By David Bautz, PhD
NASDAQ: MTVA
READ THE FULL MTVA RESEARCH REPORT
Business Update
Phase 1 Part 3 Trial of DA-1726 to Initiate in April 2026
On March 18, 2026, MetaVia, Inc. (NASDAQ: MTVA) announced the approval of the clinical trial plan by the Institutional Review Board (IRB) at Clinical Pharmacology of Miami for the company’s Phase 1 Part 3 study of DA-1726, a dual oxyntomodulin analog that functions as a dual agonist of the GLP-1 and glucagon (GCGR) receptors. The approval allows for the initiation of the trial, which is expected to begin in April 2026.
The Phase 1 Part 3 trial is planning to enroll 40 obese but otherwise healthy individuals across two parts, with 20 subjects per part randomized 4:1 (16 active; 4 placebo). Part 3A will evaluate a one-step titration regimen with 16 mg DA-1726 for four weeks, followed by 48 mg DA-1726 for 12 weeks. Part 3B will evaluate a two-step titration regimen, with 16 mg DA-1726 for four weeks, 32 mg DA-1726 for four weeks, and 64 mg DA-1726 for eight weeks. The primary endpoints include monitoring adverse events (AEs), serious adverse events (SAEs), treatment-emergent adverse events (TEAEs), and AEs leading to discontinuation. Secondary and exploratory endpoints include pharmacokinetic (PK) profiling and evaluation of metabolic, glycemic, lipid, and body composition measures, including weight, waist circumference, and body mass index (BMI). We anticipate topline results in the fourth quarter of 2026.
