By John Vandermosten, CFA
NYSE: PLX
READ THE FULL PLX RESEARCH REPORT
While Protalix BioTherapeutics, Inc. (NYSE: PLX) has been quiet since the last financial update in mid-March, the company has been active with its development portfolio while continuing to support its commercialization partners for Elfabrio and Elelyso. The Phase II RELEASE trial, which is evaluating PRX-115 in gout, continues to enroll patients and expects topline results in 2H:27. PRX-119 persists in its preclinical work as Protalix narrows the indication in NETs-related diseases, and work continues with Secarna to identify indications in renal rare disease using RNA-based therapeutics. Total revenues for 1Q:26 were ahead of our estimates due to the earlier recognition of the milestone related to European approval of every-four-week dosing for Elfabrio. Elelyso revenues were ahead, but Elfabrio sales were behind our estimates.
2026 guidance of $78 to $83 million was reaffirmed. This includes the now-recognized Chiesi milestone. By product, Protalix expects Elfabrio revenues of $30 to $35 million and Elelyso revenues of $20 to $23 million.
Protalix’s financial and operational results were reported in a May 13th, 2026, press release and Form 10-Q filing. The reports were followed by a conference call, which provided further updates. Total revenues were up 234% due to the recognition of the Chiesi milestone, and net income was $18.3 million or $0.22 per share. Further discussion of financial results for the year ending March 31st, 2026, is provided below, compared to the same prior year period:
- Revenues were $33.8 million, up 234% from $10.1 million, attributable to the recognition of the Chiesi milestone related to European Commission approval of Elfabrio for every-four-week dosing. Elelyso sales to Pfizer fell 79% to $1.5 million, and to Brazil fell 19% to $2.5 million. The decline was attributable to shipment timing for both partners. Elfabrio sales of $3.5 million compared to zero sales in the prior year. License and R&D services revenues increased to $26.3 million from $118,000 related to the abovementioned milestone;
- Cost of revenue was $4.1 million vs. $8.2 million. Product gross margin increased to 44.4% from 18.2% due to product mix;
- Research and development expenses increased 56% to $5.4 million from $3.5 million. The increase was due to higher expenses across the board, including salary, subcontractor expenses, materials, and other expenses. The rise is due to greater activity related to the initiation of the RELEASE study;
- Selling, general, and administrative expenses rose 17% to $3.1 million versus $2.6 million due to higher salaries;
- Net financial expense was $5,000 compared to a net financial expense of $413,000. The change in this line item was due to exchange rate fluctuations;
- Income tax expense of $2.8 million vs. a tax benefit of $113,000;
- Net income was $18.3 million or $0.22 per share, versus net loss of $3.6 million or $0.05 per share.
The cash and equivalents balance on March 31st, 2026, totaled $51.1 million versus $30.3 million at the end of 2025. During the first quarter, Protalix’s cash from operations was $21.2 million, derived primarily from net income and from a decrease in accounts receivable. Cash from financing was minimal. We believe that Protalix holds sufficient cash to support operations for the foreseeable future.
PRX-115 Phase II Trial Enrolling
Protalix is enrolling subjects in its Phase II study for PRX-115. Under the acronym RELEASE, the trial is entitled A Study to Investigate the Clinical Effect and the Safety of PRX-115 Infused Intravenously at Different Dosing Regimens, With and Without Methotrexate, Versus Placebo in Adult Gout Patients. It is listed on clinicaltrials.gov under the designator NCT07280156. The multicenter, randomized, double-blind, placebo-controlled study will assess the efficacy, safety, and dosing regimen selection of multiple IV infusions of PRX-115 over 24 weeks, with or without methotrexate (MTX), versus the respective placebos in adult patients with gout. One site is active in Miami, Florida.
Protalix anticipates that RELEASE topline results will be available in 2H:27.
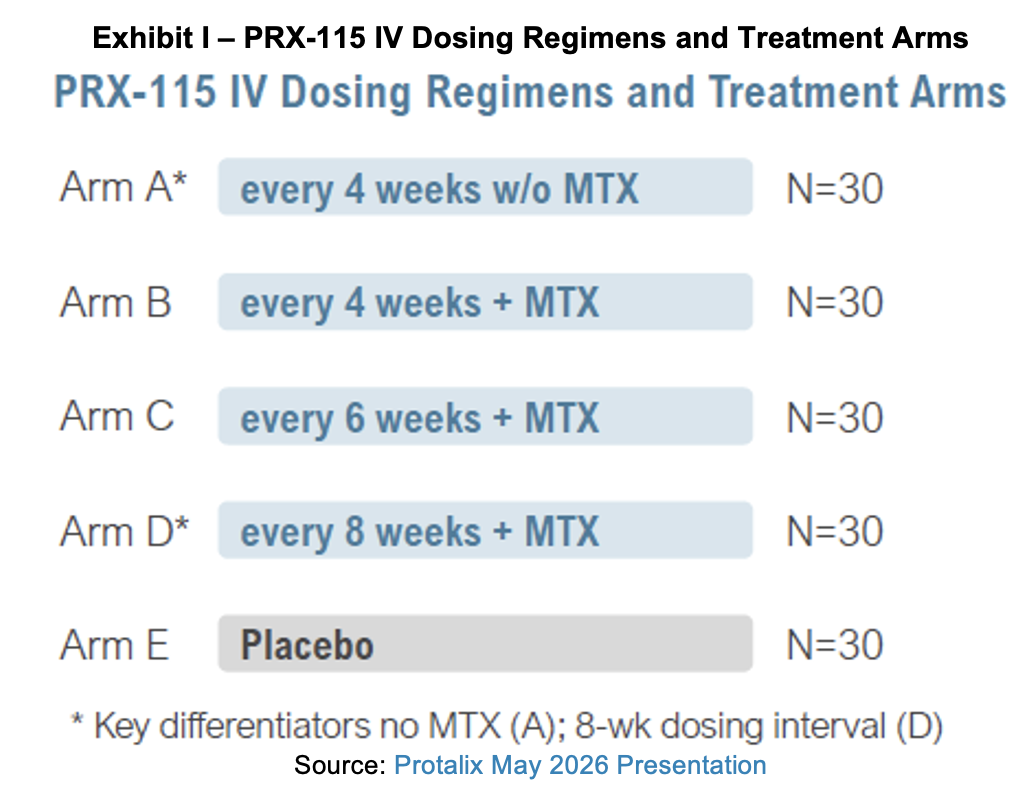
The primary endpoint is the proportion of patients who achieve a reduction in serum uric acid to less than 6.0 mg/dL for at least 80% of the time during month six. Secondary endpoints will measure additional uric acid parameters, safety, and immunogenicity. The study will additionally record tophi, flares, swollen and tender joints, quality of life, and pharmacokinetics.
