sepsis requiring continuous renal replacement therapy (CRRT) and antibiotic therapy. One of the conditions of the approval included an FDA-mandated post-market surveillance registry (SAVE) to monitor safety and clinical outcomes in all treated patients. The poster presented an update on 32 patients enrolled as of Dec. 11, 2025. A total of 31/32 (97%) of the patients were on intensive mechanical ventilation, indicative of a very sick patient population. The 28- and 60-day survival were both 69% (22/32), which is consistent with the clinical trial experience of the SCD-PED in the same population.
The Selective Cytopheretic Device Induces Monocyte Phenotype Switching Towards an Anti-Inflammatory, Reparative Phenotype
In this study, whole blood from four healthy donors was circulated through an in vitro closed-loop circuit containing a miniaturized SCD. Monocytes were collected at four timepoints to analyze how gene expression changed during treatment. Following enrichment, the monocytes were subjected to single-cell RNA sequencing, which indicated that the SCD shifted the monocytes from a pro-inflammatory state toward an anti-inflammatory, tissue-reparative phenotype. Treated cells demonstrated upregulation of pathways associated with immune regulation and wound healing, while maintaining the ability to respond to inflammatory stimuli. These findings support the proposed mechanism of action of the SCD as targeted immunomodulation rather than broad immunosuppression.
Phase 3 NEUTRALIZE-AKI Trial Over 50% Enrolled
SeaStar is currently conducting the NEUTRALIZE-AKI pivotal study to assess the safety and efficacy of the SCD in adults with AKI. It is a two-arm, randomized, open label, controlled, multi-center trial that will enroll up to 339 adult patients in the ICU with AKI requiring CRRT and at least one additional organ failure across 30 clinical centers. SCD therapy will be administered for up to 10 days with a primary outcome of all-cause mortality or dialysis dependency at day 90. An overview of the trial is given below.
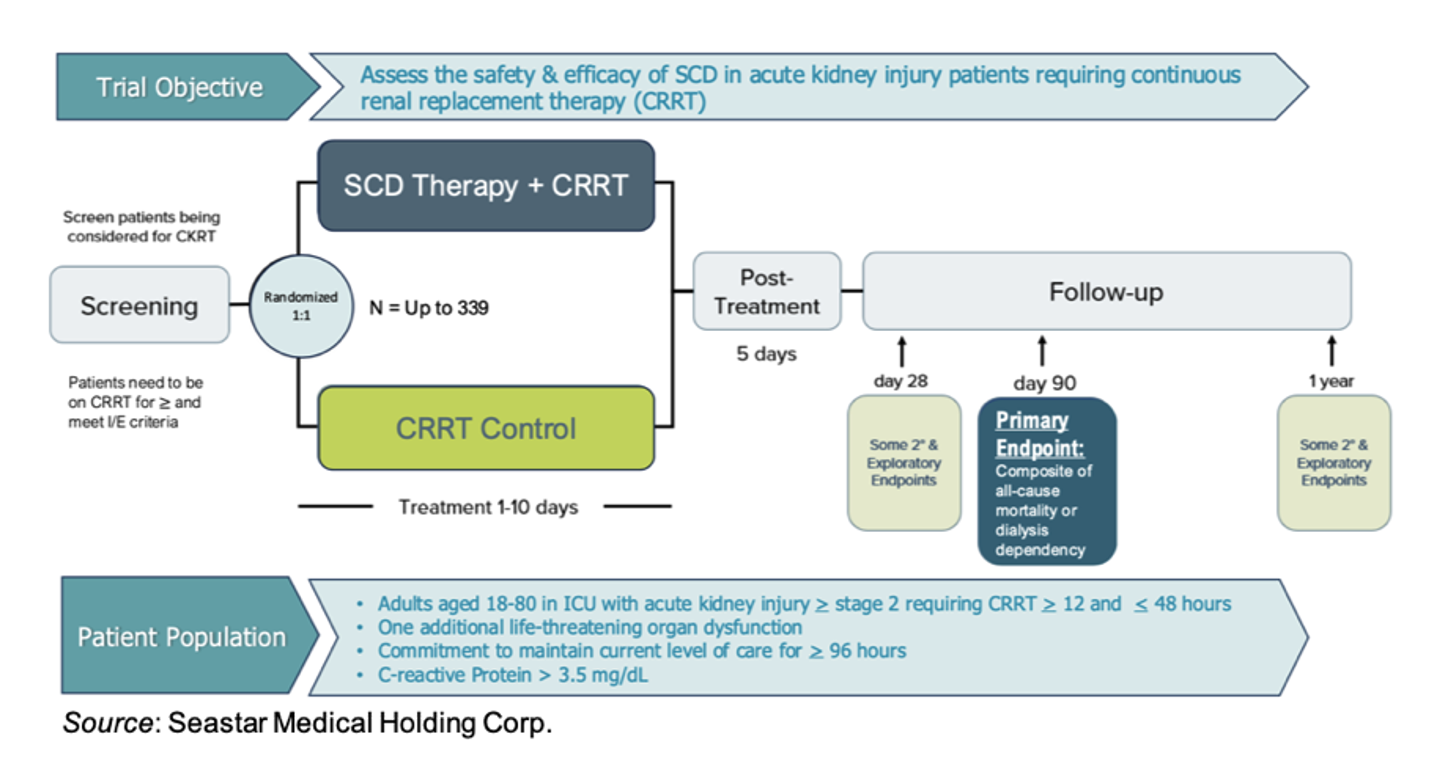
The company recently reported that 181 of 339 patients have been enrolled to date. We estimate that patient enrollment will complete near the end of 2026. SeaStar has initiated efforts for the submission of a modular PMA, which may be expedited since the company was previously granted Breakthrough Device Designation by the FDA.
Enrollment Complete in SAVE Surveillance Registry
On March 5, 2026, SeaStar announced that it completed the required enrollment in the SAVE Registry. Fifty patients were successfully enrolled, and SeaStar will be reporting the 28-day safety results from the SAVE Registry to the FDA following analysis of the data.
Financial Update
On March 25, 2026, SeaStar announced financial results for 2025. For the years ended December 31, 2025 and 2024, net revenue was approximately $1.2 million and $135,000, respectively. The increase is due to the first full year of QUELIMMUNE sales compared to only half a year in 2024. R&D expenses for 2025 were $7.5 million compared to $9.1 million in 2024. The decrease in R&D expenses was primarily due to declines in personnel costs, consulting expenses, pre-clinical costs, and external clinical services costs, partially offset by increased clinical trial site costs, supply costs, and medical affairs expenditures. G&A expenses in 2025 were $5.9 million compared to $8.9 million in 2024. The decrease was primarily due to reduced personnel costs and director, legal, accounting, consulting related fees, licensing costs, and a one-time settlement cost with a former distributor in 2024, partially offset by a one-time due diligence fee paid to a third-party financial institution.
SeaStar exited 2025 with $12.0 million in cash and cash equivalents. As of March 24, 2026, the company had approximately 4.0 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of 6.7 million.
Conclusion
With the SAVE Registry fully enrolled and the recent presentation showcasing how the real-world experience with QUELIMMUNE is matching the clinical experience, we anticipate an uptick in the number of centers onboarding the SCD for use in pediatric patients. In support of this, management has indicated a goal for an additional 15 pediatric centers to be using QUELIMMUNE by the end of the year. The company is continuing to steadily enroll patients into the NEUTRALIZE-AKI trial, and we expect enrollment concluding near the end of 2026. With no changes to our model, our valuation remains at $12 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.