Positive Results for 8-week 48 mg Cohort in Phase 1b Trial of DA-1726
In January 2026, MetaVia announced positive and statistically significant results from the 8-week 48 mg cohort from the Phase 1b trial of DA-1726. The results showed a robust reduction in weight and waist circumference, along with large improvements in glucose control and reductions in liver stiffness. The drug also continued to be safe and well-tolerated.
The following figure on the left shows the weight loss seen thus far for the different cohorts from the Phase 1 study that were treated for four weeks. There is a clear dose response starting from the 8mg dose through the 48 mg dose. The figure on the right compares the 4-week data to the 8-week data for the 48 mg cohort.
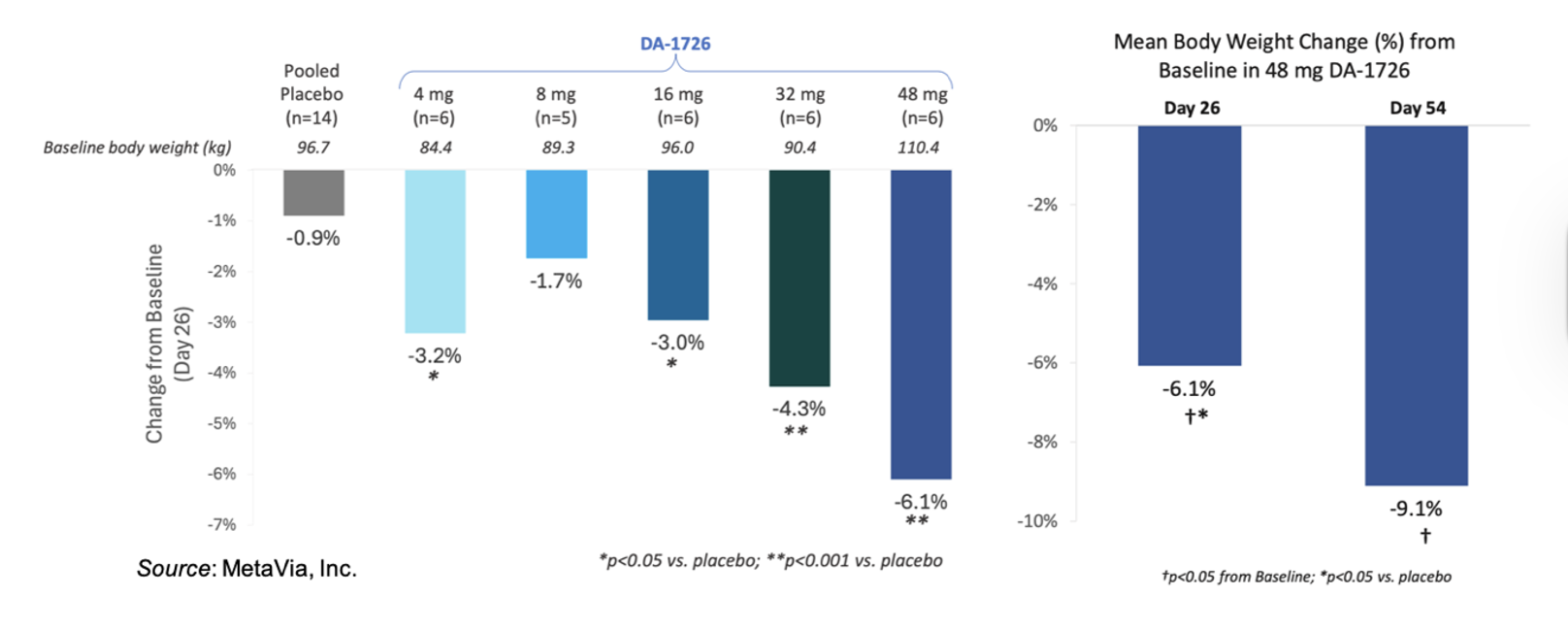
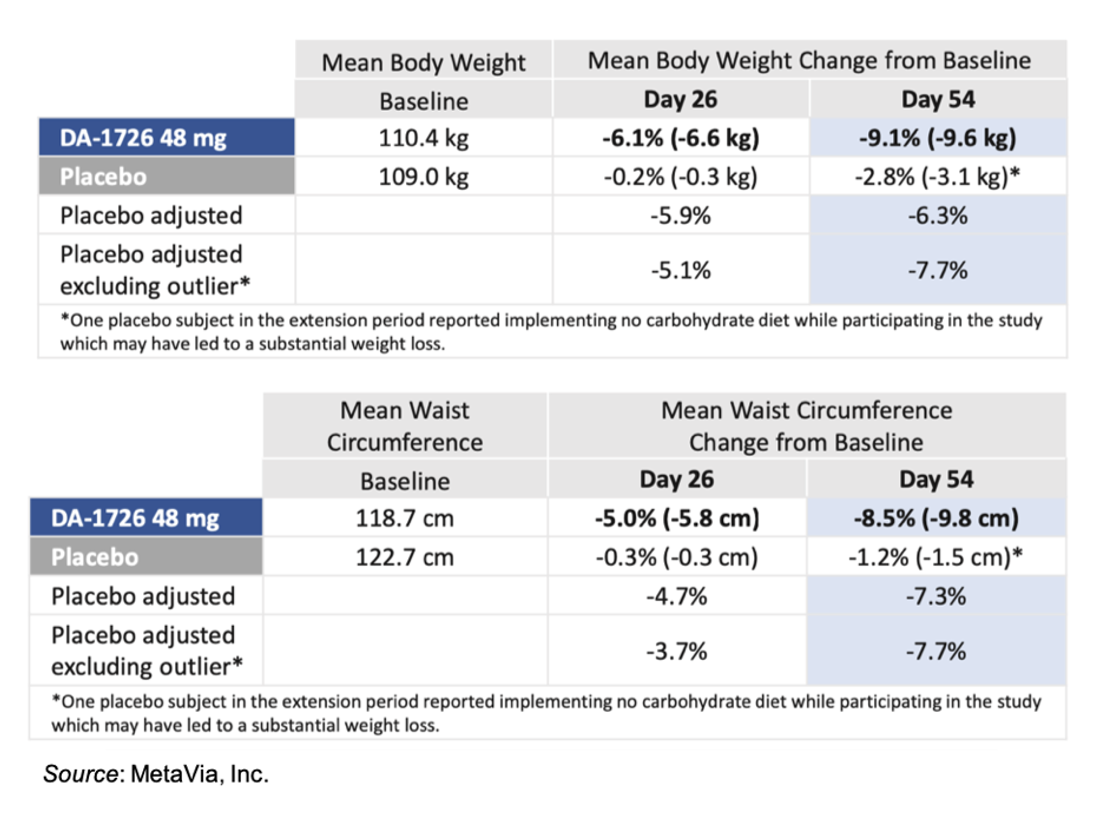
The following tables give the placebo-adjusted results for body weight and waist circumference for the 48 mg cohort. One of the subjects in the placebo group reported implementing a “no carbohydrate” diet while participating in the study, which may have led to a substantial weight loss. Thus, the company performed a placebo-adjusted analysis with and without that individual. For waist circumference, the placebo outlier did not have a substantial reduction in waist circumference, which further supports DA-1726’s direct effect on waist circumference.