Participants will receive PRX-115 by intravenous (IV) infusions according to different treatment schedules, with and without MTX. MTX itself does not reduce uric acid levels or manage flares directly, but instead is used in combination with treatments that may activate antibodies and an immune response. In a study evaluating pegloticase, MTX was used to reduce antidrug antibody development and infusion reactions as well as improve efficacy. The use of MTX with pegloticase improved the response rate for gout patients compared to the use of pegloticase alone.[1]
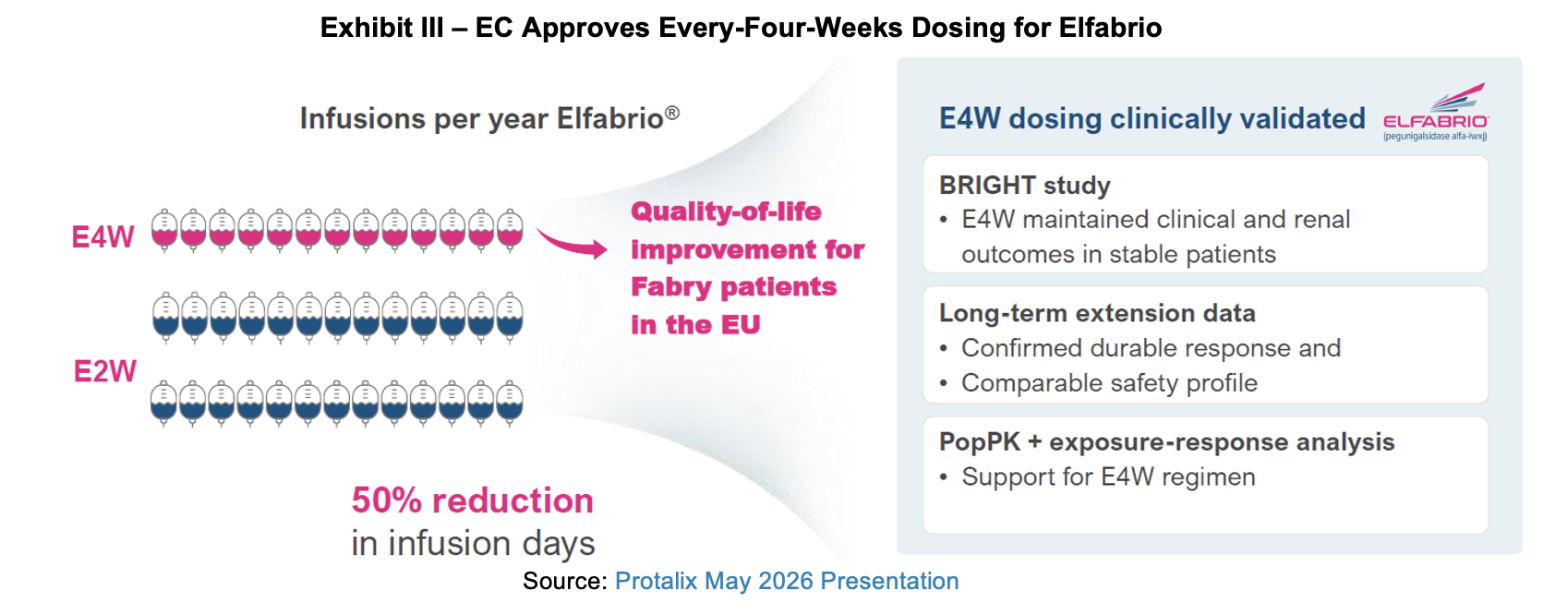
European Commission Approval of Elfabrio Four Week Dosing
On March 9th, Protalix announced that the European Commission (EC) approved the 2.0 mg/kg every-four-weeks dosing regimen for Elfabrio in Fabry disease. The addressable population is adult patients who are stable on enzyme replacement therapy (ERT). Protalix has received a $25 million milestone payment from Chiesi following the approval.
While the four-week dosing regimen has been approved, it will take time for patients to make the shift. There are country-specific logistics and regulatory requirements that must be satisfied before patients and providers can make the change.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] Botson, J.K., et al. A Randomized, Placebo‐Controlled Study of Methotrexate to Increase Response Rates in Patients with Uncontrolled Gout Receiving Pegloticase: Primary Efficacy and Safety Findings. Arthritis Rheumatology. December 2022.