By M. Marin
NASDAQ:QNRX
READ THE FULL QNRX RESEARCH REPORT
Quoin Pharmaceuticals (NASDAQ:QNRX), a late clinical stage, specialty pharmaceutical company focused on developing treatments for rare and orphan diseases, is advancing lead asset QRX003 and has generated positive data from ongoing studies. QRX003 has multiple regulatory designations in place that provide economic and potentially timeline benefits and others under review. In preparation of commercializing QRX003 and potentially other assets in its pipeline designed to treat multiple dermatological indications, the company has established a worldwide sales infrastructure.
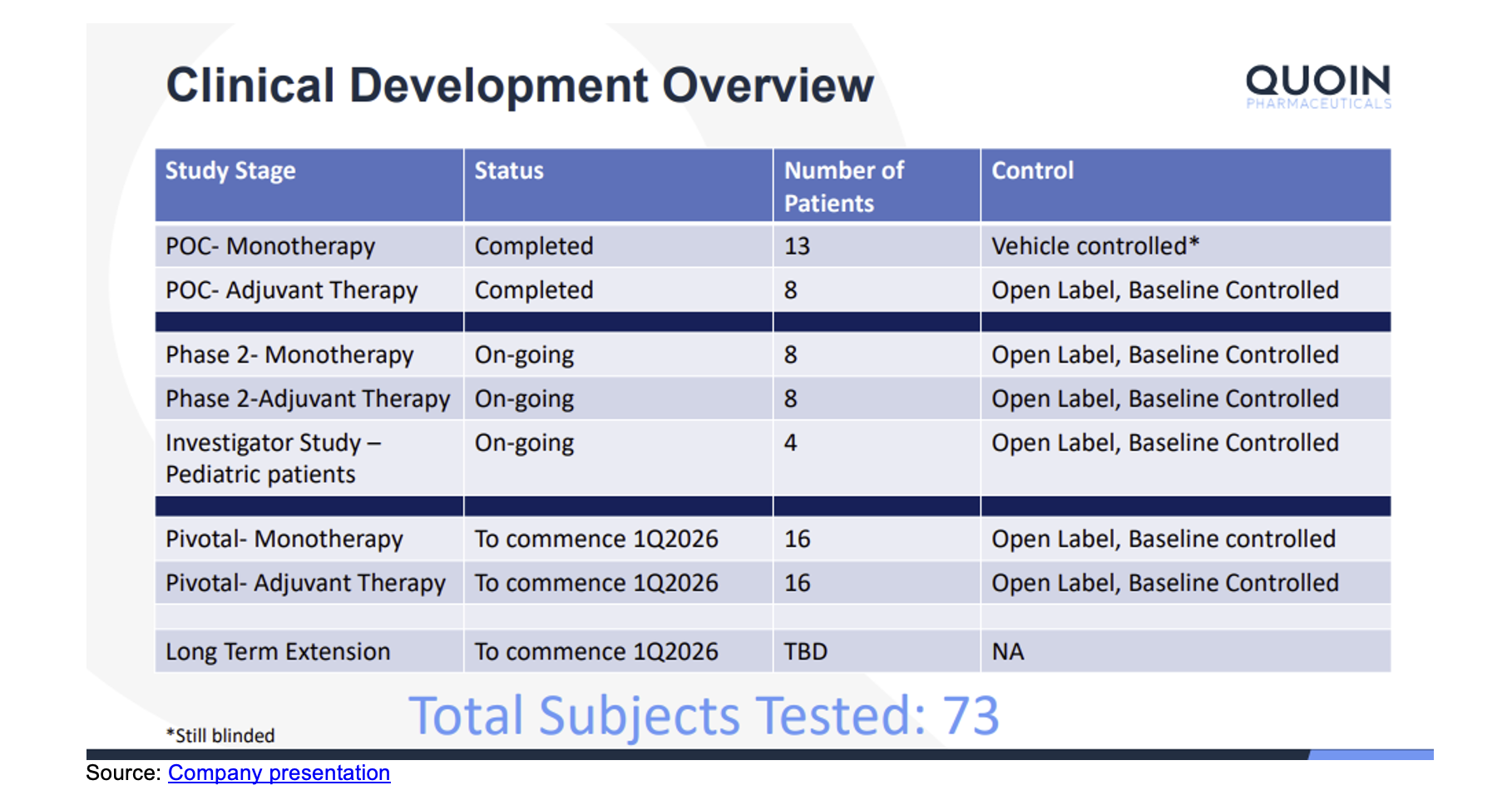
QRX003 is being evaluated for the treatment of Netherton Syndrome (NS), a Netherton Syndrome is a debilitating skin disorder, pediatric NS and Peeling Skin Syndrome (PSS). There are no approved NS treatments currently and Quoin is optimistic about QRX003’s prospects to be the first approved treatment for NS. QRX003 is a topical lotion with a broad-spectrum serine protease inhibitor that has demonstrated the ability to significantly downregulate the hyperactivity of the kallikreins in the skin that are responsible for the excessive skin shedding that is associated with NS and other dermatological diseases. QRX003 is being assessed in two late-stage whole-body pivotal clinical trials in patients with NS. Early data has supported the benefits of QRX003 and top-line data is expected in 2H 2026.
QRX003 has multiple regulatory designations
Once QRX003 and potentially other assets are commercialized, Quoin targets three core markets for commercialization through a proprietary sales network - the U.S., E.U, and Japan - and has multiple regulatory designations for QRX003 in place and/or under review in these and other regions. Specifically, the FDA and European Medicines Agency (EMA) have granted QRX003 Orphan Drug Designation (ODD) for the treatment of NS. Following a meeting with Japan’s regulatory authority, the Japanese Ministry of Health, Labour and Welfare (MHLW), Quoin recently submitted an application for Orphan Drug Designation for QRX003 for the treatment of NS for that market and is optimistic that it will be granted.
Among other benefits, these designation provide economic incentives or credits and potentially can accelerate the timeline for attaining regulatory approval and market exclusivity for a period if QRX003, if approved. The drug candidate has also been granted FDA Rare Pediatric Disease (RPD) Designation for the treatment of NS.
The company believes these designations reinforce the potential of QRX003 as a therapeutic candidate as it advances clinical studies towards a potential New Drug Application (NDA). Moreover, the potential for an expedited regulatory pathway is supported by the significant increase in approvals of products to treat rare and orphan diseases in recent years.
Separately, Quoin also filed an application for Breakthrough Medicine Designation for QRX003 with the Saudi Food and Drug Authority (SFDA) for the treatment of NS. Such designation would allow for accelerated regulatory review and could enable earlier patient access and reimbursement in Saudi Arabia, where Quoin has formed a distribution partnership with Genpharm for QRX003 (for Saudi Arabia and other MENA countries), potentially as early as 2H 2026, establishing QRX003 as the first approved treatment anywhere for NS.
NDA submission expected by early 2027; extensive distribution network in place to support potential QRX003 commercialization
Quoin plans to submit a New Drug Application (NDA) in the U.S. and other markets in late 2026 or early 2027. As QNRX moves QRX003 forward in clinical development towards potential regulatory approval and commercialization, The company’s go-to-market plan is to establish a proprietary sales infrastructure for core markets the U.S., E.U. and Japan, as noted, and QNRX has established an extensive distribution network for QRX003 and potentially other assets in its pipeline encompassing at least 61 countries through distribution agreements.
Demonstrating evidence of benefits of QRX003; currently no approved NS treatments
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.