DA-1726 also showed positive effects on glucose control and liver health. Fasting glucose decreased from a mean baseline of 105.3 mg/dL to a mean of 93 mg/dL after eight weeks of treatment. Mean HbA1c was also controlled in this non-diabetic population, with a mean change of -0.22% after eight weeks of DA-1726 therapy. One pre-diabetic patient began the study with an HbA1c of 6% at baseline, and that was reduced to 5.5% after eight weeks of treatment. For liver health, the mean VCTE (FibroScan®) was 5.9 kPa at baseline, and that decreased to a mean 4.5 kPa after eight weeks of treatment with DA-1726. This is in contrast to placebo-treated patients that had a mean baseline VCTE of 5.1 kPa that increased to a mean 6 kPa after eight weeks. These results show that DA-1726 is having a positive effect on liver inflammation and stiffness.
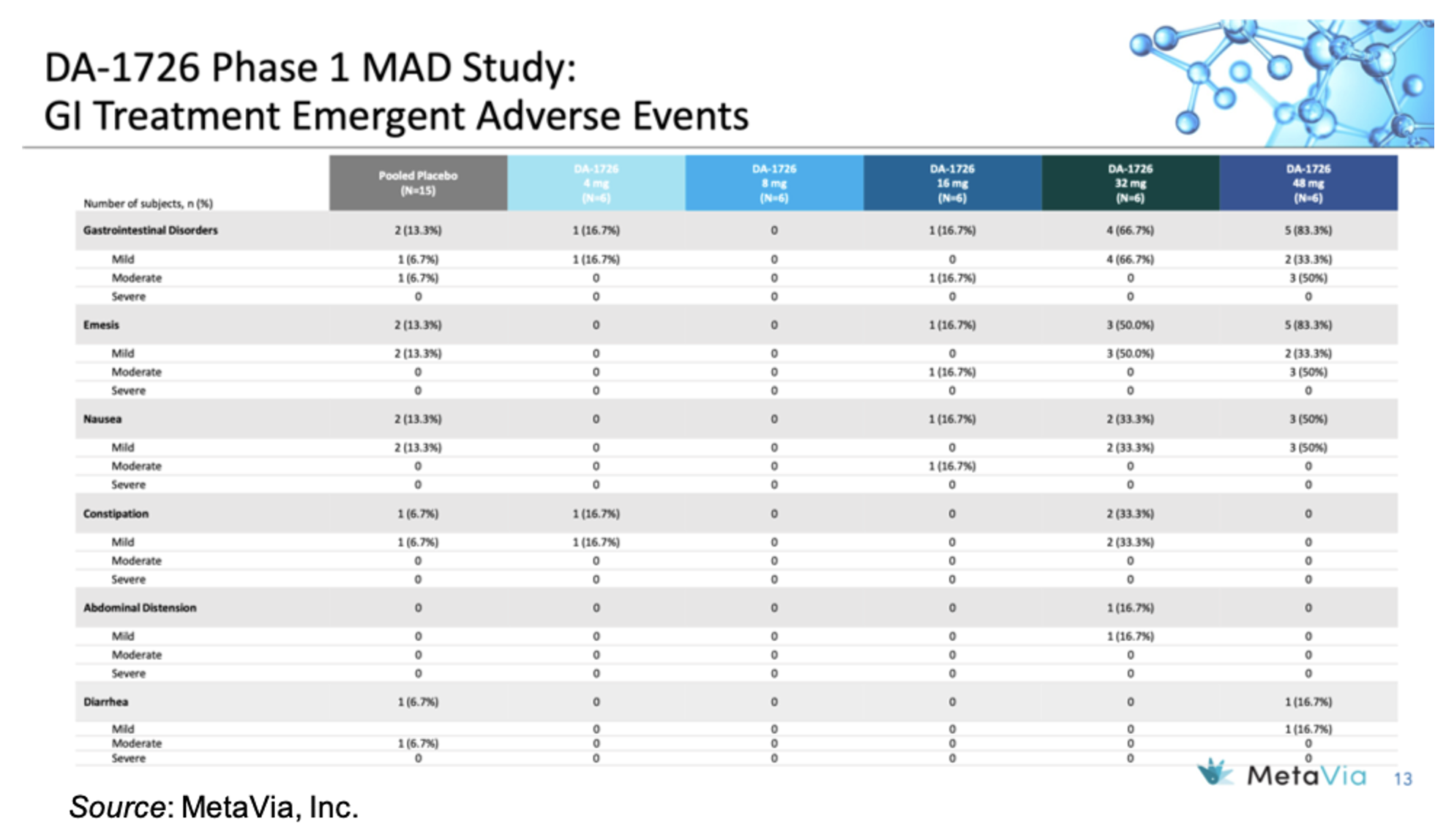
In regards to safety and tolerability, there was only one DA-1726-treated subject who discontinued from the study, which was due to being in the hospital as the result of a car accident while riding in a passenger seat. The following table gives an overview of the GI treatment emergent adverse events, which show an increase in mild to moderate events in the 48 mg cohort. However, there was no titration with that cohort, thus we believe the incidence of GI events will likely decrease in Part 3 of the trial when titration is introduced.
DA-1726 Competitive Analysis
