By David Bautz, PhD
NASDAQ:SNGX
READ THE FULL SNGX RESEARCH REPORT
Business Update
Important Readouts in 2026 for Phase 3 CTCL Study
Soligenix, Inc. (NASDAQ:SNGX) is currently conducting the Phase 3 FLASH2 Trial of HyBryte™ (SGX301 or synthetic hypericin) in the treatment of cutaneous T cell lymphoma (CTCL). As of the latest update, the company has enrolled 66 of the planned 80 patients. The FLASH2 trial is very similar in design to the successful Phase 3 FLASH trial, as shown in the following figure, which provides a comparison between the two studies. One key difference between the trials is that in the FLASH trial patients were treated for three cycles of six-weeks each, with a two-week break in between cycles and the primary efficacy endpoint was measured after the first treatment cycle, while in the FLASH2 trial patients will be treated for 18 consecutive weeks before the primary efficacy endpoint is assessed. In addition, the overall blinded aggregate response rate remains consistent with what the company reported in November 2025 (48% for all patients that have completed the treatment phase of the study, discussed further below), which is higher than the overall response rate used to design the study (25%), thus our confidence is high that the study will have a positive readout. We anticipate an interim analysis from the study being conducted in the second quarter of 2026 and topline results being reported in the second half of 2026.
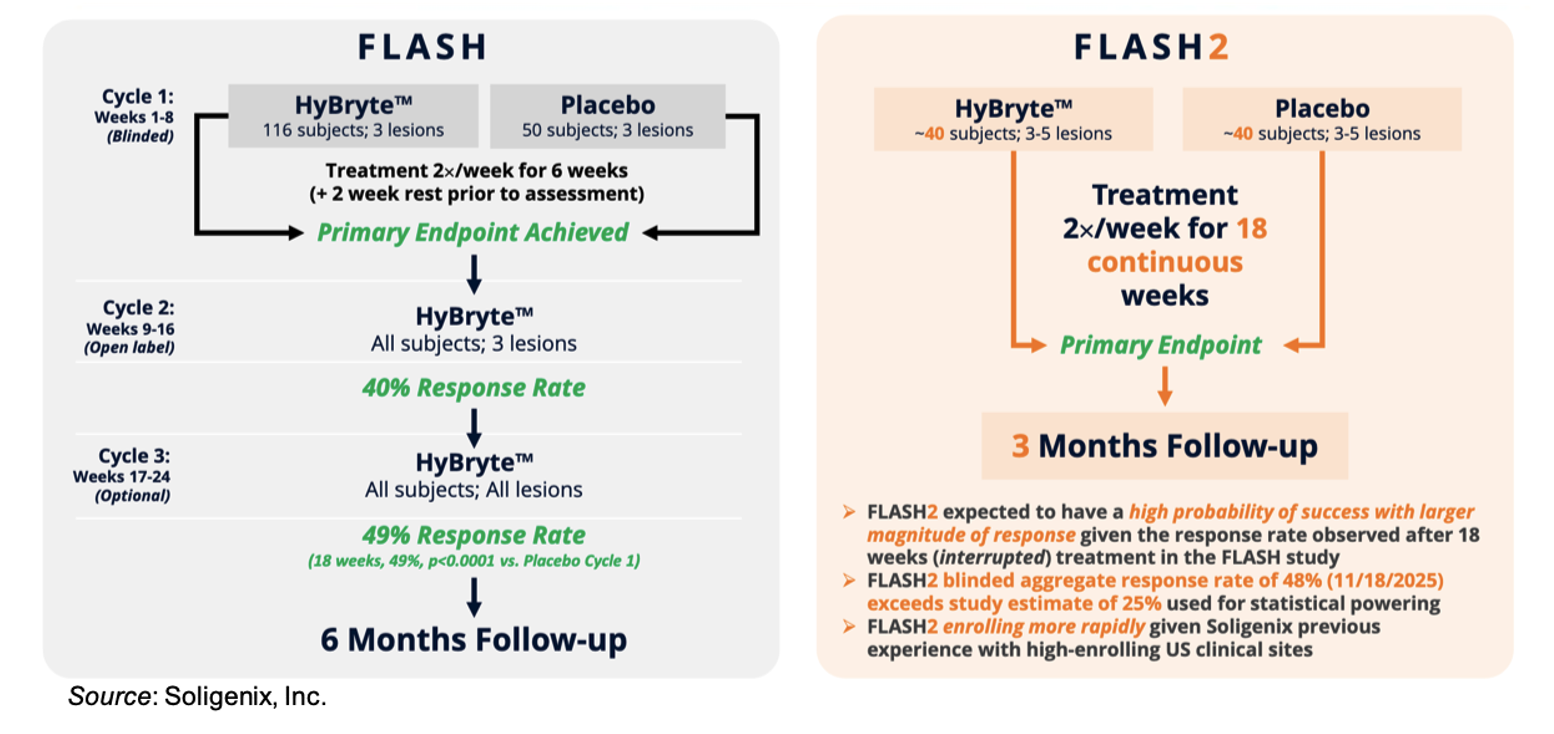
Soligenix powered the FLASH2 trial using an anticipated overall blinded study response rate of 25%, which included a very conservative 40% response rate in the HyBryte arm and a 10% response rate in the placebo arm through 18 weeks. Assuming an even number of patients were enrolled into each cohort, the 25% is calculated by combining the two response rates ((40%+10%)/2). Thus, in order for the blinded response rate to be 48%, we believe the treated cohort would need to be far exceeding the powering assumptions so long as the placebo response rate is not unusually high.
If the two cohorts in the blinded response rate analysis were equal, and the placebo response rate was 10% (the same that was used in the powering assumptions for the trial), that would mean the active response rate would be 86% ((86%+10%)/2=48%)! Thus far, Dr. Ellen Kim has seen a response rate of 75% after 18 weeks of treatment in the open-label, investigator-initiated study currently being conducted at the University of Pennsylvania, so 86% would even far exceed that. While the numbers may not be exact, the point of this exercise is to show that for the blinded response rate to be 48%, assuming there have been a relatively equal number of patients that have finished treatment from each cohort and unless the placebo response is drastically higher than previously seen, the active arm is likely exhibiting a very robust response rate. Of course, we will not know the details until the data are unblinded, however that update gives us a lot of confidence that the trial is at the very least trending in the right direction.
