By David Bautz, PhD
NASDAQ: IMNN
READ THE FULL IMNN RESEARCH REPORT
Business Update
Updated OVATION 2 Data Shows Continued Improvement in OS
On March 25, 2026, Imunon, Inc. (NASDAQ: IMNN) announced updated data from the Phase 2 OVATION 2 trial of IMNN-001 in women with newly diagnosed ovarian cancer. The OVATION 2 trial evaluated IMNN-001 in combination with standard of care (SoC) neoadjuvant and adjuvant chemotherapy in 112 women with newly diagnosed advanced ovarian cancer. Previously, the company had reported a 11.1-month increase in overall survival (OS) (40.5 vs. 29.4 months) in the IMNN-001 treatment arm compared to SoC chemotherapy alone. The updated data shows a median 14.7-month increase in OS (45.1 vs. 30.4 months) in women in the IMNN-001 treatment arm compared to SoC chemotherapy. In addition, for women being treated with IMNN-001, SoC chemotherapy, and poly ADP-ribose polymerase (PARP) inhibitors, the increase in median OS is now 24.2 months (65.6 vs. 41.4 months) compared to SoC chemotherapy and PARP inhibitors.
These results continue to show the positive impact that IMNN-001 is having on patients with newly diagnosed advanced ovarian cancer. There have been few notable advancements in the SoC for ovarian cancer in the past 30 years, thus the now 14.7-month increase in OS, if it can replicated in the ongoing Phase 3 OVATION 3 trial, could lead to a paradigm shift in how these patients are treated. Lastly, the increase in OS was accomplished with a favorable safety and tolerability profile and is another reason why IMNN-001 has received such a positive response from the medical community.
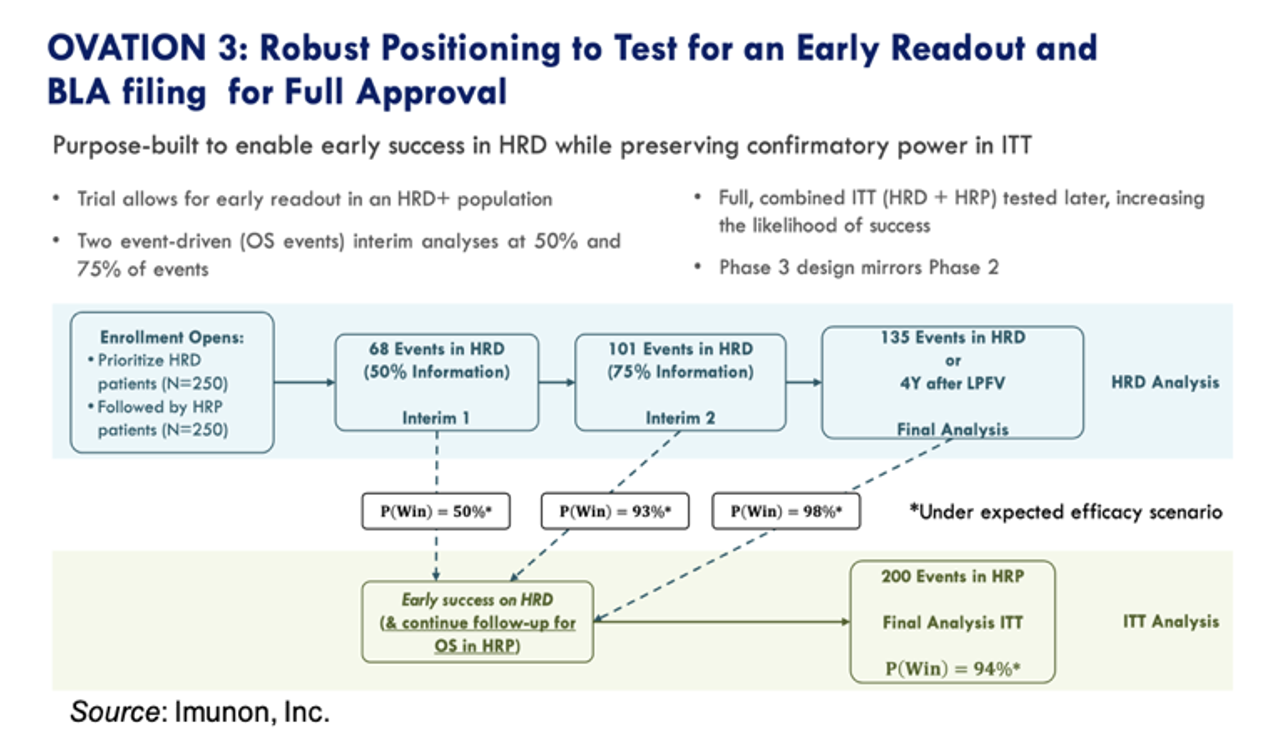
OVATION 3 Trial Enrollment Continues
Imunon is continuing to enroll the Phase 3 OVATION 3 trial of IMNN-001 in women with newly diagnosed advanced ovarian cancer. The trial has been designed to prioritize the enrollment of women positive for homologous recombination deficiency (HRD+) and has two interim analyses at 50% and 75% of events. The trial has 98% power to detect a statistically significant increase in overall survival in the HRD+ population, with a probability for stopping early for success at the interim readout of >90%.
Conclusion
The increase in OS for the OVATION 2 trial adds to the already robust data set that Imunon has accumulated on IMNN-001 showing its positive impact in the treatment of advanced ovarian cancer. The company is continuing its full focus on the OVATION 3 and anticipates having the trial approximately 20% enrolled within the next year. We look forward to additional updates from the company this year on additional trial sites being opened for the OVATION 3 trial, which may increase the rate of enrollment further. With no changes to our model, our valuation remains at $33 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.