By John Vandermosten, CFA
NASDAQ:LNTH
Lantheus Reports 2025 Financial and Operational Results
Lantheus Holdings, Inc. (NASDAQ:LNTH) reported financial results yesterday. Revenue was $407 million in the fourth quarter and $1.54 billion for the full year; adjusted fully diluted earnings were $1.67 and $6.08 per share. This compares to November’s guidance, which called for full year revenues in the range of $1.49 to $1.51 billion and EPS of $5.50 to $5.65. The better-than-guided results came from less severe declines in Pylarify and Definity sales and lower than expected expenses, especially for research and development. At year-end 2025, cash and equivalents were $359 million compared to $913 million at the end of 2024. Cash generation from Lantheus’ portfolio of assets was offset by payments of almost $630 million for the acquisitions of Evergreen Theragnostics and Life Molecular Imaging, along with $300 million in stock repurchases.

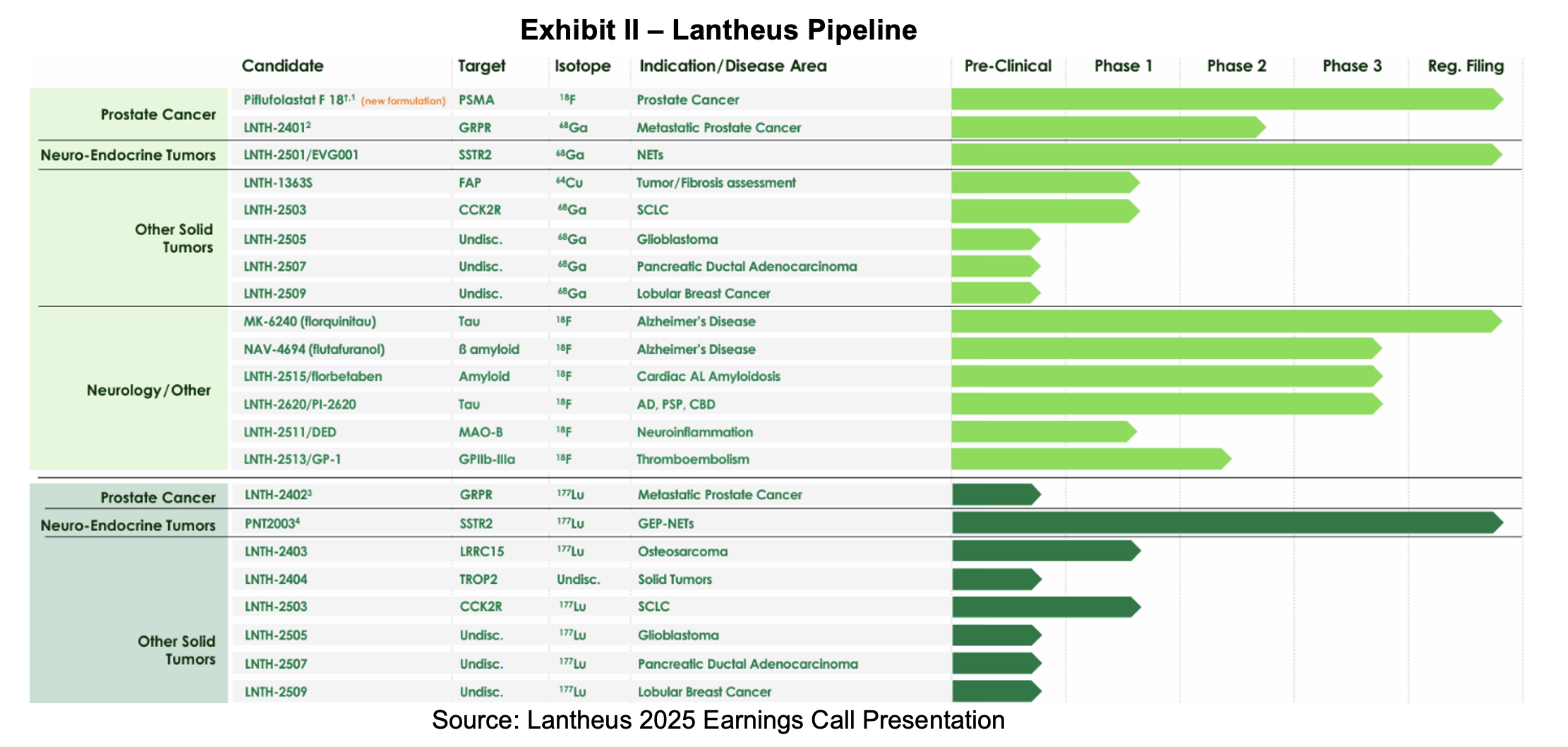
At the beginning of the year, prior Chairperson of the Board and former CEO, Mary Anne Heino, returned as the chief executive following the retirement of Brian Markison. Her return ushers in a new strategic focus to prioritize investment in the development and commercialization of innovative PET radiodiagnostics, alongside a decision to pursue value‑maximizing alternatives for radiotherapeutic assets to support long‑term growth. We interpret this to mean Lantheus will prune some early-stage therapeutic assets. Since the third quarter update, the company has divested its SPECT business to SHINE Technologies, expanded its PET portfolio with the addition of the NeuraCeq franchise and submitted three assets to the FDA for which target action dates have been assigned.
