By David Bautz, PhD
NASDAQ: TNXP
READ THE FULL TNXP RESEARCH REPORT
Business Update
Encouraging Initial Data from Tonmya™ Launch
Following the approval of Tonmya™ (cyclobenzaprine HCl sublingual tablets) for the treatment of fibromyalgia in adults, Tonix Pharmaceuticals Holding Corp. (NASDAQ: TNXP) initiated the commercial launch of the drug on November 17, 2025, with an approximately 90-member salesforce. Early prescription trends from the launch through December 31, 2025, include:
- More than 1,500 healthcare providers have prescribed Tonmya to patients
- ~2,500 patients have initiated treatment with Tonmya
- Cumulative total prescriptions totaled approximately 4,200. This number includes bridge prescriptions that are facilitated through Tonix’s specialty pharmacy channel. Bridge prescriptions are initial patient fills provided while coverage determinations are ongoing and do not immediately generate net product revenue.
These are very encouraging numbers for the initial launch of the drug, especially considering this was over both the Thanksgiving and Christmas holidays, which are known to depress pharmaceutical sales. Tonix continues to focus on expanding payer engagement, establishing contracts with commercial payers, and continuing discussions with Medicare and Medicaid.
TNX-4800 Development Plan
Tonix is developing TNX-4800 (formerly 2217LS), a long-acting, human monoclonal antibody (mAb) prophylactic treatment for the prevention of Lyme disease. It is being developed for annual seasonal use as a single subcutaneous administration in the spring to protect against Lyme disease through the entire tick season in the U.S. There are currently no FDA-approved vaccines or prophylactics to protect against Lyme disease.
Tonix will be meeting with the U.S. Food and Drug Administration (FDA) this year to explore Phase 2 development and registration options for TNX-4800. The company currently expects to have GMP drug product available for clinical testing in early 2027. A Phase 2 field study is likely to begin in 2027, with the potential for a Phase 2 controlled human infection model (CHIM) study, also called a human challenge study, likely to take place in 2028.
Phase 2 Study of TNX-102 SL in MDD to Initiate in Mid-2026
In November 2025, Tonix announced that the U.S. FDA cleared the Investigational New Drug (IND) application for the development of TNX-102 SL 5.6 mg for the treatment of major depressive disorder (MDD) in adults. The company is planning to conduct a potentially pivotal Phase 2 trial that will be a six-week, randomized, double blind, placebo-controlled study of TNX-102 SL as a first-line monotherapy. Approximately 360 patients will be enrolled across 30 sites in the U.S. The primary endpoint of the study will be the MADRS total score change from baseline at Week 6, with secondary endpoints that include global impression scores, anxiety ratings, and measures of sleep disturbance. Startup activities have begun, and we expect the trial to initiate enrollment in mid-2026.
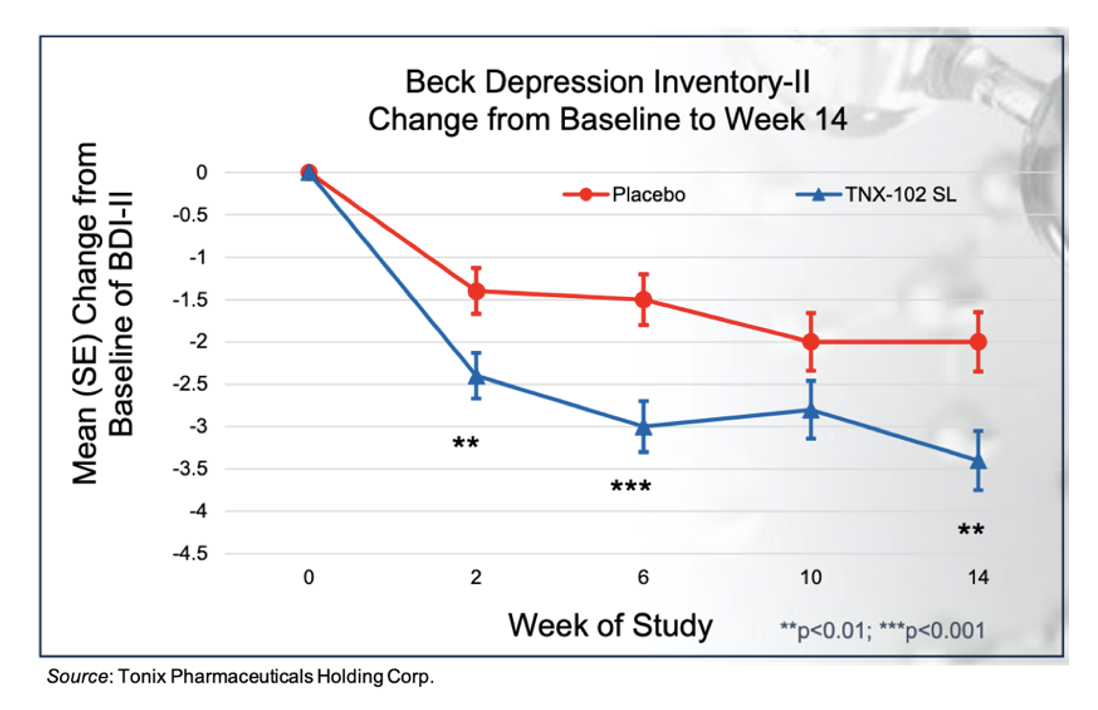