At this point, we believe the most informative comparisons of DA-1726’s early data are with other weight loss drugs that also target the glucagon receptor. The following table provides efficacy and tolerability data for different glucagon receptor-targeting therapies and tirzepatide, which is a GLP-1R/GIPR co-agonist. While a direct comparison between the various compounds is not possible due to differences in titration, inclusion/exclusion criteria, and study length, this table is provided as a means to show potential trends in the data. In addition, the competitor compounds are further along in development than DA-1726, but their results give a good indication of the type of activity DA-1726 will need to show in longer term trials. Lastly, while AE rates for nausea, vomiting, diarrhea are provided, investors should keep in mind that DA-1726 did not titrate up to 48 mg while the competitor compounds results were obtained following titration to the target dose.
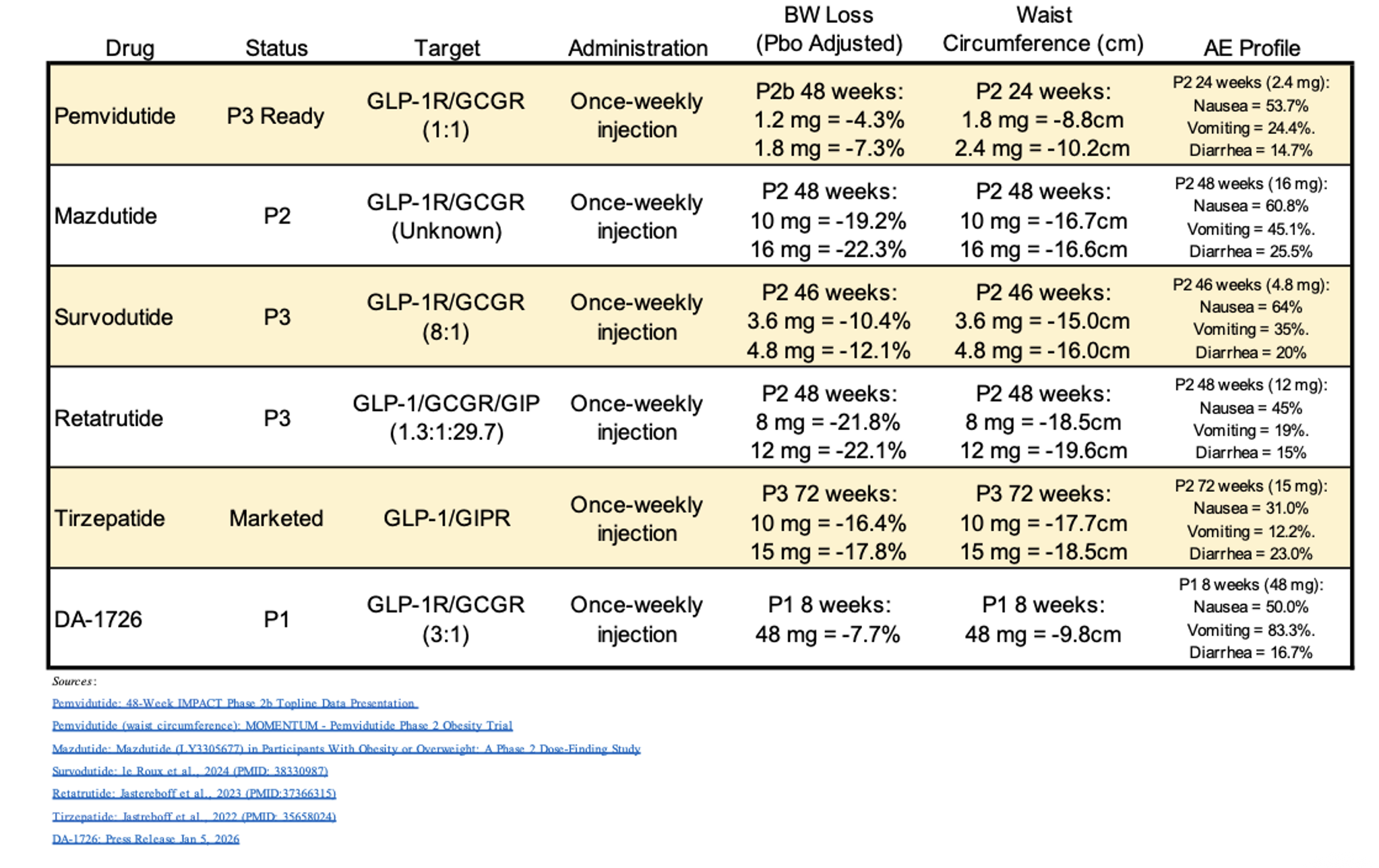
Based upon the early data for DA-1726, we believe the drug could potentially match or even exceed the efficacy results for the other glucagon-targeted therapies in a long-term trial, and if the Phase 1b Part 3 titration results show decreased GI AEs (which is typical for this class of drugs that implement a titration strategy), DA-1726 could represent a fully differentiated asset with a best-in-class efficacy and tolerability profile.
Financial Update
On March 26, 2026, MetaVia announced financial results for 2025. As expected, the company did not report any revenues in the year ending December 31, 2025. R&D expenses in 2025 were approximately $6.8 million compared to approximately $21.6 million in 2024. The decrease was primarily attributed to decreased R&D expenses related to vanoglipel (DA-1241) product development, decreased direct R&D expenses related to DA-1726 product development, and lower direct other R&D costs. G&A expenses in 2025 were approximately $6.9 million compared to approximately $7.3 million in 2024. The decrease was primarily attributable to lower consulting expenses, insurance, and other G&A expenses.
As of December 31, 2025, MetaVia had approximately $10.3 million in cash and cash equivalents. Subsequent to the end of the year, MetaVia closed a $9.3 million underwritten public offering. We estimate that the company currently has sufficient capital to fund operations into the fourth quarter of 2026. As of March 20, 2026, MetaVia had approximately 5.1 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 14.8 million.
Conclusion
We’re glad to see that MetaVia has gotten IRB approval for the Phase 1 Part 3 trial, which we anticipate initiating next month. DA-1726 has shown signs of potential best-in-class efficacy with a favorable tolerability and safety profile, thus we will be particularly interested in the tolerability profile in this trial with the titration steps. The topline results are expected in the fourth quarter of 2026, and that will mark the end of the Phase 1b trial, with the results used to design Phase 2 studies. With the continued weakness in the stock we have had to increase potential dilution for future financings into our model, which has lowered our valuation to $30 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.