The company provided 2026 revenue and earnings guidance. Revenues for 2026 are expected to fall in the range of $1.40 to $1.45 billion and adjusted, fully diluted earnings per share are forecast to be between $5.00 and $5.25 per share. Using the company-provided and adjusted baseline revenues for 2025 of $1,424 million, which reflects the SPECT divestiture and subtracts a $6 million milestone, this represents topline growth ranging from -1.7% to +1.8%. EPS at the midpoint is expected to be down 11% vs. 2025’s adjusted baseline of $5.75.
See below for links to key materials related to the report of 4Q:25 and 2025 full year results:
2025 Financial and Operational Results
Lantheus’ earnings release was followed by a conference call which included CEO Mary Anne Heino, CFO Bob Marshall and Chief Commercial Officer Amanda Morgan. The team highlighted anticipated milestones and provided additional detail regarding the new focus on the diagnostic portfolio. A financial comparison for 2025 follows.
For the year ending December 31st, 2025, relative to the prior year:
- Net sales were $1.54 billion, up 0.5% from $1.53 billion. The increase was driven by the addition of NeuraCeq revenue, an increase in revenue from Strategic Partnerships, Other Precision Diagnostics, and Definity. Revenue declines of 6.5% for Pylarify and 9.1% for TechneLite partially offset those increases;
- Cost of goods sold rose 10% to $600 million and gross margin declined to 58.1% from 63.5% due to lower prices for Pylarify;[1]
- Sales and marketing expenses were $179 million, up slightly from $178 million, driven by higher costs related to NeuraCeq sales, the provision of contract manufacturing services, and employee-related costs related to the acquisitions of Life Molecular and Evergreen. These costs were offset by lower brand campaign spending for Pylarify, the cessation of launch support for PNT2002, and a decrease in third-party vendor and other marketing expenditures;
- General and administrative expenses were $275 million vs. $194 million, increasing 42%. The change was attributable to the acquisitions of Life Molecular and Evergreen. Higher legal fees also contributed, which were related to intellectual property matters and costs related to the SPECT divestiture;
- Research and development expenses were $177 million, up 5% from $168 million. The increase was related to a payment to AstraZeneca to reduce future royalty obligations for NAV-4694, milestones owed related to RM2, and additional costs from the Life Molecular and Evergreen acquisitions. This was offset by the absence of payments to partners Life Molecular, Perspective, and Radiopharm Theranostics, which occurred in 2024. R&D was 12% of revenues;
- Other items generated net income of $22.7 million vs. loss of $6.3 million due to a larger unrealized loss in equity securities for 2024;
- Income tax expense of $80.2 million represents a 25.6% tax rate, with state income taxes, nondeductible stock compensation, and non-deductible acquisition-related costs contributing to the difference between the reported rate and the U.S. statutory rate of 21%. This was partially offset by tax credits;
- GAAP net income was $234 million or $3.41 per diluted share. Adjusted net income as presented by Lantheus was $416 million or $6.08 per diluted share. The difference is explained in part by the removal of stock and incentive plan compensation, strategic collaboration and license costs, amortization of acquired intangible assets, acquisition, integration, and divestiture-related costs, and the income tax effect of these non-GAAP adjustments.
On December 31st, 2025, Lantheus held $359 million in cash and equivalents compared to $913 million at the end of 2024. Free cash flow for 2025 was $354 million vs. $493 million in 2024. Lower net income contributed to the change. Material items contributing to the decline in the cash balance over 2025 include a $300 million repurchase of Lantheus stock and $629 million in acquisition payments.
Corporate Milestones
- Close of Evergreen Theragnostics acquisition – April 2025
- Life Molecular Imaging acquisition completed – July 2025
- Initiate Phase I/IIa study for LNTH-2401/-2402 (RM2) – 2H:25
- Completion of SPECT business unit transfer to SHINE – year end 2025
- Reformulated Pylarify target action date – March 6th, 2026
- Target action date for LNTH-2501 (Octevy) in SSTR+ NETs – March 29th, 2026
- Launch six additional PET Manufacturing Facility (PMF) sites in support of NeuraCeq - 2026
- Target action date for MK-6240 – August 13th, 2026
- MK-6240 market launch – 2H:26
- FDA approval and launch of PNT2003 after Hatch-Waxman resolution – 2026
- Launch of reformulated Pylarify – 4Q:26
Summary
Lantheus reports better than guided revenues and earnings. During 2025, Pylarify suffered price and volume pressures offset by the addition of NeuraCeq revenues and modest growth from Definity and Other Precision Diagnostics. Management set 2026 guidance, which reflects the divestiture of the SPECT business and a full year of NeuraCeq revenues. Lantheus has four assets that may be approved or cleared for sale this year. However, we do not expect them to materially contribute to the topline until 2027 due to market preparations required before commercialization. Guidance calls for lower earnings in 2026 while Pylarify laps 340B price impacts and newly approved assets begin to contribute. Despite the decline, the neurology imaging portfolio is attractive long-term and leveraged to growth in therapies targeting neurodegenerative disease. Cash generation is a strength which has been used to invest in the pipeline and repurchase stock.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] We calculate gross margin as 1-COGS/(revenues from Pylarify, Definity, TechneLite, NeuraCeq and Other Precision Diagnostics) as reported in company filings. We exclude revenues from Strategic Partnerships and Other from the calculation.