The support for evaluating TNX-102 SL as a treatment for depression stems from results of TNX-102 SL in fibromyalgia and post-traumatic stress disorder (PTSD) clinical trials. For example, in the RESILIENT Phase 3 trial, there was a greater reduction in the total Beck Depression Inventory-II score in the TNX-102 SL cohort compared to placebo at Week 14 with an effect size of 0.27, as shown in the following figure.
TNX-2900 Trial in Prader-Willi in 1Q27
Tonix is developing TNX-2900 (intranasal potentiated oxytocin) as a treatment for Prader-Willi syndrome, a rare genetic disorder that is characterized by insatiable appetite, obesity, and behavioral problems. Tonix is planning to initiate a Phase 2 trial of TNX-2900 in children and adolescents with Prader-Willi syndrome in the first quarter of 2027. TNX-2900 has Orphan Drug Designation (ODD) as well as Rare Pediatric Disease designation, which makes the company eligible for a Priority Review Voucher (PRV) upon approval. PRVs are fully transferable and have sold previously for $150 million.
Financial Update
On March 12, 2026, Tonix announced financial results for the fourth quarter and full year that ended December 31, 2025. Net revenue from product sales for the full year 2025 was approximately $13.1 million, compared to $10.1 million in 2024. Net revenue from the sales of Zembrace, SymTouch, and Tosymra was approximately $11.7 million, compared to $10.1 million in 2024. Net revenue from the sales of Tonmya from the date of launch (Nov. 17, 2205) to December 31, 2025, was approximately $1.4 million. Cost of sales for 2025 was approximately $6.6 million, compared to $7.8 million in 2024. R&D expenses for 2025 were $44.5 million, compared to $40.0 million in 2024. The increase was primarily due to pipeline prioritization and increased headcount. SG&A expenses in 2025 were $87.7 million compared to $40.1 million in 2024. The increase was primarily due to spending on sales and marketing related to Tonmya, along with increased headcount.
Tonix exited 2025 with approximately $207.6 million in cash and cash equivalents. In December 2025, the company completed a $20 million registered direct offering with Point72 Asset Management. Subsequent to the end of the year, the company raised $8.6 million using its at-the-market (ATM) facility. We estimate that the company’s current cash position is sufficient to fund operations into the first quarter of 2027. As of March 11, 2026, the company had approximately 13.4 million shares outstanding and, when factoring in stock options and warrants, a fully diluted share count of approximately 15.2 million shares.
Conclusion
We’re very encouraged by the initial prescribing numbers for Tonmya. We had only estimated for $0.1 million in revenue for the 4Q25; thus the $1.4 million in net revenue was well ahead of our estimate. For 2026, we estimate for Tonmya net revenues of $17 million. A large number of physicians have begun prescribing Tonmya, and we fully anticipate this momentum to continue in 2026 as the commercial launch of Tonmya proceeds. With no changes to our model, our valuation remains at $64 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.