Orphan Drug and Promising Innovative Medicine Designations for SGX945
Recently, Soligenix has announced that SGX945 (dusquetide) has received the following designations from European and UK regulatory agencies:
- In February 2026, Soligenix announced that the European Medicines Agency (EMA) Committee for Orphan Medicinal Products (COMP) gave a positive recommendation on the company’s request for orphan drug designation for SGX945 for the treatment of Behcet’s Disease. The European Commission will then need to ratify the positive opinion. Orphan drug designation by the EMA provides a 10-year period of marketing exclusivity in the EU following approval. In addition, there are incentives for companies seeking protocol assistance from the EMA during development along with direct access to the centralized authorization procedure.
- On March 10, 2026, Soligenix announced that the Medicines and Healthcare Products Regulatory Agency (MHRA) in the UK granted Promising Innovative Medicine (PIM) designation to SGX945 for the treatment of Behcet’s Disease. This is the first step and a prerequisite towards inclusion in the UK Early Access to Medicines Scheme (EAMS), which offers severely ill patients with life-threatening conditions and seriously debilitating conditions the ability to try ground-breaking new medicines much earlier than they would normally be able to. To meet PIM designation, a product must meet three criteria, including treating a life-threatening or seriously debilitating disease with a high unmet medical need, the product needs to offer a major advantage over therapies that are currently used in the UK, and the potential adverse effects of the product are likely to be outweighed by the potential benefits.
Conclusion
In addition to the readouts from the Phase 3 FLASH2 trial expected this year, we also anticipate additional scientific publications around HyBryte this year along with updates for the company’s Behcet’s disease program for SGX945 (dusquetide) and the psoriasis program for SGX302. Soligenix is planning to reformulate SGX945 to enable home-based treatment before interacting with regulators regarding a follow-on Phase 2b trial later in 2026. The company will also be planning follow-up studies for the psoriasis program following the release of encouraging topline results from the Phase 2a trial of SGX302 in mild-to-moderate psoriasis. Lastly, the company is continuing to engage in partnering discussions for ex-U.S. markets for HyBryte in CTCL in order to pursue marketing authorizations worldwide. With no changes to our model, our valuation remains at $25 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.